Illustrate Le Chatelier’s Principle by demonstrating the effects of concentration changes
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
13 Terms
Draw a diagram of this experiment
…
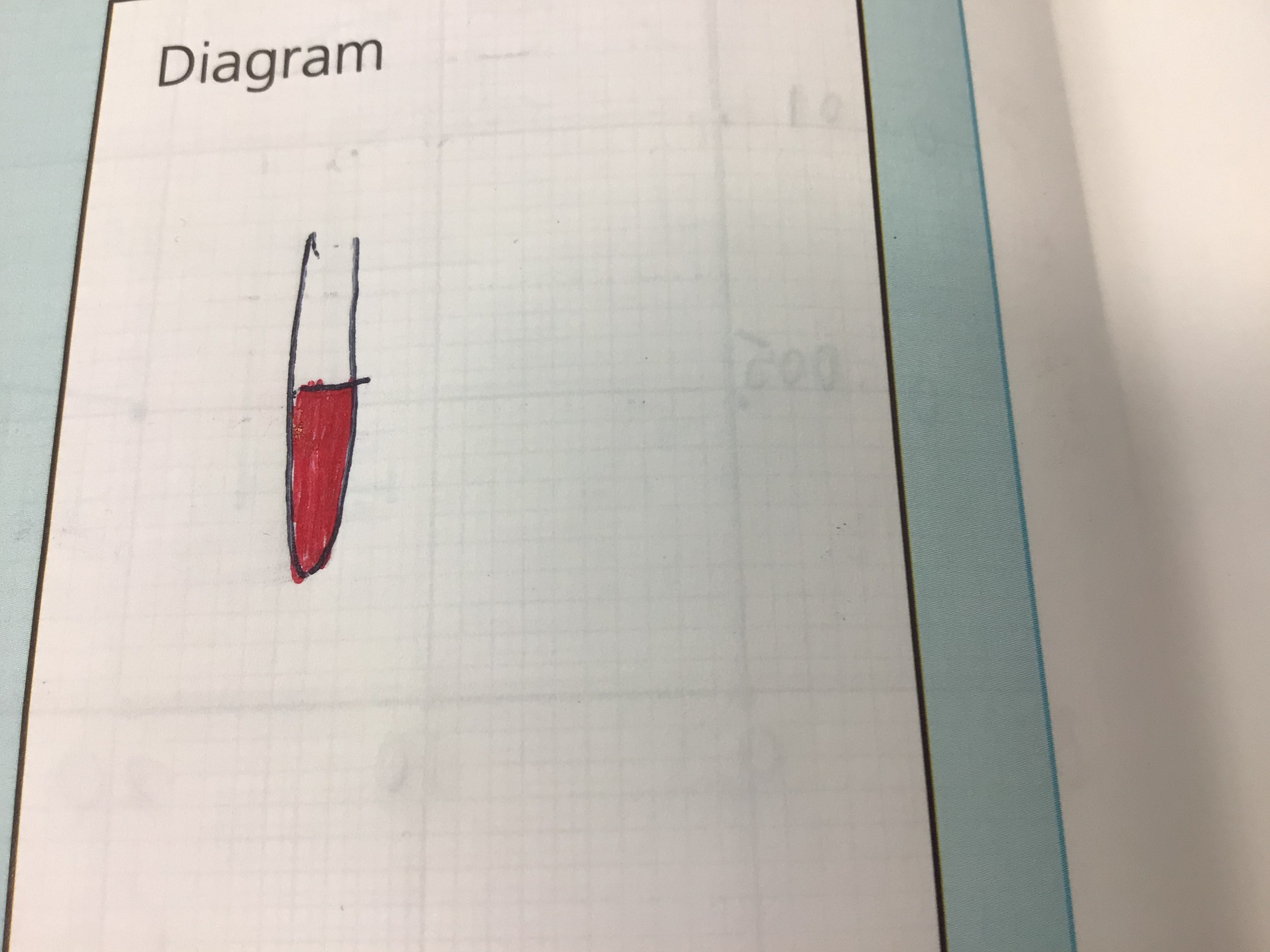
Step 1
Add a drop of potassium thiocyanate solution to iron (III) chloride
What is observed?
Red colour is observed
Explain observation
Equilibrium shifts to RHS
Equation of experiment
Fe3+ + CNS- ⇌ Fe(CNS)2+
Step 4
Add dilute hydrochloric acid until a colour change is observed
What is observed when hydrochloric acid is added?
Red colour disappears
Draw a diagram showing the experiment when hydrochloric acid is added
…

Explain the observation when hydrochloric acid is added
Equilibrium favours left side as LHS reacts with Cl- ions to produce the reactants
Step 7
Add in some potassium thiocyanate solution
What do you observe when potassium thiocyanate ions are added?
Solution turns red
Explain the observation when potassium thiocyanate is added
Equilibrium shifts to RHS absorbing CNS-
Draw a diagram of the solution when potassium thiocyanate is added
…
