PRECIPITATION REACTIONS
1/78
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
79 Terms
Antigen + Antibody
Important role in diagnosing many different diseases
Immunoassays
- Developed to detect either antigen or antibody
- Maybe Manual or Automated
Precipitation
Agglutination
Immunoassays are based on 2 Principles:
Precipitation
- Combining soluble antigen with soluble antibody to produce insoluble complexes that are visible
- First noted by Kraus (1897), culture filtrates of enteric bacteria would precipitate when mixed with antibody
Agglutination
- Process by which particulate antigens (cells) aggregate to form larger complexes when specific antibody is present
Antigen-Antibody Binding
- The primary union of binding sites on antibody with specific epitopes on an antigen depends on affinity and avidity.
Avidity
- Sum of all the attractive forces between antigen and antibody
- Involves the strength with which a multivalent antibody binds a multivalent antigen
- Measure of overall stability of an antigen-antibody complex
- High avidity can compensate for low affinity
Affinity
- Initial force of attraction between a single Fab site on an antibody molecule and a single epitope
Epitope - Paratope
- held together by non-covalent bonds
Ionic bonds
- Occur between oppositely charged particles
Hydrogen bonds
- Attraction between polar molecules that have a slight charge separation
- Positive charge resides on hydrogen atom
Hydrophobic bonds
- Occur between nonpolar molecules that associate with another and exclude molecules of water
Van der Waals forces
- Interaction between electron clouds of oscillating dipoles
1x10-7 mm
These are weak bonds that occur over a ____________ distance, so there must be a very close fit between antigen and antibody.
specificity of antibody
Strength of attraction depends on the _____________ for a particular antigen
stronger their bond (attraction).
The more specific the Ag-Ab to each other, the _______________________
Cross-reactivity
- Antibodies are capable of reacting with antigens that are structurally similar to the original antigen that induced antibody production
- High resemblance to original antigen, stronger the bond
- Perfect lock-and-key relationship = maximal affinity
Law of Mass Action
- Govern all reversible antigen–antibody binding
- States that free reactants are in equilibrium with bound reactants
Equilibrium constant
- Represents difference in the rates of the forward and reverse reaction
- can be seen as a measure of the goodness of fit.
Formula of Equilibrium constant
o [AgAb] = conc. Antibody-antigen complex (mol/L)
o [Ab] = conc. of Antibody (mol/L)
o [Ag] = conc. of Antigen (mol/L)
![<p>o [AgAb] = conc. Antibody-antigen complex (mol/L) <br>o [Ab] = conc. of Antibody (mol/L) <br>o [Ag] = conc. of Antigen (mol/L)</p>](https://knowt-user-attachments.s3.amazonaws.com/af9f5fbd-bd37-427d-94e7-5fb967fc25d1.png)
↓ tendency of complexes to dissociate
The ↑ Strength of Bind/Avidity, The ____________________
↑ K1
The ↓ K2, The __________________
↑ amount of antigen-antibody complex
The ↑ K, The ___________________
- Reaction more visible ore detectable
Precipitation Curve
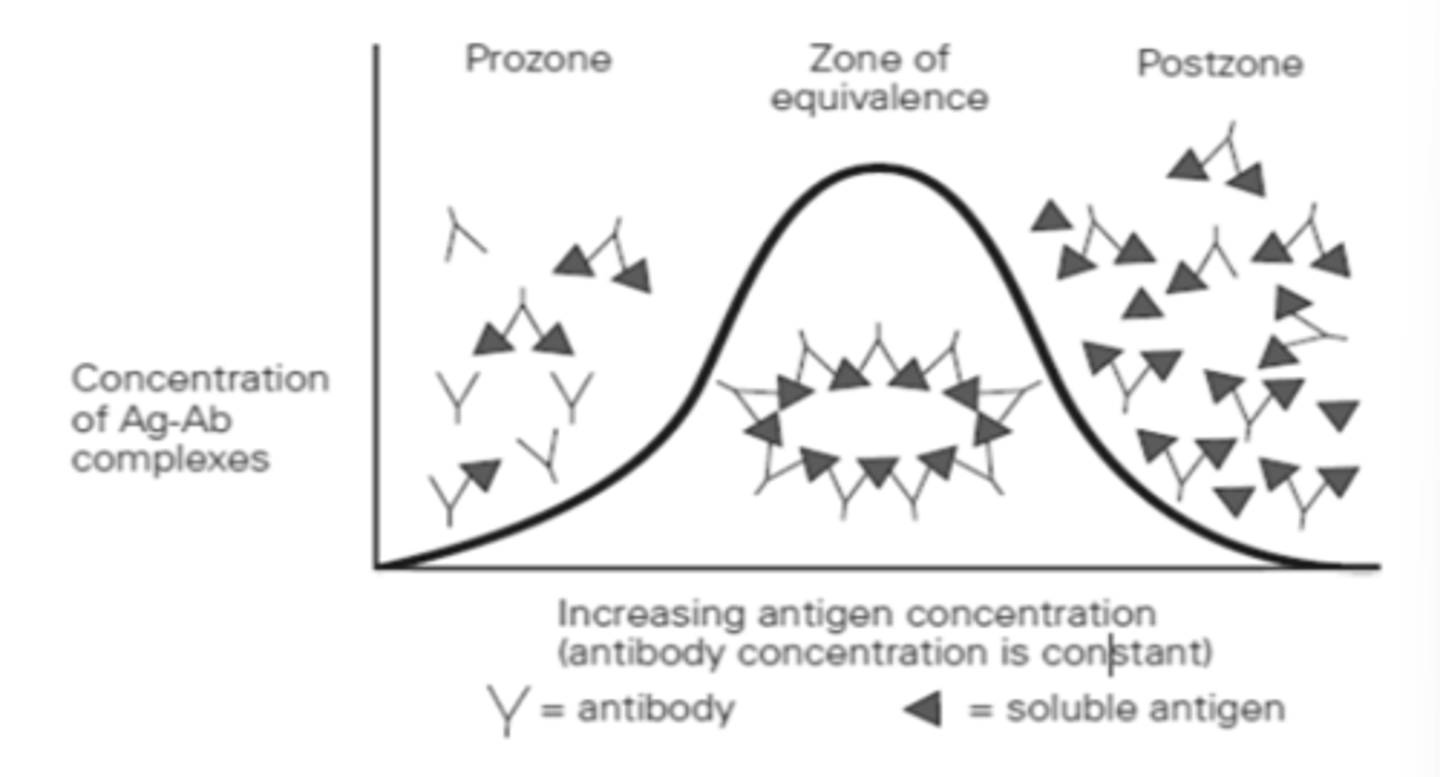
Zone of Equivalence (ZOE)
- Ab = Ag
- Where optimum precipitation occurs
- Number of multivalent sites of antigen and antibody are approximately equal
Precipitation at ZOE
Antibody binds to more than one antigen and vice versa forming a lattice (Reversible reaction)
Lattice Hypothesis
- Formulated by Marrack
1. Each antibody molecule must have at least two binding sites
2. Antigen must be multivalent
- Antigen + Antibody = Multimolecular lattice that increases in size until it precipitates
- Heidelberger and Kendall
o Performed quantitative precipitation reactions that established proof for this theory.
↑ amount of precipitation up to the ZOE
The ↑ soluble antigen, The ___________________
↓ precipitation
The ↑ amount of antigen overwhelms the number of antibody combining sites present, The ___________________________
Prozone Phenomenon (PrP)
- Ab > Ag (Antibody Excess)
- Antigen combines with only one or two antibody molecules
- No cross-linkages or visible end result
- Many free antibody molecules remain in solution
- False Negative reactions
o due to high antibody concentration
o Dilute antibody and perform the test again
Postzone Phenomenon (PsP)
- Ab < Ag (Antigen Excess)
- Small aggregates are surrounded by excess antigen
- No lattice is formed
- Excess antigen may obscure presence of small amount of antibody
o Paired sera
o Test is repeated with an additional patient specimen
Zone of Equivalence
For the Precipitation to be detectable, they must be run where
1. Turbidimetry
2. Nephelometry
a.) End-point Nephelometry
b.) Kinetic/Rate Nephelometry
MEASUREMENT OF PRECIPITATION BY LIGHT SCATTERING
Precipitation in a Fluid Medium
Turbidimetry
- Measure of the turbidity or cloudiness of a solution
- Measures the reduction in light intensity due to:
Reflection, Absorption, Scatter
*Scattering of light occurs in proportion to the size, shape, and concentration of molecules in a solution.
Absorbance units
measure of the ratio of incident light to that of transmitted light
Spectrophotometer
Automated Clinical Chemistry Analyzer
Measurements in Turbidimetry are made using:
Nephelometry
- Measures the light that is scattered at a particular angle from the incident beam as it passes through a suspension.
- can be used to detect either antigen or antibody.
- Amount of light scattered = index of the solution’s concentration
- Measure light scatter at 10° to 90°
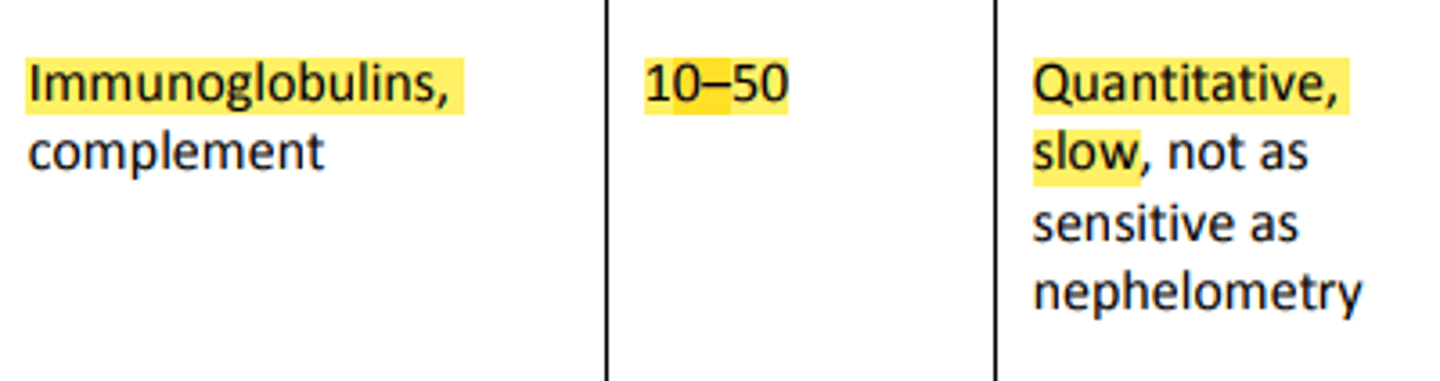
-Nephelometry is more sensitive than Turbidity with a lower limit of detection of 1-10 mg/dL

End point nephelometry
- Reaction is allowed to run essentially to completion, but large particles tend to fall out of solution and decrease the amount of scatter.
Kinetic/ Rate nephelometry
- Rate of scattering increase is measured immediately after the reagent is added.
- Rate change is directly related to antigen concentration of antibody concentration is constant.
- provides accurate and precise quantitation of serum proteins, and the cost per test is typically lower than other methods.
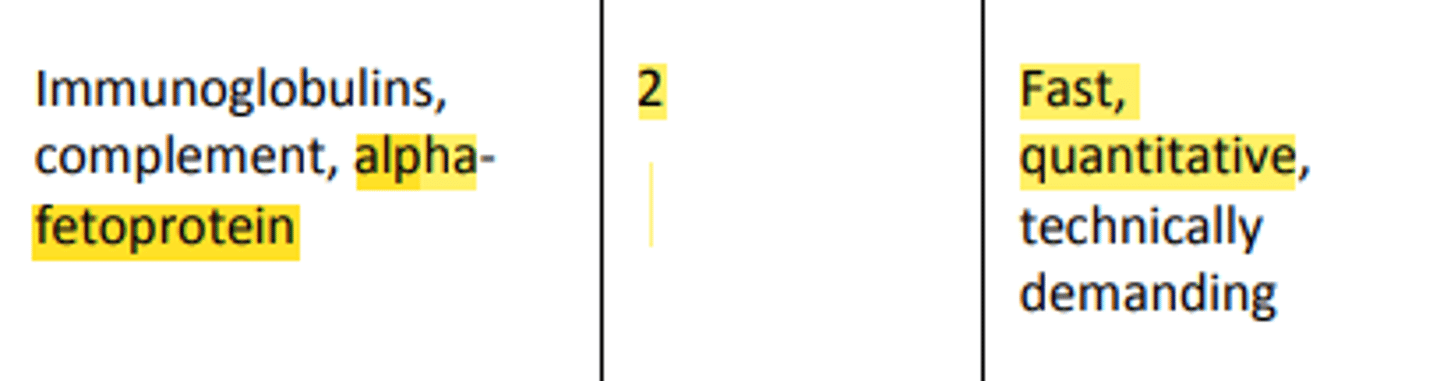
o Quantification of immunoglobulins (IgG, IgA, IgM, and IgE)
o Kappa and Lambda chains
o Complement proteins
o C Reactive Protein o Clotting Factors
Kinetic/Rate Nephelometry is used in:
Agar
- High-molecular-weight complex polysaccharide derived from seaweed
- Has negative charge
Agarose
- Purified agar
- Used as support medium (used to read/determine precipitation results)
- More preferred than Agar because has almost no charge
Agar and agarose
- Stabilize diffusion process
- Allow visualization of precipitin bands
Passive Immunodiffusion
- When no electrical current is used to speed up the antigen-antibody combination by means of diffusion.
- Combination of Ag and Ab occurs by allowing them to diffuse towards each other (in a medium).
1. Size of particles
2. Temperature
3. Gel viscosity
4. Amount of hydration
Factors that affect Rate of Diffusion
0.3 to 1.5%
Percentage of agar concentration allows diffusion of most reactants
Single Diffusion technique
Radial Immunodiffusion
- End-point method
- Kinetic method
One-Dimensional techniques
Single Diffusion technique
- Pioneered by James Oudin
- Oudin Single Diffusion (Single-linear Diffusion)
- Antibody was incorporated into agarose in a test tube
- Only one reactant migrates (either Ag or Ab)
- Medium: Agarose gel in a test tube
- Positive result: Precipitin band
* If after 24hrs incubation and NO PRECIPITIN LINE appeared, DO NOT IMMEDIATELY REPORT as Negative. Incubate again for another 24hrs at 37C.
Radial Immunodiffusion
- Modification of single-diffusion technique
- used to measure IgG, IgM, IgA, and complement components.
- Antibody is uniformly distributed in the support gel, and antigen is applied to a well cut into the gel
- Antigen-antibody combination changes proportions until the zone of equivalence is reached and stable lattice is formed
- Area of the ring = measure of Antigen concentration
- Disadvantage: fairly expensive to run
End-point method
Kinetic method
Two techniques of Radial Immunodiffusion
End-point method
- Developed by Mancini
- Antigen is allowed to diffuse to completion
- When equivalence is reached, no further change in the ring diameter (occurs between 24 and 72 hours)
- Square of diameter is directly proportional to antigen concentration
- Disadvantage: takes time to obtain results
Kinetic method
- aka Fahey and MacKelvey Method
- uses measurements taken before point of equivalence is reached
- Diameter is proportional to the log of concentration
- Readings are taken at about 18 hours
Monospecific antiserum
- Must be used to increase clarity of precipitation (High affinity)
1. Overfilling/Under filling the wells
2. Nicking the side of the wells when filing
3. Spilling sample outside the wells
4. Improper incubation time and temperature
5. Incorrect measurement
Sources of Error for One Dimensional Techniques:
1. Overfilling/Under filling the wells 2. Nicking the side of the wells when filing 3. Spilling sample outside the wells 4. Improper incubation time and temperature 5. Incorrect measurement
Ouchterlony Double Diffusion
Double-linear Ouchterlony
Double-radial Ouchterlony
Two - Dimensional Techniques
Ouchterlony Double Diffusion
- Old, classic immunochemical techniques
- Both Antigen and Antibody diffuse independently through a semi-solid medium in two dimensions
- Density of the lines reflects the amount of immune complex formed
- Most Ouchterlony plates are set up with a central well surrounded by 4 - 6 equidistant outer wells.

Aspergillus
Blastomyces
Coccidioides
Candida
Ouchterlony Double Diffusion is used to identify:
Arc
Cross-Line
Fusion of two lines with spur
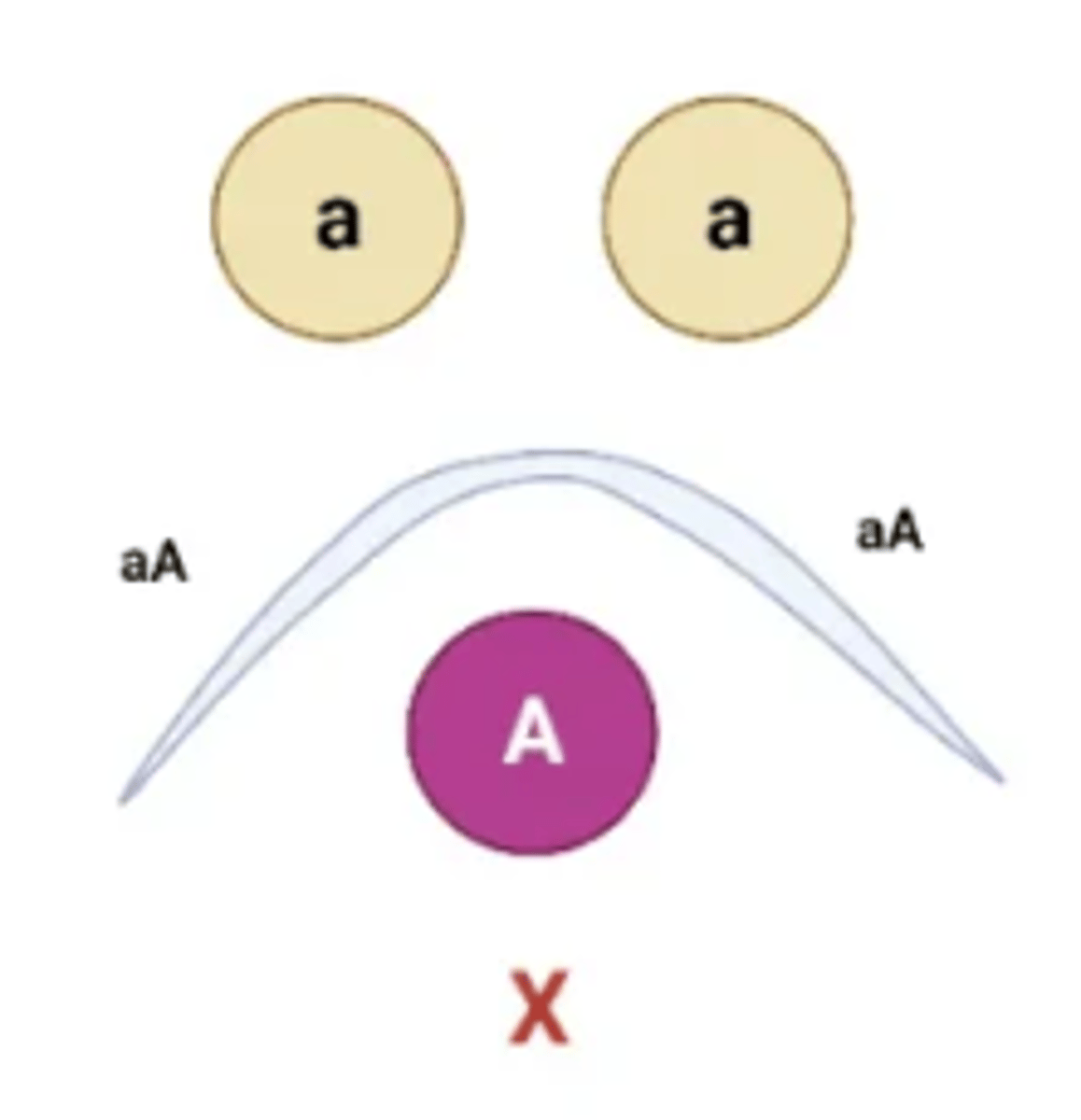
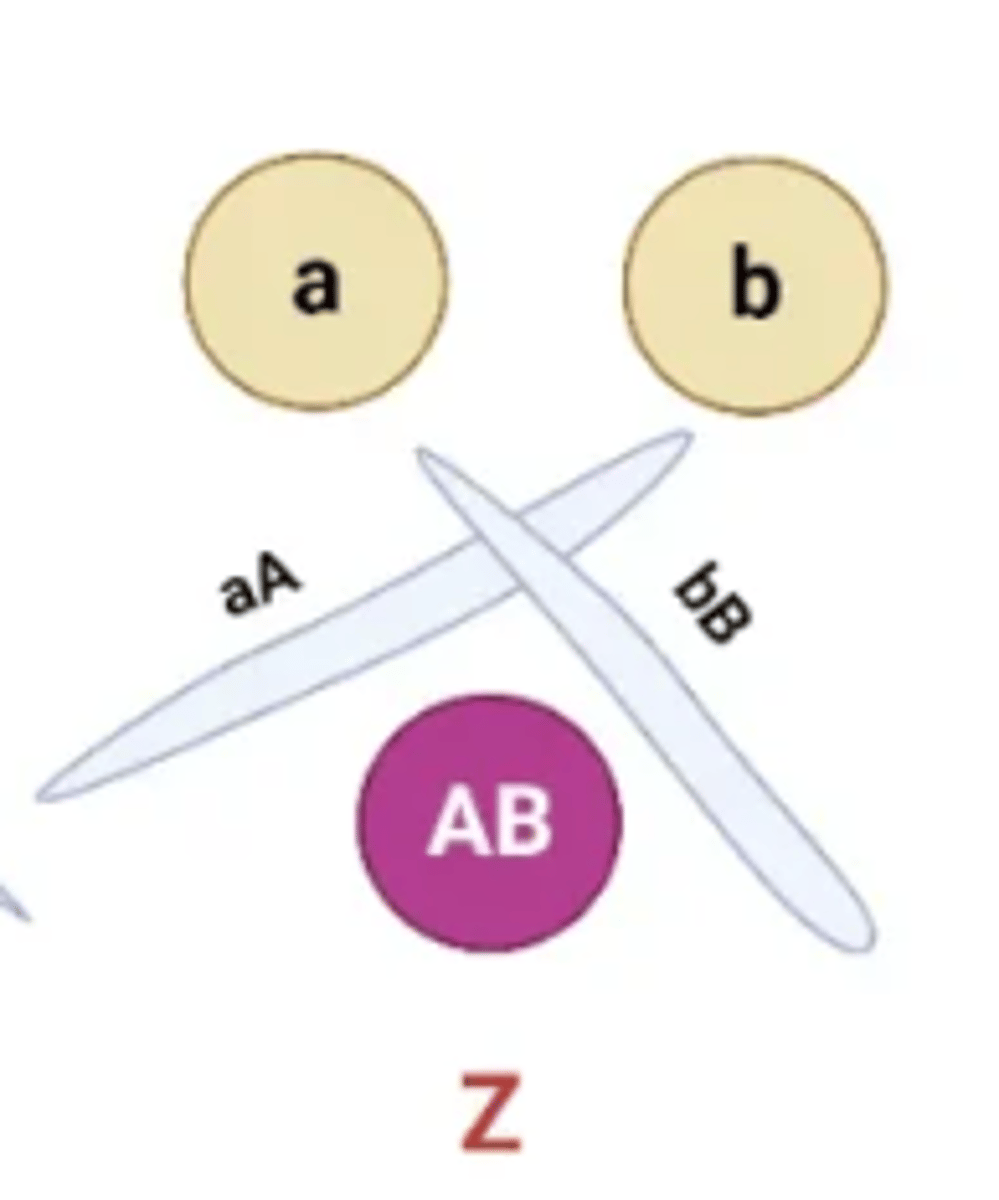
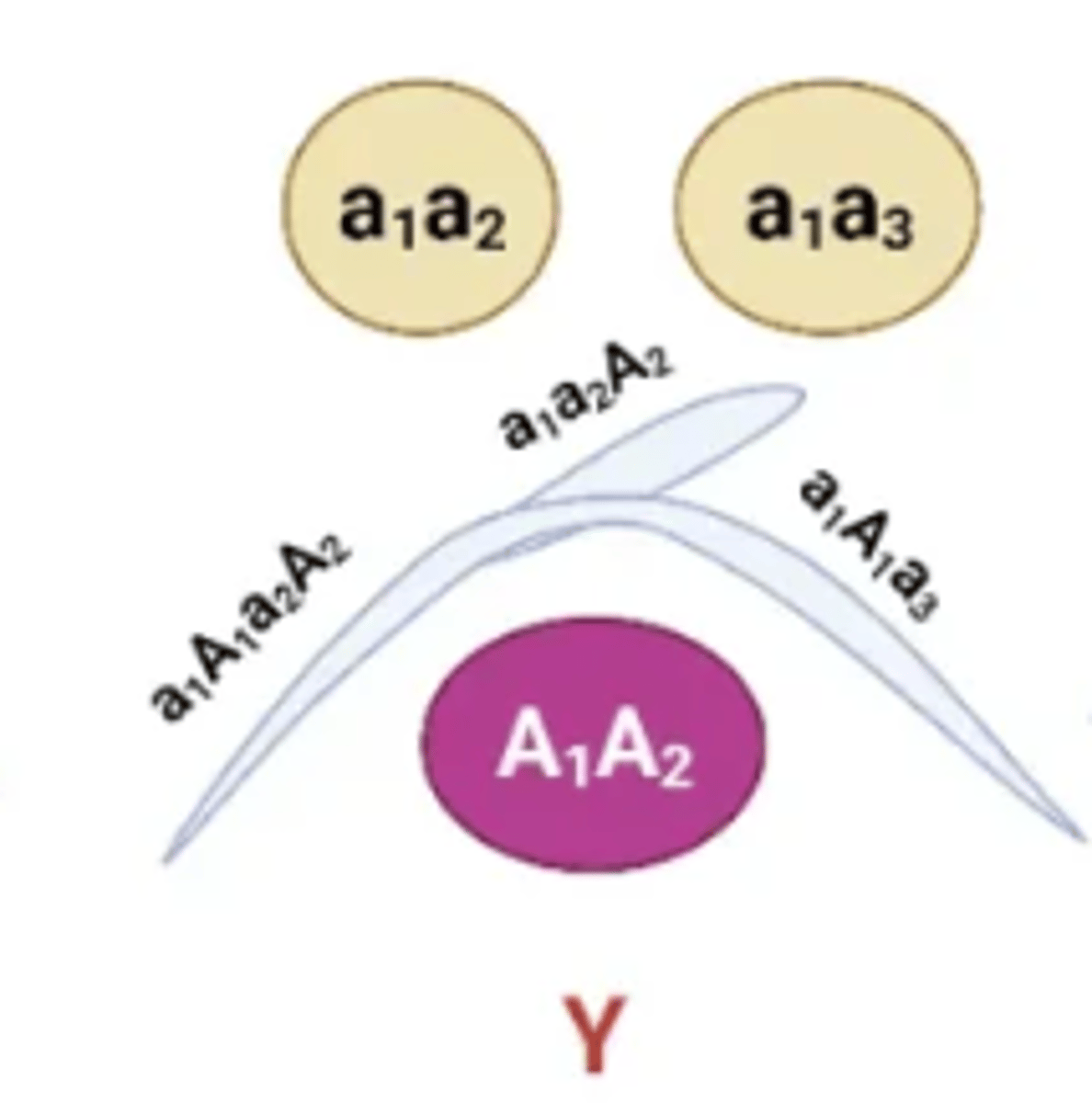
The position of the precipitin bands between wells allows for the antigens to be compared with one another. Several patterns are possible:
Arc
Represents serological identity
Cross-Line
Two separate reactions, Compared antigens share no common epitopes
Fusion of two lines with a spur
Indicates partial identity, Spur always points to the simpler antigen
1. Overfilling the wells
2. Irregular hole punching
3. Nonlevel incubation
4. Drying out of gel
5. Inadequate time for diffusion
Factors affecting accuracy of results for Ouchterlony Double Diffusion
Rocket Immunoelectrophoresis
Countercurrent Immunoelectrophoresis
Immunoelectrophoresis
Immunofixation Electrophoresis
Western Blot
ELECTROPHORETIC TECHNIQUES
Precipitation by Electrophoretic Techniques
Electrophoresis
- Separates molecules according to differences in their electric charge when they are place in an electric field
- Can be applied to both single and double diffusion
Rocket Immunoelectrophoresis
- Just like RID but uses electrophoresis to facilitate migration of the antigen into the agar
- End result: Conical precipitin line resembling a rocket
- Height of rocket (well to apex) directly proportional to the amount of antigen - Much more rapid than RID
- Used to quantitate immunoglobulins using pH 8.6 buffer.
One-dimension electroimmunodiffusion
o Adaptation of radial immunodiffusion (RID)
o Developed by Laurell (early 1960)
Immunoelectrophoresis
- Double diffusion technique that occurs at right angles
- Incorporates electrophoresis current to enhance results
- Introduced by Grabar and Williams (1953)
- Two-step process
- Source of Antigen: SERUM
- Antiserum is placed in the trough, and the gel is incubated for 18- 24 hours.
- Limit of Detection: 3-20 uL

Immunofixation Electrophoresis
- First described by Alper and Johnson
- Similar to Immunoelectrophoresis EXCEPT that after electrophoresis takes place, antiserum is applied directly to the gel’s surface.
- Uses Agarose or Cellulose Acetate
- Requires shorter time and results higher resolution
- Gel thickness: Approx. 1mm
- Hypogammaglobulinemia = faintly staining bands
- Polyclonal hypergammaglobulinemia = darkly staining bands in gamma region
- Waldenstrom’s macroglobulinemia = Monoclonal bands with dark and narrow bands in specific lanes

Hypogammaglobulinemia
faintly staining bands
Polyclonal hypergammaglobulinemia
darkly staining bands in gamma region
Waldenstrom's macroglobulinemia
Monoclonal bands with dark and narrow bands in specific lanes
Western Blot
- Adaptation of immunofixation electrophoresis
- Confirmatory tests to detect HIV-1
- Uses Nitrocellulose paper
Sources of Error in Electrophoresis
1. Applying current in wrong direction – samples may run off the gel or not separated
2. Incorrect pH of buffer
3. Incorrect electrophoresis time
4. Concentrations of antigen and antibody
5. Amount of current
a. Weak – incomplete separation
b. Too strong – generation of heat leading to denaturation of proteins
Nephelometry

Radial immunodiffusion
Ouchterlony double diffusion

Rocket electrophoresis
Immunoelectrophoresis

Immunofixation electrophoresis
