Unit 2 - Biomolecules + Enzymes
1/61
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
62 Terms
Biomolecules make up :
All living things
Carbon color
Black
Nitrogen Color
Blue
Oxygen Color
Red
Hydrogen Color
White
Phosphorus Color
Purple
Sulfur Color
Yellow
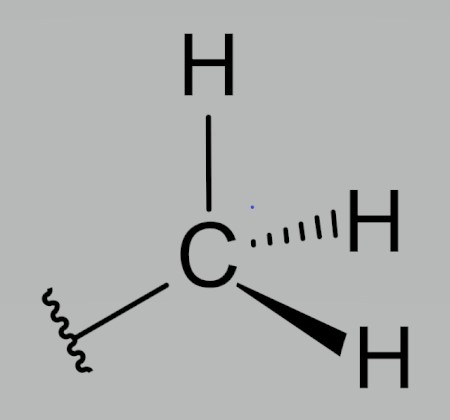
Methyl Group
Hydrophobic
No effect on pH
Alcohol Group
Hydrophilic
No effect on pH
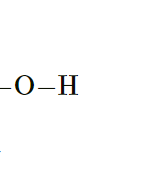
Amino Group
Hydrophilic
Basic
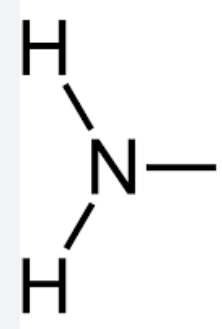
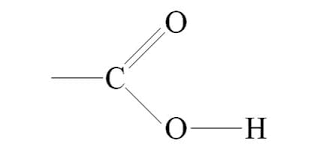
Carboxyl Group
Hydrophilic
Acidic
Phosphate Group
Hydrophilic
Acidic
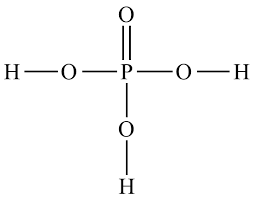
How many bonds does Carbon have?
4
How many bonds does Oxygen have?
2
How many bonds does Nitrogen have?
3
How many bonds does Hydrogen have?
1
The 4 types of biomolecules
Carbohydrates
Proteins
Lipids
Nucleuic Acids
Most types of Biomolecules are
Polymers
What is a polymer?
A long molecule formed by binding many small molecules together.
Polymers are made of
Monomers
Dehydration Synthesis
A chemical reaction that joins monomers together by removing water.
Synthesis
To create
Dehydration
Removing water
Lipids are Hydro-
-phobic
Nitrogenous Bases
C,T,A,U,G
DNA ‘s only job
Permenant genetic information storage
RNA ‘s multiple jobs
temporary information storage
Carrier molecules
Ribozymes
Ribozymes
Cellular machinery that speeds up chemical reactions
Primary Structure (1º)
The sequential order of amino acids in a protein
Encoded by genes in the DNA
What is the only way to change primary structure
To change the DNA
Secondary Structure (2º)
Portions of the protein that fold into specific structures
Alpha helix
Beta pleated sheet
Not all parts of the protein have secondary structure
Tertiary Structure (3º)
The overall 3D shape of the entire protein
Quaternary Structure (4º)
The final shape of a complex made up of multiple problems working as a single unit
Not found in all proteins (optional)
Denaturation
Disruption of Protein Structure : the Protein unwinds and loses its shaoe
Caused by change in: temperature, pH, salt, and attack by toxin
Can sometimes be renatured
Examples of Proteins unfolding (change in temperature)
Cooking and Fevers
Examples of change in pH
Stomach acid
Examples of change in salt
Overhydration or dehydration
Monomers
small subunits
Hydrolysis
Reverse reaction of dehydration synthesis
What is the monomer of Nucleis acids?
Nucleotide
What is the monomer of Carbohydrates?
Simple Sugars
What is the monomer of Lipids?
Fatty acids
What is the monomer of Proteins?
Amino Acids
What bases does RNA use?
A,U,C,G
What bases does DNA use?
A,T,C,G
Chitin
Structural Polymer of glucose
cell walls of fungi
exoskeleten of arthopeta
Cellulose
Structural polymer of Glucose
cell walls of plants
Starch
Storage polymer of Glucose
stores sugar in plants
Glycogen
Storage polymer of Glucose
stores sugar in animals
Phospholipids
Storage polymer of Lipids
Makes up all membranes
Cholestrol
Found in membranes of animals
Steroids
Built from cholestrol
Communication molecules in an organism
Triglycorde
Energy storage in animals
If a protein loses its shape
It cannot function
Some proteins can (opposite of denature)
renature
Name rule for Carbohydrates
end in -ose
Name rule for proteins
end in -in and -ase
If a protein ends in -ase
it is an enzyme
Polymers of carbohydrates
Cellulose, Chitin, Startch, Glycogen
Polymers of nucleic acids
DNA + RNA
Polymers of Lipids
Phospholipids, Cholestrol, Steroids, Triglycoride
Polymers of proteins
Proteins