7.1-7.4- ES 4/7- Equilibrium and Reversible Reactions
1/76
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
77 Terms
Give two uses of chlorine
1. Extract bromine from sea water
2. Make bleaches
Describe how bleach is made
Chlorine gas passed through a cold solution of sodium hydroxide
Sodium hydroxide solution reacts with chlorine- forming sodium chlorate (I)
Give the equation for the formation of bleach
Cl2 (g) + 2NaOH (aq) (reversible reaction sign) NaCl (aq) + NaClO (aq) + H2O
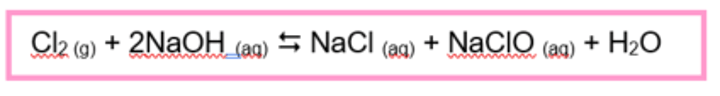
What is chemical equilibria also known as?
Dynamic equilibrium
What does one arrow in a chemical equation indicate, what does a double arrow indicate?
One arrow- the reaction has gone to completion
Double arrow- reversible reaction, it will reach a state of equilibrium
Describe a closed system (4 points)
1. Closed container and its contents
2. Nothing can enter or leave the closed system- mass/ number of molecules remains constant
3. Kept at same pressure/ temperatures the closed system will reach dynamic equilibrium
What sort of a system is needed in dynamic equilibrium?
Equilibrium only applies if the system remains isolated in a closed system so nothing gets in or out- no materials added/ taken away and no other external conditions (e.g. temperature and pressure) are altered
What appears to happen at a macroscopic and microscopic scale when a system is at dynamic equilibrium?
· Macroscopic scale: when at equilibrium nothing appears to change (macroscopic scale- scale you can see)
· Microscopic scale the individual atoms continue to move from one state to the other constantly- system is actually dynamic and in constant movement
Describe how liquid bromine in a closed tube can reach dynamic equilibrium
· Bromine in bottle- bromine present in two states- liquid and gas above the liquid (both diatomic)
· Standing at steady temp for some time- the depth of the orange colour above the liquid remains constant
· Mass of bromine that is a gas and the mass of bromine that is a liquid in a closed system remains constant
· The system is at equilibrium and nothing appears to change in the macroscopic scale (scale you can see)
· If you could see microscopic scale (individual molecules)- in gas molecules constantly moving rapidly in random directions and so collide with the molecules on the surface of the liquid
· Some of these molecules that collide bounce back into gas phase and some entre liquid phase
· Same time molecules of bromine in the liquid are constantly moving around colliding with each other
· Near surface of the liquid some of these molecules escape into the gaseous phase
There are molecules both entering and leaving the liquid phase- a reversible change
Exam Question: Define dynamic equilibrium
Dynamic equilibrium is established in a closed system (where nothing is added or taken away) when;
· Forward and reverse reactions are both happening (so it is dynamic)
· The rate of the forward reaction is the same as the rate of the reverse reaction so reactants are turning into products at the same rate as products are turning into reactants
· The concentrations of the reactants and products remain constant (but concentrations of reactants and products may not be the same as each other)
In a chemical equation which direction is the forwards reaction and which is the reverse, which are called the products/ reactants?
· Reaction left to right is the forwards reaction, reaction right to left (opposite) is the reverse reaction
· Reaction is reversible but substances on left still called reactants and substances on right still called products
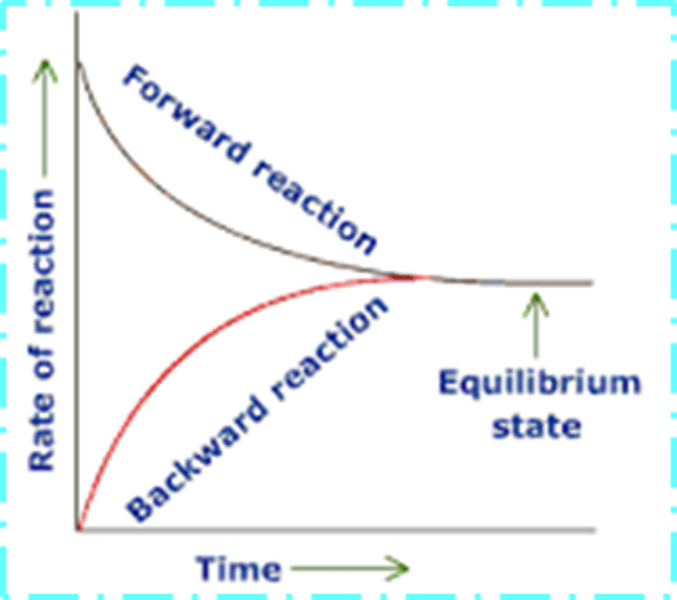
Describe and explain what happens to the rate of the forwards reaction during a reaction to reach dynamic equilibrium?
· The rate of the forward reaction decreases as the reactants are used up
· Rate of the forwards reaction starts high when there are high concentrations of reactants, as the concentration of reactants decreases the rate of reaction decreases- fewer molecules to collide and react
Describe and explain what happens to the rate of the reverse reaction during a reaction to reach dynamic equilibrium?
· The rate of the reverse reaction increases as the products are made
· Rate of the backwards reaction starts low when there are low concentrations of products, as the concentration of products increases the rate of reaction increases- more product molecules to collide and react
Describe what happens to the rate when the system reaches dynamic equilibrium
The system reaches dynamic equilibrium when the rate of the forward and reverse reactions are equal, once final rate is reached it does not change
How do you calculate the rate of reaction from a graph?
The gradient of the graph= the rate- draw a tangent on the graph to calculate it= change in concentration/ time
What factor must be kept constant in order for the rate of reaction to reach a state of equilibrium?
Temperature (affects kinetic energy of the particles)
Draw a rate-time graph of the forwards and backwards reactions up to equilibrium?
What happens to the concentration of the reactants and the products at equilibrium, how is this shown on a concentration- time graph?
All concentrations become constant at equilibrium, shown by the levelling off then horizontal lines on a concentration-time graph
Is the concentration of the reactants and the products exactly the same at equilibrium?
No- concentrations are not the same they are just constant
Why do the concentrations of the reactants and the products remain constant at equilibrium?
Because the forwards and backwards reactions occur at the same rate
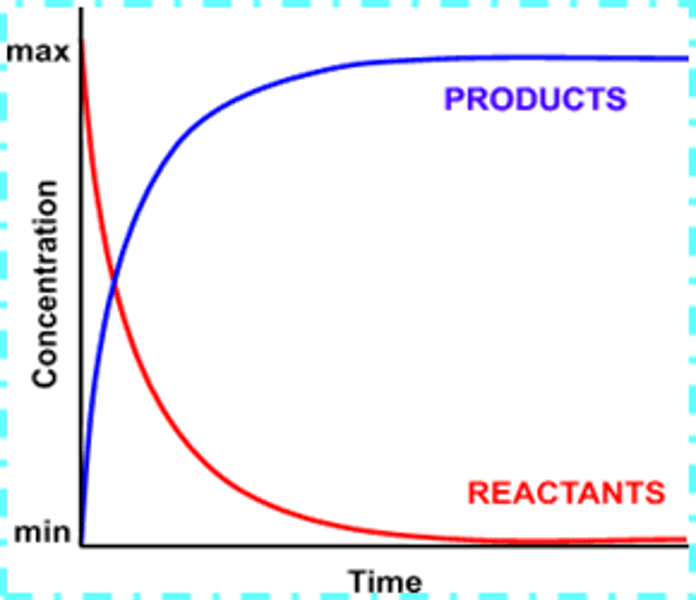
Describe the conversion in a non-equilibrium/ non-reversible reaction and draw a graph
This reaction goes to completion because there are not reactants remaining- In an ordinary reaction; all reactants end up as products; there is 100% conversion
On graph- the concentration of the reactants is 0 at the end which shows it has gone to completion
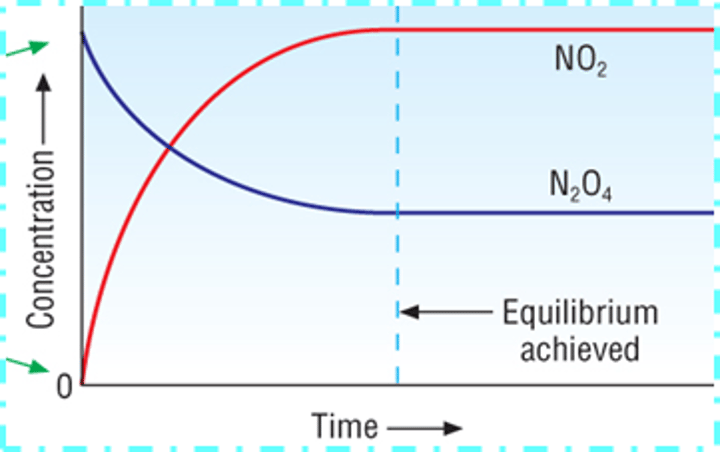
Describe the conversion in an equilibrium reaction and draw a graph, at which point on the graph is equilibrium achieved?
· In an equilibrium reaction, not all the reactants end up as products; there is not a 100% conversion.
· BUT IT DOESN'T MEAN THE REACTION IS STUCK IN THE MIDDLE
· Point when the two lines levels off- when equilibrium is achieved
Exam Tip: What key word must be used when describing the "location" of the equilibrium?
Say POSITION of equilibrium not just equilibrium
Exam Tip: Which principle should you link all equilibrium questions back to?
Always link your answer to Le Chatelier's Principle and state according to Le Chatelier's Principle...
What sort of reactions are acid-base titrations?
Equilibrium reactions
Exam Tip: When talking about the observations made during an reaction which reached equilibrium what key thing should you discuss?
The colour changes- indicates more or less products
What does the position of equilibrium tell you?
How much products/ reactants you will have at equilibrium
What does it mean if equilibrium lies to the left/ right?
1. If the equilibrium lies to the left you'll have more reactants than products
2. If the equilibrium lies to the right you'll have more products than reactants
Which three factors affect the position of equilibrium?
1. Concentration of the reactants or products
2. Pressure in reactions with gases
3. Temperature
Which principle is used to predict changed in equilibrium in response to change?
Le Chatelier's Principle
Define Le Chatelier's Principle
Le Chatelier's principle states that when a system in dynamic equilibrium is subjected to change, the position of equilibrium will shift to minimise change/ oppose the change.
When a system encounters a change what 6 steps are taken to return to equilibrium?
1. Concentration/ change in the system
2. System opposes the change
3. System changes to decrease the change
4. Rate of one of the reactions increases as there is a greater concentration of it
5. More of products/ reactants made
6. Position of equilibrium has shifted to the left/ right
Describe what happens if the concentration of reactants increases?
Increasing the concentration of reactants: The system opposes the change by decreasing the concentration of the reactant by removing it so the position of equilibrium shifts to the right-hand side (away from the reactants) forming more products
The rate of the forwards reaction increases as there are more reactants to collide and react
Describe what happens if the concentration of reactants decreases?
Decreasing the concentration of the reactants causes the equilibrium position to shift further towards the side with the reactants to counter the decrease
Rate of the forwards reaction decreases- fewer molecules to collide
Describe what happens if the concentration of products increases?
Increasing the concentration of a product causes the position of equilibrium to move in the direction that decreases this increased product concentration
The system opposed the change by decreasing the concentration of the products by removing it, the position of equilibrium will move to the left-hand side forming more reactants
The rate of the backwards reaction increases as there are more products molecules to collide and react
Describe what happens if the concentration of the products is decreased?
Decreasing the concentration of the products causes the equilibrium position to move to the product side
The rate of the reverse reaction decreases as there are fewer molecules to divide
What must be present in the equilibrium mixture in order for pressure changes to affect the position of equilibrium?
Changing the total pressure of a system will only change the position of equilibrium if gases are present- causes a shift in the position of equilibrium to oppose the change
Which side of the reaction is at the greatest pressure?
The side of the equation with more gaseous moles is the side at the higher pressure
What happens if the pressure of the system is increased?
Increasing the total pressure of the system causes the position of equilibrium to move to the side with fewer gas molecules as this will decrease the pressure and counter the increase
What happens if the pressure of the system is decreased?
Decreasing the total pressure of the system will cause the position of equilibrium to move to the side with the greater number of as molecules as this will increase the pressure
What does increasing the concentration of one of the gases in the system have the same effect as?
Same effects as if the pressure was increased
Where should the position of equilibrium aim to be for industrial processes?
As far to the right as possible- as many products as possible
What happens if the pressure of a system is increased but there are the same number of gaseous moles on their side of the equation?
The position of equilibrium does not change
If the gas was coloured and it is compressed the colour will still get marker and the same number of colourless molecules are contained within a smaller area but the amount of product/ react will not change
What does changing the temperature of a system affect other than the position of equilibrium?
Changing the temperature will also change the rate- kinetic energy of the molecules changes
Exam Tip: If stating that "the reaction is exothermic" what else must you also say?
The direction it is exothermic in- the forwards reaction is exothermic (if you just say exothermic this could go both ways)
If the forwards reaction is exothermic (- x delta H) what will the reverse reaction be?
The reverse reaction will be endothermic and will have the same magnitude of delta H with a different sign- Links to Hess' Law
What happens if the temperature of the system is increased?
Increasing the temperature of the system causes the position of equilibrium to shift in the endothermic direction (delta positive)/ favours the endothermic (forward/ reverse) reaction so the system absorbs more heat energy and decreases the temperature, the system opposes the change by taking in heat and the position of equilibrium shifts in the necessary direction
What happens if the temperature of the system is decreased?
Decreasing the temperature of the system causes the position of equilibrium to move to increase the temperature, the system opposes the change by releasing heat energy and the position of equilibrium shifts in the exothermic direction
When +/- delta H is written by a reaction is this referring to the forwards or reverse reaction?
Forwards reaction
How does the addition of a catalyst affect the position of equilibrium, what does the catalyst do?
Does not alter the position of equilibrium or the composition of the equilibrium system·
A catalyst speeds up the rate of the forwards and reverse reactions equally
A catalyst increases the rate at which equilibrium is established- increases the rate of the forwards and backwards reactions by equal amounts
Speeds up the rate of reaction by using an alternative route of reaction with a lower activation energy and speeds up the forwards and reverse reactions by the same amount
Exam Question: Why you shouldn't add other acidic cleaners with bleach?
Adding acid removes the NaOH so NaOH concentration decreases
The equilibrium position moves to the left producing more NaOH and Cl2
Chlorine is a toxic gas- unsafe
What are the problems associated with the use of high pressure, high temperatures and low temperatures in industry?
High pressure: expensive (pumps, pipes, plants), dangerous/ safety concerns
High temperature: expensive to maintain
Low temperature: rate of reactions is too low to produce sufficient yield for it to be economically worth it- particles do not have enough kinetic energy
What might be used in industry to increase the production of products but not affect the equilibrium position, why?
A catalyst- does not change the position of equilibrium. However, it does increase the rate that equilibrium is established by increasing the rate of both the forward and reverse reactions equally. Can ensure that a reaction can occur at lower temperature- cheaper.
Exam Tip: 4 things to mention when answering question about equilibrium
1. Direction of the Equilibrium shift
2. Observations
3. Reasons
4. Le Chatelier reference
What does the equilibrium constant tell you about the reaction?
Gives an idea of where the equilibrium position lies
Does the equilibrium constant change whether the starting reaction mixture is the products or the reactants?
No it stays the same
What condition changes the equilibrium constant?
The temperature- Remains constant unless the temperature is changes
What is the temperature for an equilibrium constant stated in?
Kelvin
Is the equilibrium constant the same for all equations or does it change?
Each equilibrium constant refers to a different reaction
What at the symbol for equilibrium constant?
Kc (small c)
What are the units of Kc?
Units must be determined for each equation by creating a fraction containing all of the units of the reactants and products and turning it into a single term
What must you ensure is done to the equation before calculating the equilibrium constant?
It is balanced
What is the general equation for equilibrium constant?
General Equation:
aA + bB → cC + dD
(at constant temperature)
Expression for Kc:
[C]c [D]d = Equilibrium constant (Kc)
[A]a [B]b
When stating the equilibrium constant what value must also be stated along side it?
Temperature
What happens when the top line in the Kc equation is </> the bottom line?
When top line in the Kc relationship> than the bottom line- more products than reactants- value more than 1?
When the bottom in the Kc relationship< than the bottom line- more reactants than products, the value is less than 1- can be very very small
Are reactions with Kc << or >> equilibrium reactions or not?
Yes they are- All reactions are equilibrium reactions, even if the reaction seems to go to completion- always actually have a little bit of reactant left at equilibrium with the product
What does a value of Kc >1 mean?
Equilibrium lies to the right- Slightly more products than reactants at equilibrium
What does a value of Kc >>1 mean?
(greater than 10^10)
Equilibrium lies far to the right- Many more products than reactants at equilibrium, reaction appears to have gone to completion
What does a value of Kc =1 mean?
Equilibrium lies in the middle- the amount of products and reactants at equilibrium will be the same
What does a value of Kc <1 mean?
Equilibrium lies to the left- Slightly more reactants than products at equilibrium
What does a value of Kc <<1 mean?
(less than 10^-10)
Equilibrium lies far to the left- Much more reactants than products present at equilibrium means the reaction appears not to have happened
What happens to the system, in terms of the equilibrium constant, when the concentrations change?
When a system is in equilibrium and more reagent (or product) is added the concentrations change and the equilibrium is disrupted
The system is trying to keep Kc the same- position of equilibrium changes so that Kc gets back to Kc value
Changing concentrations so not alter Kc once equilibrium has been reached assuming temperature remains constant
More product/ reactant must be formed to rebalance the mixture- restore/ maintain Kc
The position of the equilibrium shifts to the right/ left- more to the product/ reactants side to re-establish Kc
What happens, in relation to Kc, if the concentration of the reactants is increased?
Increase the concentration of the reactants: the position of equilibrium will move to the right to increase the concentration of products and keep Kc constant
What happens, in relation to Kc, if the concentration of the products is increased?
Increase the concentration of the products: the position of equilibrium will move to the left to increase the concentration of reactant and keep Kc constant
Name two important chemical processes in industry that use equilibrium systems?
· Preparation of ammonia from nitrogen and hydrogen in the Haber process
· Conversion of sulphur dioxide into sulphur trioxide in the Contact Process
What are the aims of equilibrium reactions in industrial reactions?
Highest possible yields, quickly and cheaply as possible
Explain the equilibrium reaction in cobalt chloride when there are two flasks and each flask contains the same solution of cobalt (II) chloride dissolved in water containing chloride ions and one is in low and on is in high temperatures
Cobalt (II) complexes with water forming pink-coloured [Co(H2O)6]2+ ions- exothermic favoured by low temperatures
Cobalt (II) complexes with chloride ions to form blue-coloured [Co(Cl)4]2- ions- endothermic favoured by high temperatures