CHM 232: Common Infrared Absorption Frequencies
1/33
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
34 Terms
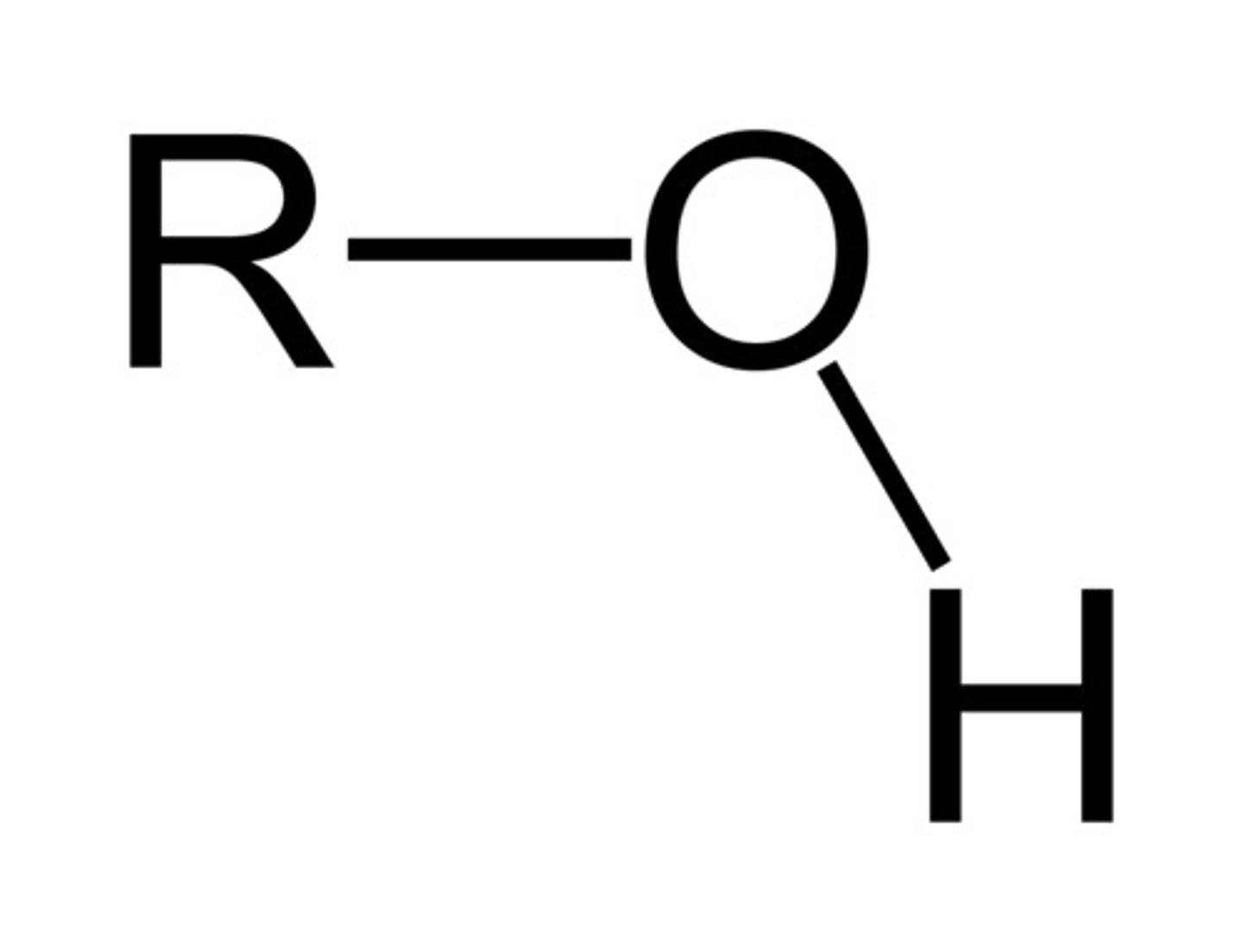
Alcohol
3200-3600 cm^-1
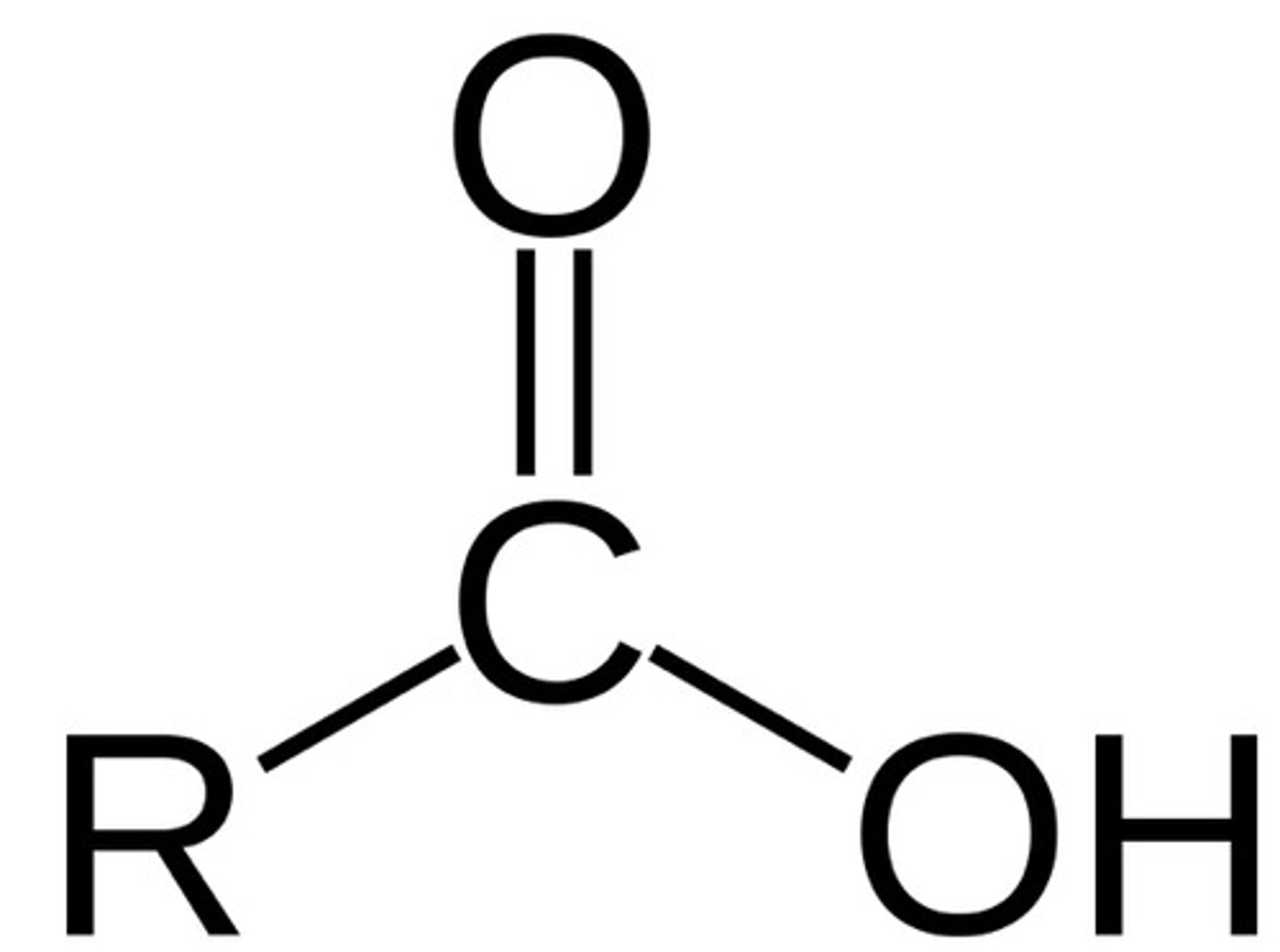
Carboxylic Acid
2500-3600 cm^-1
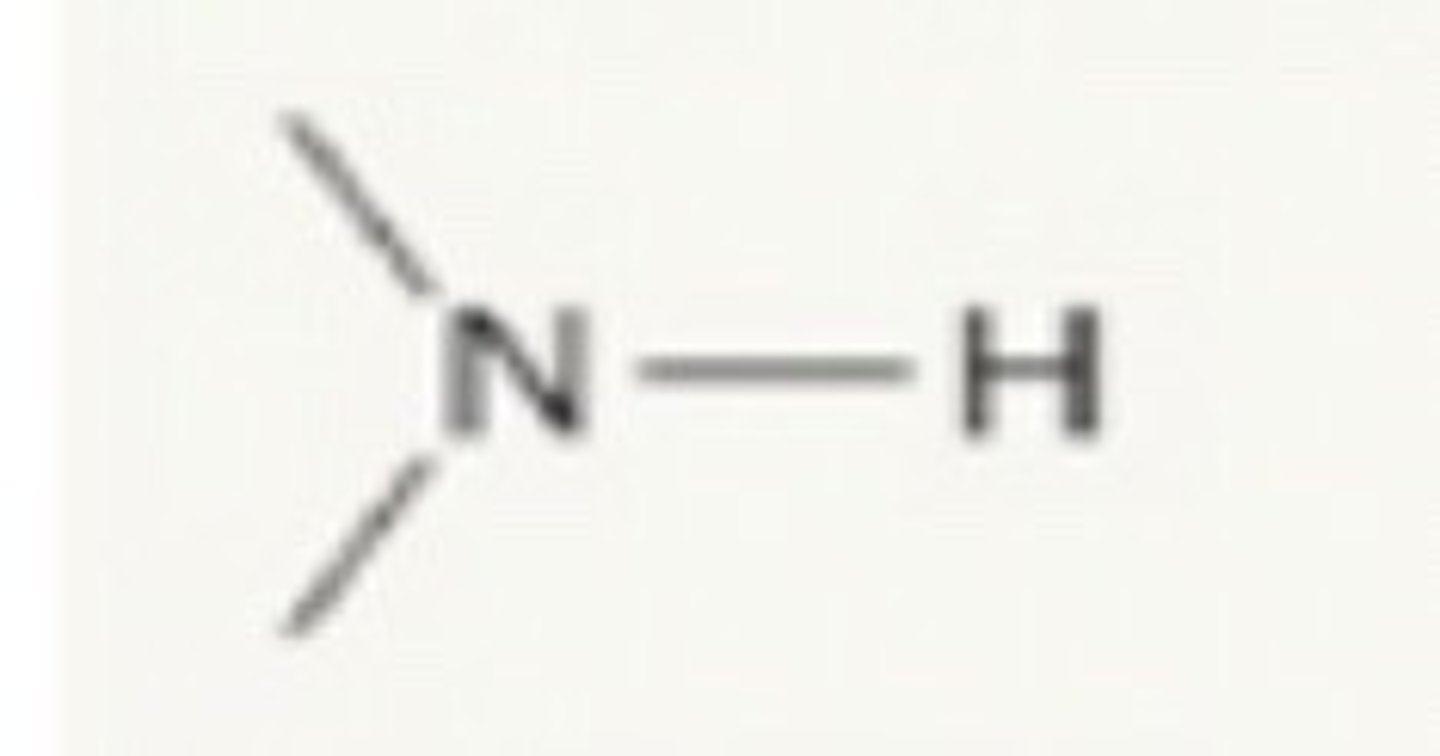
N-H
3350-3500 cm^-1
sp C-H
3310-3320 cm^-1
sp2 C-H
3000-3100 cm^-1
sp3 C-H
2850-2950 cm^-1
sp2 C-O
1200 cm^-1
sp3 C-O
1025-1200 cm^-1
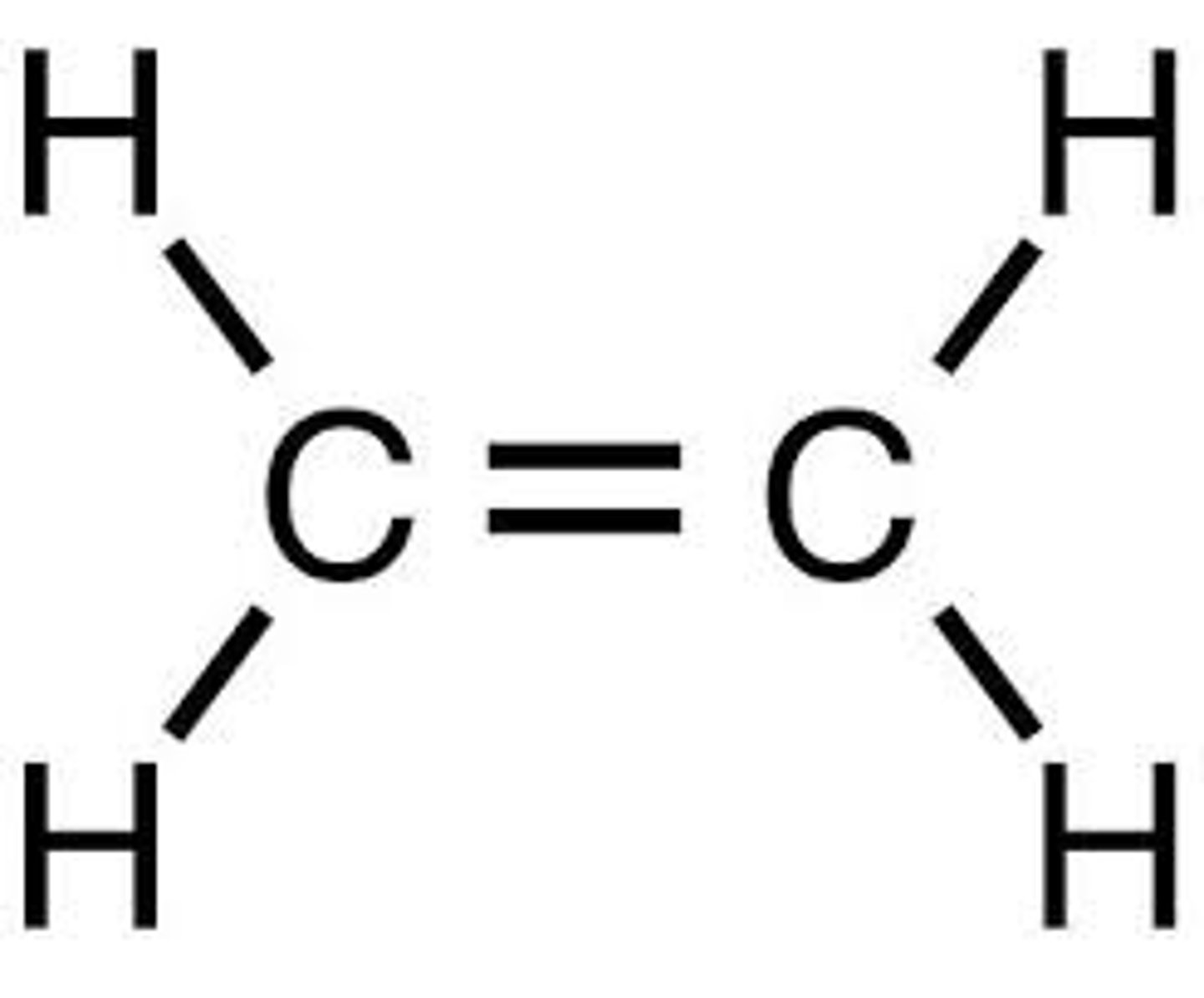
Double Bonds e.g. C=C, C=O
1620-1680 cm^-1
Aldehydes and Ketones
1710-1750 cm^-1
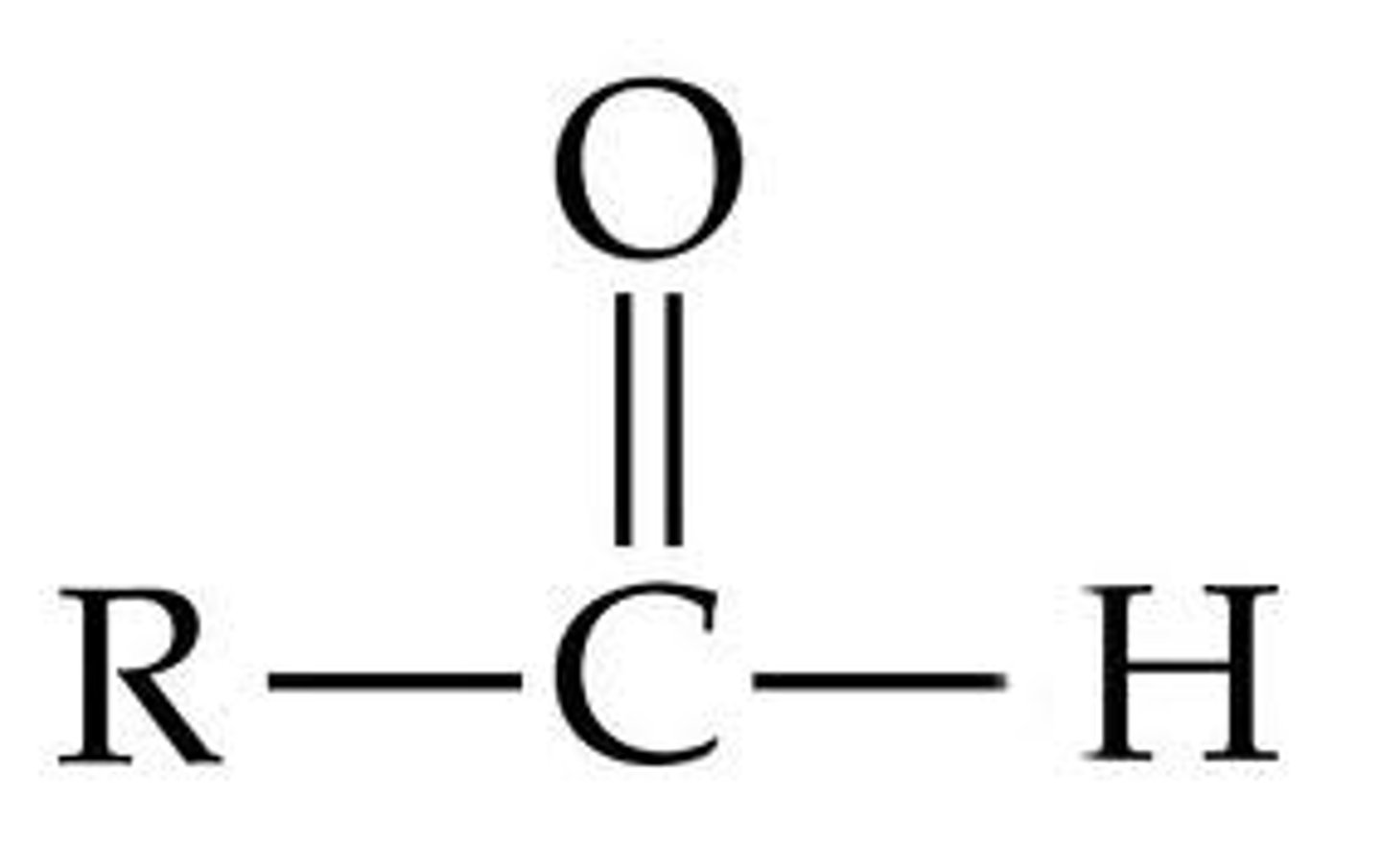
Carboxylic acids (double bonds)
1700-1725 cm^-1
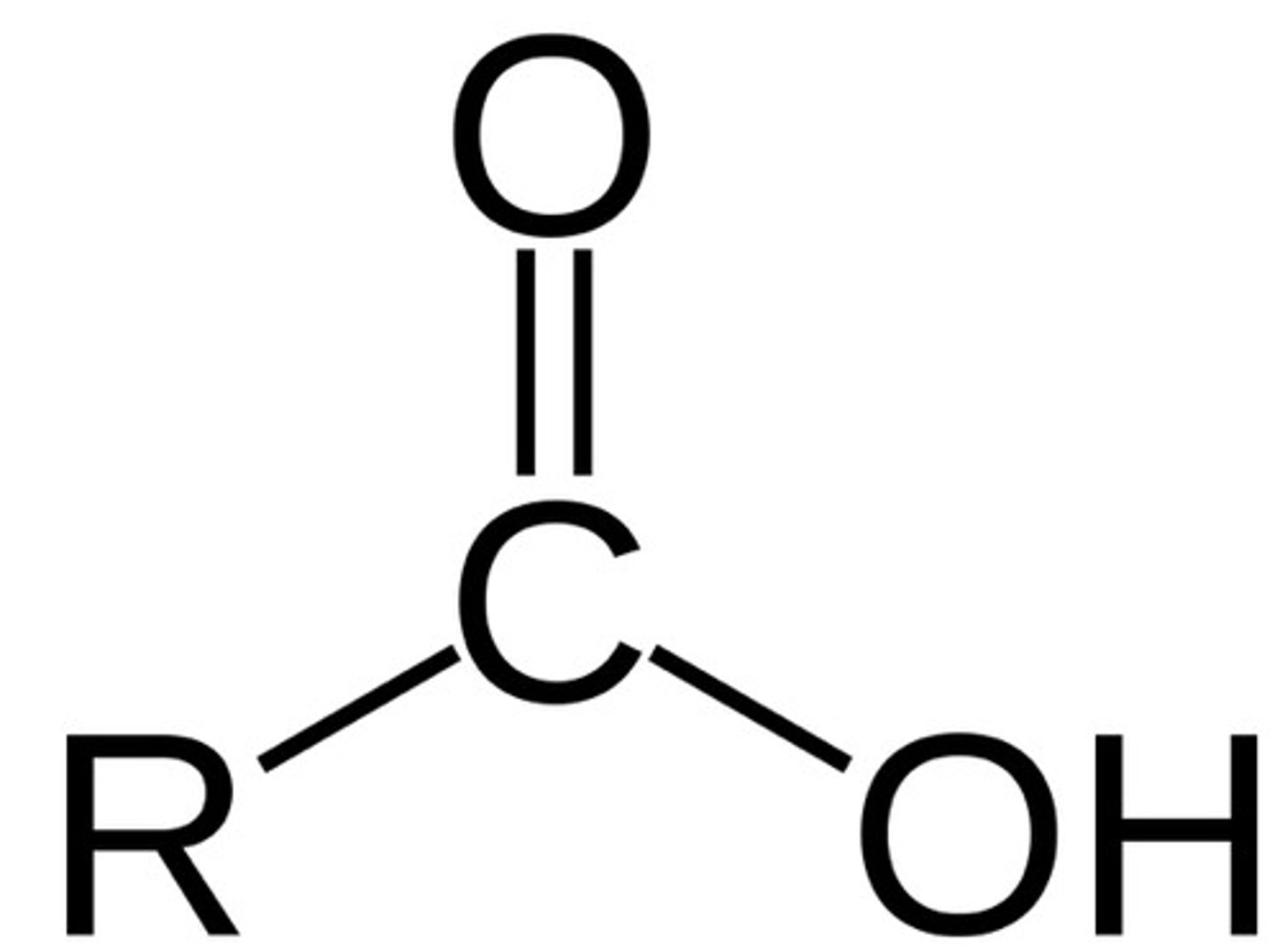
Acid anhydrides
1800-1850 and 1740-1790 cm^-1
Acyl halides
1770-1815 cm^-1
Esters
1730-1750 cm^-1
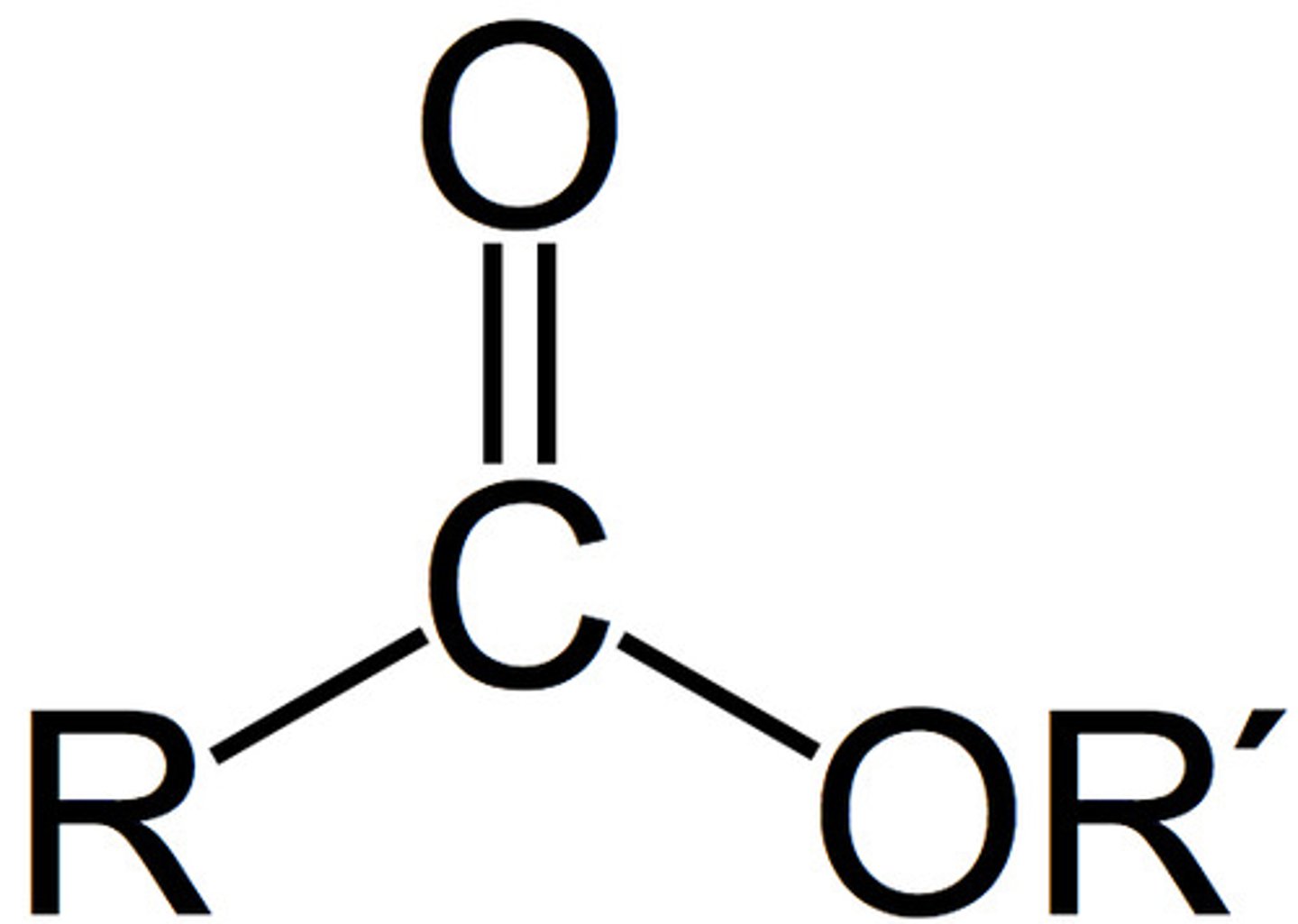
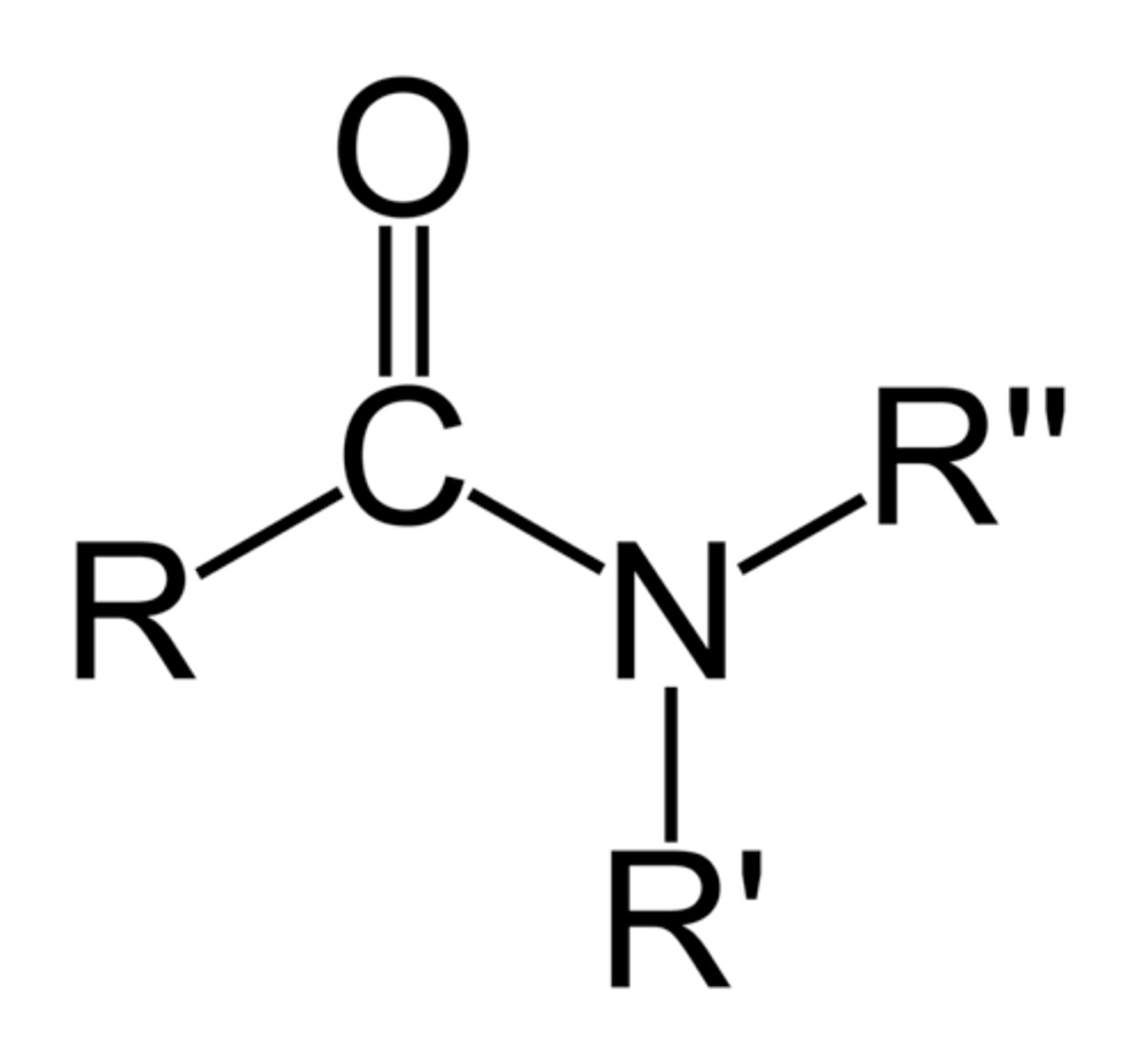
Amides
1680-1700 cm^-1
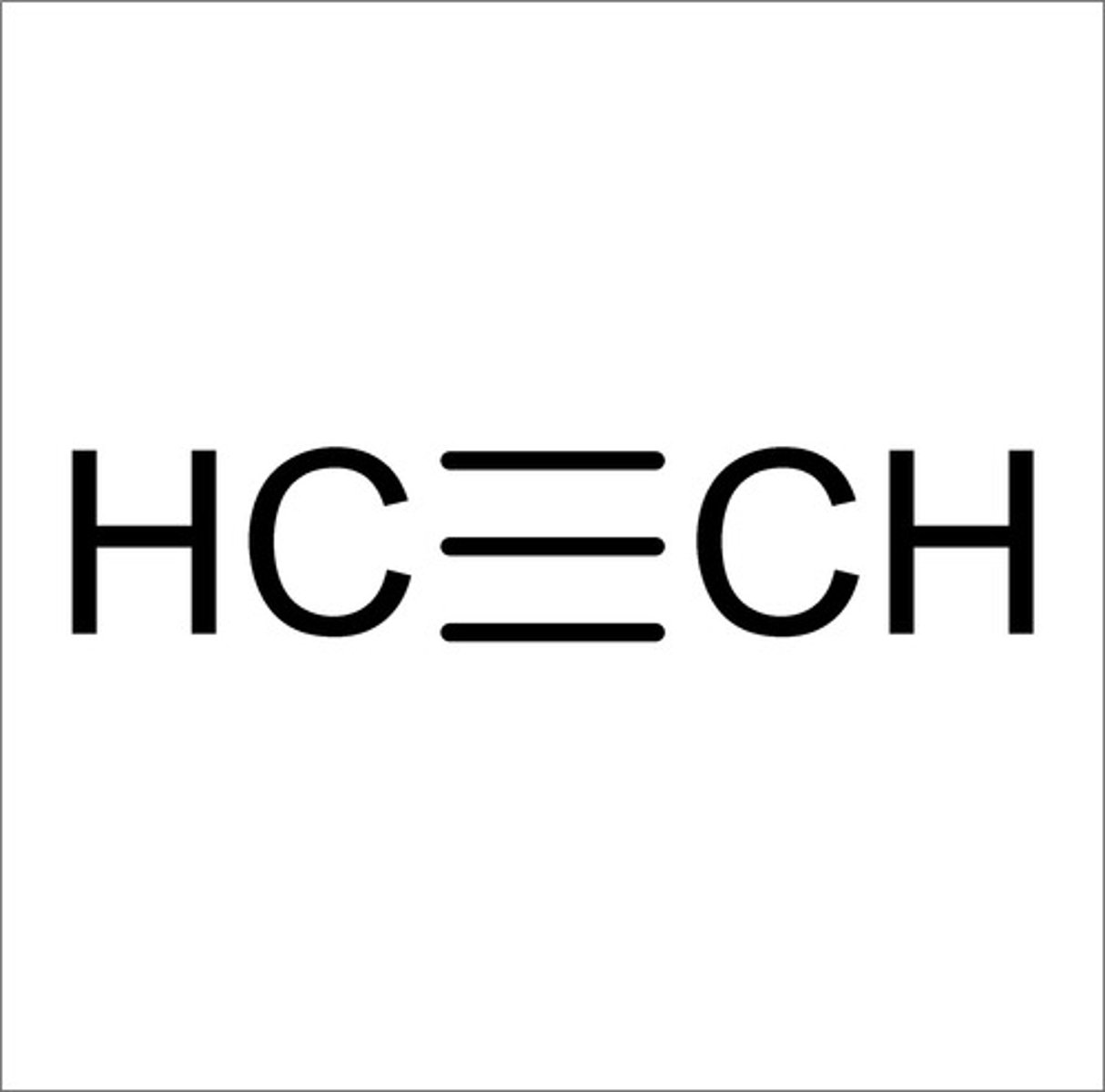
C≡C
2100-2200 cm^-1
C≡N
2240-2280 cm^-1
What are alkenes?
hydrocarbons with double bonds
RCH=CH2
910,990 cm^-1
R2C=CH2
890 cm^-1
cis-RCH=CHR'
665-730 cm^-1
trans-RCH=CHR'
960-980 cm^-1
R2C=CHR'
790-840 cm^-1
Substituted derivatives of benzene: Monosubstituted
730-770 and 690-710 cm^-1
Substituted derivatives of benzene: Ortho-disubstituted
735-770 cm^-1
Substituted derivatives of benzene: Meta-disubstituted
750-810 and 680-730 cm^-1
Substituted derivatives of benzene: Para-disubstituted
790-840 cm^-1
Higher bond strength...
Higher frequency
Higher mass...
Lower frequency
Wavenumber
cm^-1
Reciprocal of the wavelength measured in centimeters (cm); directly proportional to frequency
Used to measure frequency
Higher wavenumber = higher frequency = higher energy vibration
What is Spectroscopy?
Study of the interaction of electromagnetic radiation with matter; typically involves the absorption of electromagnetic radiation
What is Spectrometry?
Evaluation of molecular identity and/or properties that does not involve interaction with electromagnetic radiation
What is Infrared Spectroscopy?
IR spectroscopy is used to determine the functional groups in a substance
Measures the stretching and bending frequencies of covalent bonds that contain a dipole moment
What is Nuclear Magnetic Resonance (NMR) Spectroscopy?
Used to determine the connectivity (C,H framework) of a molecule
Measures the energy associated with spin states of nuclei in the presence of a magnetic field