AP CHEM AP TEST NOTES
If a substance is impure
it would contain less moles than expected (final value is less than theoretical value
What property is important when measuring the purity of a substance?
Mass percent
1/45
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
46 Terms
If a substance is impure
it would contain less moles than expected (final value is less than theoretical value
What property is important when measuring the purity of a substance?
Mass percent
In order to calculate molar mass you need
The mass of the substance and molar mass
Why does an ion of phosphorous, P3- have a larger radius than a neutral atom of phosphorus?
The electrons in P3- have a greater Coulombic repulsion than those in the neutral atom
***The ion has three more electrons than the neutral atom meaning the overall repulsion will be greater. The electrons will “push” one another away more effectively creating a bigger radius
As atomic radius decreases across a period what value increases and why?
The electronegativity value will increase b/c # of protons increases
The average atomic mass of an element on periodic table is equal to
the average mass of a mole of atoms of that element in grams as well as being the average mass of a single atom of that element in amus.
Which of the following can be inferred from examining mass spectroscopy data?
The existence of isotopes
Mass Spectrometry
used to determine the masses for individual atoms of an element which proves that each element has more than one possible mass which proves the existence of isotopes.
Atoms with more protons have a greater
effective nuclear charge as well as electronegativity
When electrons are at a higher energy level
they are more shielded from the nucleus and are easier to remove so they have a lower ionization energy
Is it easier to remove a proton from a higher energy sub shell or a lower energy subshell?
Higher energy sub shell b/c less effective nuclear charge
Elements with the same electronegativity are more likely to
share electrons and form a non polar covalent bond
What must a substance have to conduct electricity?
For a substance to conduct electricity it must contain charged particles that are mobile. In molten salts such as NaCl those charged particles are ions
Metals conduct electricity via
mobile electrons within a lattice of metal atoms
What is a property of ionic solids and why?
Brittleness
The Brittleness of ionic solids is due to the internal repulsion of ion layers within the crystal lattice that occurs when the layers are displaced by an outside force parallel to the layers
Structure of Metallic Solid
The structure of a metallic solid consists of a lattice of positive ions immersed in a sea of electrons in which electrons can move freely (therefore being able to conduct electricity)
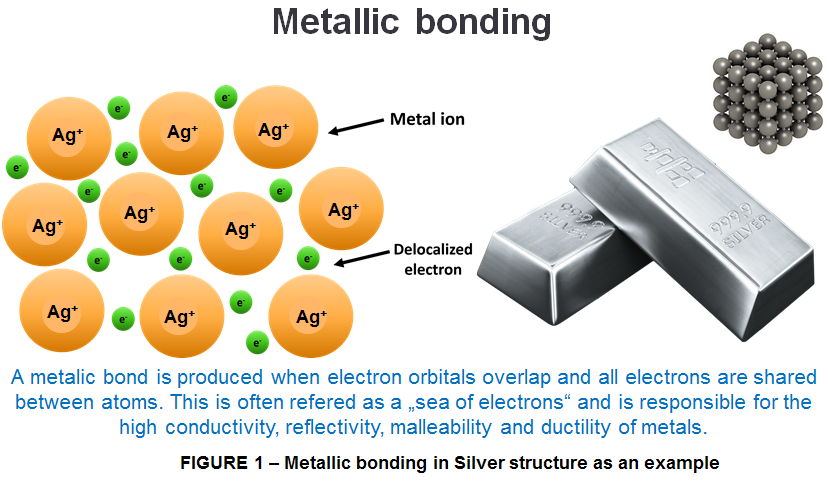
Why are metallic solids malleable?
Adjacent layers of positive ions can move relative to each one another while still remaining in full contact with the sea of electrons
When a molecule has two different charges on its sides like CH2Cl2 is it polar or nonpolar?
The separation of charge constitutes a dipole and the molecule is polar.
Lattice energy
The energy released when one mole of an ionic compound forms from its constituent ions in the gas phase
Charge of ions- Higher charges lead to stronger attractions and larger lattice energy
Ionic Radius- Inversely proportional to lattice energy so the larger the ionic radius the lower the lattice energy
Bond Order Formula
1/2(Bonding—Antibonding)
When is a molecule most stable?
When it has the lowest amount of potential energy
Sigma bonds
Single bonds (including those in pi bonds)
Charges on ions contribute to the
valence electron count
Ionic Compounds have
high melting points
Which property would solid compounds of sulfate be expected to have?
High Melting point
***Since Sulfate is an anion (adds electrons) it tends to form ionic compounds which have high melting points
Which substances do not conduct electricity no matter the phase?
Network Covalent and Molecular Covalent
When do ionic substances conduct electricity?
Ionic substances conduct electricity in aqueous and liquid form NOT solid or gas
When do metallic substances conduct electricity?
ONLY as solids or liquids
Nonpolar Covalent substances have a
Low melting point
Covalent Network Substances
Have a high melting point
When do Interstitial alloys form?
When atoms of greatly different sizes combine
Interstitial Alloys
Alloys formed when small atoms (like carbon or hydrogen) fit into the interstitial spaces (gaps) between the larger metal atoms in a metal lattice
Example: Steel, where carbon atoms fit between iron atoms
Analogy: Like pebbles fitting the gaps between basketballs in a box
Substitutional Alloys
Alloys formed when atoms of similar size to the host metal replace or substitute for some of the metal atoms in the lattice
Example: Brass where zinc atoms replace some copper atoms
Analogy: Like swapping out some basketballs for slightly smaller soccer balls in the same box
If an ionic compound dissociates into a greater number of ions then
it will be a better conductor of electricity
Example: SrBr2 is a better conductor of electricity than BaS because SrBr2 dissociates into one Sr2+ and two Br- for a total of 3 ions while BaS dissociates into only one Ba+ and one S- for a total of two ions
A sample of liquid NH3 is brought to its boiling point. What occurs during the boiling process?
The hydrogen bonds holding separate NH3 molecules together break apart
****When a covalent substance undergoes a phase change the IMF holding its molecules together get broken apart****
Columb’s Law
Predicts that the attractive force between the partial negative charge in the O atom and the cation will be directly proportional to the charge of the ion (greater charge) and inversely proportional to the size or ionic radius (smaller radius). This is similar to lattice energy
Why do covalent network solids have such a high melting point?
Atoms that are covalently bonded in an network solid are stronger than dispersion forces leading to a higher melting point
At a fixed temperature what best describes the speeds of the particles in a gas?
The particles have different speeds
***At a given temperature gas particles follow the Maxwell Boltznann distribution meaning that not all particles move at the same speed. Instead their speeds vary with some moving faster and some moving slower. This distribution results from random collisions and energy exchanges between particles
If gases are at the same temperature what determines which one has the particles with greater speed?
The gas with the lower molar mass has the greater speed.
Molecules with larger more polarizable electron clouds have
stronger IMF and deviate more from ideal gas behavior
Gas pressure is due to
collisions of gas particles with walls of the container. If gas particles about to hit the walls of a container are attracted to nearby gas (strong IMF) particles they will hit the walls of the container with less force than expected.
Large molecules deviate from the kinetic molecular theory because
According to the Kinetic Molecular theory, ideal gas molecules have negligible volumes so the larger its molecules are, the more the behavior of the gas will deviate from that of an ideal gas.
Distillation
A separation technique that takes advantage of differences in boiling points
Visible Light
Absorption of photons in the visible range will cause transition of electrons between electron energy levels
Infrared Radiation
The absorption of photons of infrared radiation is associated with transitions in Molecular vibrational levels
Under which conditions would gases from a container deviate the most from ideal condtions and why?
Low temperatures b/c the intermolecular forces between gas molecules would increase since molecules are moving more slowly