Chem Naming
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
30 Terms
OH-
Hydroxide
NH4^+
Ammonium
NO3^-
Nitrate
NO2^-
Nitrite
ClO4^-
Perchlorate
ClO3^-
Chlorate
ClO2^-
Chlorite
ClO-
Hypochlorite
CO3^ 2-
Carbonate
HCO3^-
Hydrogen Carbonate(bicarbonate)
CN^-
Cyanide
C2H3O2^-
Acetate
SO4^ 2-
Sulfate
HSO4^-
Hydrogen Sulfate(bisulfate)
SO3^ 2-
Sulfite
HSO3^-
Hydrogen sulfite(bisulfite)
PO4^ 3-
Phosphate
HPO4^ 2-
Hydrogen Phosphate
H2PO4^-
Dihydrogen phosphate
PO3^ 3-
Phosphite
Ionic
metal and nonmetal
Molecular/covalent
nonmetal and nonmetal
polar molecules
e- unequally shared and polar bonds don’t cancel
nonpolar molecules
all nonpolar bonds or polar bonds all cancel symmetrically
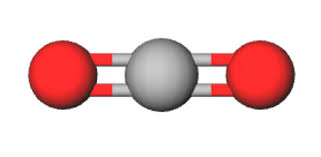
linear
Bonded-2 , Lone pairs - 0, bond angle- 180 degrees, molecular shape- linear, example CO2
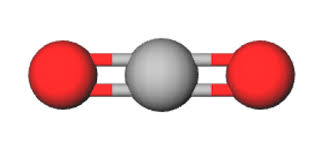
Triagonal Planar
Bonded- 3, Lone pairs-0, bond angle- 120, molecular shape- trigonal planar, example H2CO
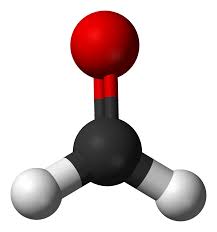
Trigonal Planar
Bonded-2, Lone pairs- 1, bond angle-120, molecular shape-bent, example SO2
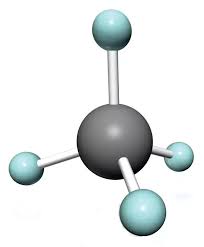
Tetrahedral
Bonded-4, lone pairs- 0, bond angle-109, molecular shape- tetrahedral, example CH4
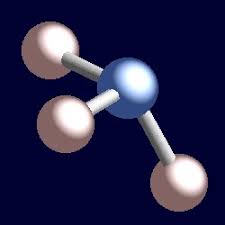
Tetrahedral
Bonded- 3, lone pairs-1, bond angle-109, molecular shape- trigonal pyramidal, example NH3
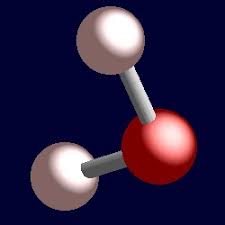
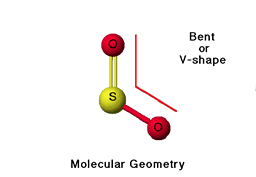
Tetrahedral
bonded- 2, lone pairs-2, bond angle-109, molecular shape- bent, example H2O