Development and Plasticity of the CNS: Key Concepts and Processes
1/166
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
167 Terms
What is the source of the human nervous system during embryonic development?
The ectoderm, the outermost tissue layer of the embryo.
What key structure forms in the third week of embryonic development that will become the nervous system?
The neural plate.
What happens to the neural plate by the fourth week of development?
It wraps in on itself to form a hollow neural tube.
What are the potential consequences of mutations during the formation of the neural tube?
Lethal deformities like anencephaly or lifelong disabilities like spina bifida.
What is the anterior part of the neural tube called, and what does it give rise to?
The telencephalon, which expands to form the brain.
What is neurogenesis, and where does it continue throughout life?
The formation of new neurons, continuing in areas like the hippocampus.
What does neurulation refer to in CNS development?
The process of forming the neural tube.
What is neuronal proliferation?
The rapid increase in the number of neurons.
What is neuronal migration?
The movement of neurons towards their targeted sites.
What is synaptogenesis?
The formation of synaptic connections between neurons.
What is myelination in the context of CNS development?
The process of myelinating nerve fibers.
What is the typical progression of CNS development?
Neuronal proliferation, neuronal migration, glial proliferation, and neuronal differentiation.
How do younger neurons migrate during neuronal migration?
They migrate past older neurons using radial glial cells as scaffolding.
What is the role of glial cells in CNS development?
To increase in number to allow for the myelination of fibers.
What is neuronal differentiation?
The process where the function of the neuronal pool becomes more specific.
What analogy is used to describe the stages of CNS development?
Mexican food: neural plate as tostada, neural groove as taco, neural tube as enchilada, and closed ends as burrito.
What part of the neural tube typically impacts disorders related to lack of closure?
The ends of the neural tube, affecting the cerebrum and spinal cord.
What is the significance of synaptic communication in the developing brain?
It establishes functional neural circuits that mediate sensory and motor processing.
What is the time frame for most of the human brain's development?
Within the first 20 years of life.
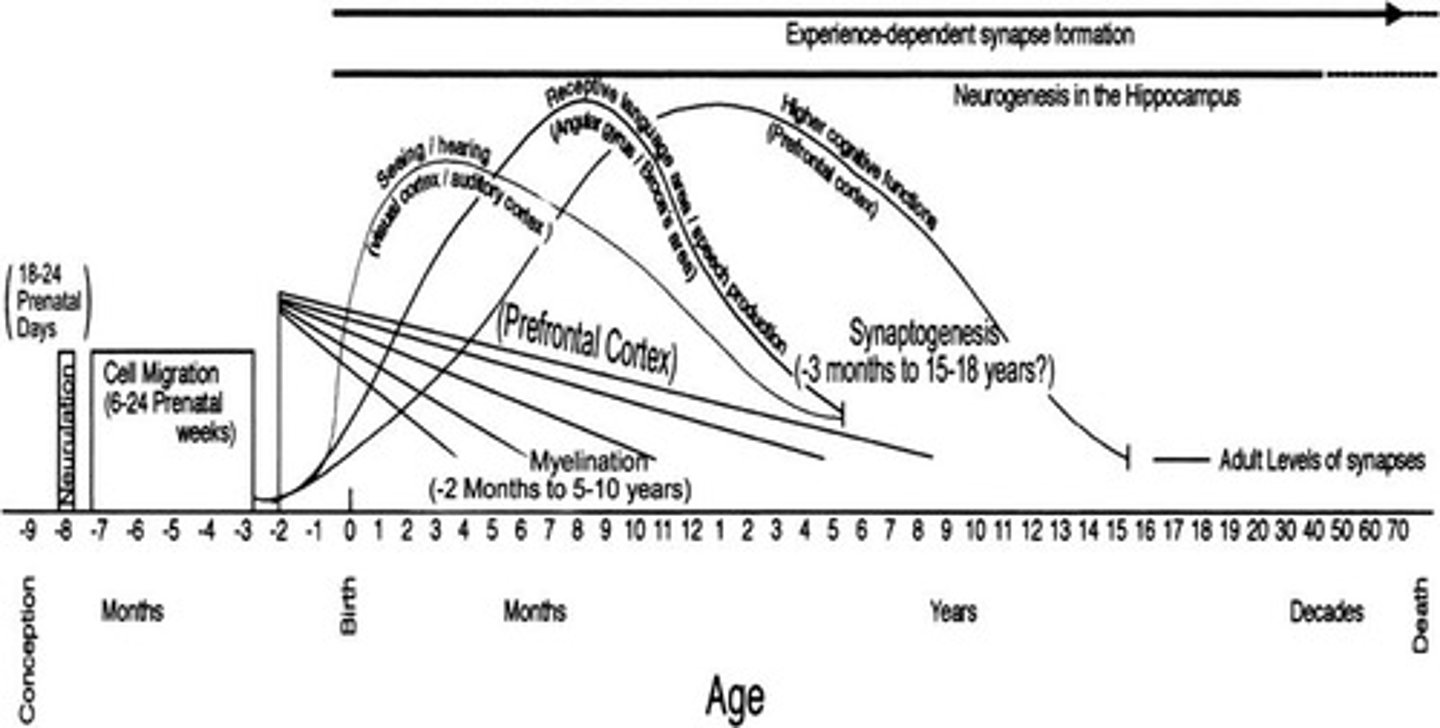
What is the process by which neurons extend axons and dendrites?
Neuronal maturation after reaching their regional positions.
What is the role of the neural plate in the development of the CNS?
It is the source of the majority of neurons and glial cells in the mature human.
What happens to some cells in the neural plate during development?
They stop dividing and differentiate into neurons and glial cells.
What is the significance of the neural tube in relation to the brain and spinal cord?
It gives rise to both structures, making its proper formation crucial.
What is the initial structure that forms the central nervous system?
The neural tube.
What are the three primary sections of the neural tube?
Prosencephalon, mesencephalon, and rhombencephalon.
What happens if the neural tube fails to close in the cephalic region?
It results in anencephaly.
What is the consequence of neural tube closure failure in the caudal region?
It results in myelomeningocele.
At what week does the cephalic portion of the neural tube begin to bend?
At 4 weeks.
What does the alar plate of the prosencephalon develop into?
The cerebral hemispheres (telencephalon).
What does the basal plate of the prosencephalon develop into?
The diencephalon.
What is the role of the optical vesicle in CNS development?
It eventually becomes the optic nerve, retina, and iris.
What are the two classes of neurodevelopmental processes?
Activity-independent mechanisms and activity-dependent mechanisms.
What do activity-independent mechanisms include?
Differentiation, migration, and axon guidance.
What is required for synapse modification and elimination?
Neural activity.
Who formulated the neurotrophic hypothesis?
Victor Hamburger and Rita Levi Montalcini.
What did Hamburger discover about motor neurons and extra limbs?
Implanting an extra limb increased the number of spinal motor neurons by preventing cell death.
What is the neurotrophic hypothesis?
Growing axons compete for limiting amounts of target-derived trophic factors, and those that fail to receive sufficient support die by apoptosis.
What is Nerve Growth Factor (NGF)?
The first trophic factor purified by Rita Levi Montalcini and Stanley Cohen.
What are the three NGF-related trophic factors?
BDNF, NT3, and NT4.
What is the function of Trk proteins?
They act as receptors for NGF and related factors.
What does Trk dimerization and phosphorylation activate?
Various intracellular signaling pathways including the MAP kinase, Akt, and PKC pathways.
What is Ciliary Neurotrophic Factor (CNTF)?
A protein that acts as a survival factor for motor neurons.
What receptor complex does CNTF act via?
A receptor complex that includes CNTFRα, GP130, and LIFRβ.
What is the significance of trophic factors in neuron survival?
They regulate the survival of neurons and prevent apoptosis.
What is the primary outcome of neuronal migration during development?
Neurons move from their origin in the embryo to their final positions.
What is the role of the growth cone in neurodevelopment?
It guides the outgrowth of axons and dendrites towards postsynaptic partners.
What lifelong changes in synapses are thought to underlie?
Learning and memory.
What is the role of JAK kinase in receptor activation?
JAK kinase is recruited upon receptor activation, phosphorylating LIFRβ and STAT proteins, which then regulate gene expression by translocating to the nucleus.
What is GDNF and its significance?
GDNF, or Glial derived neurotrophic factor, is a potent trophic factor for striatal neurons and a member of the TGFb family of proteins.
What is the composition of the functional GDNF receptor?
The functional receptor is a heterodimer composed of type 1 and type 2 receptors.
What occurs when the type 1 receptor of GDNF is activated?
Activation leads to phosphorylation of Smad proteins, which translocate to the nucleus to activate gene expression.
Why are critical periods important in synaptogenesis?
Critical periods are times when neuronal pruning occurs due to competition for growth factors, affecting the normal development of unused processes.
What is the difference between 'critical periods' and 'sensitive periods' in neuronal development?
Critical periods refer to times when specific functions must develop, while sensitive periods indicate times when development is optimal but can still occur later with more effort.
What is the neuromuscular junction (NMJ)?
The NMJ is a well-characterized synapse composed of a motor neuron, myofiber, and Schwann cell, facilitating muscle contraction.
What neurotransmitter is released at the neuromuscular junction?
Acetylcholine (ACh) is released by the motoneuron when it depolarizes.
How does acetylcholine affect the myofiber?
ACh binds to acetylcholine receptors (AChR) on the myofiber's plasma membrane, opening ion channels that lead to muscle contraction.
What role do Schwann cells play at the NMJ?
Schwann cells provide a myelin cover to insulate the junction and may facilitate preliminary signals by increasing spontaneous neurotransmitter release.
What is the origin of the cells at the neuromuscular junction?
Motor neurons arise from the neural tube, myoblasts from the mesoderm, and Schwann cells from the neural crest.
How do axons find their target during development?
Axons are guided by growth cones that actively search for neurotrophins released by the myotube.
What is the pattern of synapse development at the NMJ?
Most muscles are innervated at their midpoints, but this is due to growth from the point of innervation rather than axons specifically targeting the midpoint.
What happens to AChR density during synapse development?
AChR density results from axonal contact rather than causing it, influenced by both myotatic growth and axonal innervation.
What is the nature of the initial synaptic transmission between motoneurons and myotubes?
The initial synaptic transmission is weak but occurs almost immediately after preliminary contact.
What factors contribute to the growth of the myotube after initial innervation?
The myotube grows symmetrically from the point of innervation, influenced by both axonal contact and growth factors.
What is the significance of neurotrophins in neuronal development?
Neurotrophins released by myotubes guide the growth cones of axons to their target destinations.
What is the impact of competition for neural growth factors during critical periods?
Competition can lead to pruning of processes that are not used, affecting normal development later in life.
What is the difference in terminology between authors regarding periods of neuronal development?
Some authors use 'critical periods' to denote strict timelines for development, while others prefer 'sensitive periods' to indicate optimal times for development.
What is the role of myoblasts in the formation of the NMJ?
Myoblasts fuse to form multi-nucleated myotubes, which are essential for the formation of the NMJ.
How does the initial contact between motoneurons and myotubes affect synaptic development?
Initial contact generates weak synaptic transmission, which is crucial for subsequent synaptic development.
What is the function of the growth cone in neuronal development?
The growth cone is a filamentous projection that helps axons navigate towards their target by sensing neurotrophins.
What is formed after about a week following differentiation in muscle cells and motoneurons?
A fully functional synapse.
Why is the pioneer axon important in synapse formation?
It has a high propensity for forming contacts with well-established synapses.
What changes occur during pre-synaptic differentiation at the axon terminal?
Increase in synaptic volume and area, increase of synaptic vesicles, clustering of vesicles at the active zone, and polarization of the pre-synaptic membrane.
What mediates the changes in pre-synaptic differentiation?
Neurotrophin and cell adhesion molecule release from muscle cells.
What is the role of gene expression in pre-synaptic differentiation?
Changes in gene expression and redistribution of pre-existing synaptic components contribute to pre-synaptic differentiation.
What happens to immature synapses at birth?
They are multiply innervated due to new axons innervating pre-existing synapses.
What is synapse elimination?
A process where all axonal inputs except for one retract as the synapse matures.
How does the post-synaptic end plate change during synaptic maturation?
It grows deeper and creates folds through invagination to increase surface area for neurotransmitter reception.
What happens to Schwann cells as synapses mature?
They become dedicated to a single synapse and form a myelinated cap over the entire neuromuscular junction.
What is the presumed nature of synapse elimination?
An activity-dependent process that involves competition between axons.
How do strong synapses affect the release of synaptotrophins?
A strong synapse can trigger the release of synaptotrophins from myonuclei to strengthen and maintain well-established synapses.
What happens to weaker synapses during synapse elimination?
They are starved out and do not receive the same strengthening signals as stronger synapses.
What is the distinction between fast and slow-twitch muscle fibers in motoneuron innervation?
Fast-twitch muscle fibers are innervated by 'fast' motoneurons, while slow-twitch muscle fibers are innervated by 'slow' motoneurons.
What are the two hypothesized paths for motoneuron axon specificity?
One involves active recognition of muscle fibers by axons, and the other involves non-selective guidance by the matrix.
What does the selective path for motoneuron axons entail?
Axons recognize fiber type through specific factors or signals released by the muscle fibers.
What does the non-selective path for motoneuron axons suggest?
Axons are guided to their destinations by the surrounding matrix, without active decision-making.
What can a 'fast' motoneuron potentially do to muscle fibers?
It can convert any muscle fiber into a fast-twitch muscle fiber.
What evidence exists regarding synapse formation specificity?
There is evidence supporting both selective and non-selective paths, indicating a combination of factors in the process.
What is synaptogenesis in the context of the central nervous system (CNS)?
Synaptogenesis refers to the formation of synapses within the CNS, which is a more recent area of study compared to the neuromuscular junction (NMJ).
What are some structural similarities between CNS synapses and neuromuscular junctions (NMJs)?
Both CNS synapses and NMJs have a nerve terminal separated from the postsynaptic membrane by a cleft, localized vesicles at active sites, clustered receptors at the postsynaptic membrane, and glial cells encapsulating the synaptic cleft.
What is a key difference between CNS synapses and neuromuscular junctions?
CNS synapses are strictly neuronal and do not involve muscle fibers, using different neurotransmitters and receptors, while NMJs innervate muscle fibers.
How do inputs to neurons in the CNS differ from those to muscle fibers?
Neurons in the CNS often receive multiple inputs that must be processed and integrated, whereas muscle fibers are innervated by a single input and operate in an all-or-none fashion.
What is CNS plasticity?
CNS plasticity refers to the brain's ability to reorganize itself to meet functional demands, which is particularly beneficial following brain damage.
What factors contribute to CNS plasticity following injury?
Factors include age (younger individuals show greater plasticity), the extent of injury, and the location of damage.
What surgical procedure is mentioned as a treatment for uncontrollable seizures in children with Rasmussen's encephalitis?
Hemispherectomy, which involves removing a cerebral hemisphere where seizure activity is generated.
What is regulatory pruning in the context of CNS development?
Regulatory pruning is the process where neurons in the visual and motor cortices selectively prune connections to appropriate processing centers, refining the circuitry.
What are the two main methods of severing undesired neuronal connections?
Apoptosis, where the neuron is killed along with its connections, and pruning, where axons are retracted from inappropriate synaptic connections without killing the neuron.
What is the believed purpose of synaptic pruning and apoptosis?
To remove unnecessary neuronal structures and improve the networking capacity of the brain as it develops, replacing simpler childhood associations with more complex structures.
How does pruning contribute to brain function maintenance?
Pruning helps maintain efficient brain function by removing neurons based on their synaptic efficiency.
What happens to neurons during apoptosis?
The neuron is killed, and all connections associated with that neuron are eliminated.
What occurs during the pruning process?
The neuron does not die; instead, it retracts axons from synaptic connections that are not functionally appropriate.