Week 3: Earth's Atmosphere and Greenhouse Effect
1/35
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
36 Terms
How is matter bound together?
Electric fields
Atoms bound by sharing electrons
Atoms aggregated into gases, liquids/ solids bound by electric forces
Why is it important for the climate that ice is less dense than water?
if water froze and it sunk instead of sitting on top of the water
lower albedo of earth as albedo of ice is very high
How can the state of matter be changed?
Only if EM of the right frequency interacts with it
The Molecular Absorption Spectra
Molecules absorb at more frequencies than atoms
Various processes broaden the absorption spectra of molecules
collisions/ pressure broadening
doppler shift
What makes a molecule a good absorber of EM radiation?
Molecules with an electric dipole
Symmetric electrical fields e.g N2 and O2 don’t absorb IR
CO2 is symmetric but can produce an electric dipole if distorted → IR absorption for vibrations
H2O has a dipole in resting state → IR absorption for vibrations and rotations
What does Planck’s Law give?
the intensity of black-body radiation as a function of wavelength and temperature, i.e. the shapes of the black-body radiation curves
Planck’s law is an idealisation → real bodies don’t absorb or emit all wavelengths of light
How can you calculate thermal emission from real bodies?
Product of Planck’s Law and the Body’s Emissivity
Bλ (λ,T) × ε(λ
both functions of wavelength
Planck’s law sets the maximum thermal emission for the body.
The emissivity sets the fraction of this maximum that is realized at each frequency / wavelength, i.e. the efficiency with which it emits
What is Kirchoff’s Law?
Absorptivity (aλ ) = emissivity (ελ )
Things are as good at absorbing EM radiation of a specific wavelength as they are at emitting it → absorption lines = emission lines
How are emissivity and absorptivity measured?
As a fraction
Range between 0 and 1
How are concentrations of gases in the atmosphere expressed?
parts per million by volume (ppmv)
Which gases in the atmosphere have a significant radiative effect?
CO2 (~410 ppmv), Methane (1.9 ppmv), Water Vapour
^ have 3 or more parts and so more complex vibrations
What is the dominant greenhouse gas in the Earth’s atmosphere?
Water Vapour
Absorbs strongly in many bands
Weakly absorbing in atmospheric window
Where is the atmospheric window?
Between ~8 and ~14 μm
Around the peak of Earth’s black-body spectrum
Why does CO2 have a greater greenhouse effect than H2O?
CO2 absorbs over a narrower range of frequencies than water but it has a strong absorption band in the atmospheric window
Gases which absorb wavelengths in the atmospheric window can have a greenhouse effect
Greenhouse gases (GHGs) are most effective if they absorb frequencies that other GHGs don’t.
If they were all absorbing the same frequencing→ less of the spectrum in total absorbed
Absorption of light by the atmosphere
Some frequencies of incoming light are absorbed by the atmosphere
Water vapour absorbs some bands of the near-infrared
Ozone absorbs UV strongly
Where does the blackbody spectrum show emission from?
Reflects the temperature and altitude of the last layer which emitted it
The Earth’s longwave spectrum follows the black-body spectrum for the surface where it is transparent and the cooler upper atmosphere where it is strongly absorbing
What is radiative forcing?
measure of the influence a factor has in altering the balance of incoming and outgoing energy in the Earth system.
Units of radiative forcing
Wm-2
What effect does a positive radiative forcing have?
A positive radiative forcing will cause the Earth to absorb more energy and warm.
As it warms it will emit more longwave radiation (Stefan-Boltzmann law) and eventually a new equilibrium temperature will be reached when incoming and outgoing energy balances again
As you increase the amount of CO2 in the atmosphere, what effect does it have on outgoing longwave radiation
0 → 10 ppmv takes a big bite out of the spectrum and has a large radiative forcing
400 → 410 ppmv has a tiny radiative forcing.
What does band saturation do?
At current CO2 conc. → increasing conc. leads to diminishing increases in radiative forcing due to band saturation
Frequencies most effectively absorbed have all been absorbed
Increasing CO2 increases absorption at tails of distribution
CO2 radiative forcing does not increase linearly with CO2 concentration
CO2 absorbs all long-wave emitted from the surface around its peak absorption frequency, and exponentially weakly away from this peak.
Radiative Forcing of CO2
proportional of the number of doublings of the CO2 concentration, i.e., it follows a logarithmic function:
RFCO2 = 3.71 x ln(C/ C0) / ln (2) Wm-2
This is because of band saturation
Ideal gas assumptions
Molecules are point particles (imagined as tiny balls)
Have elastic interactions (don’t lose energy after collision)
When do assumptions about ideal gases break down?
At very high pressure and low temperature
Assumption works well for atmospheric conditions
Equation showing the relationship between kinetic energy of an ideal gas and its temperature
EK = 3/2 nRT
n = number of moles
R= ideal gas constant
T= temperature in Kelvin
Force =
Mass x Acceleration
Unit of force
1 Newton (N) = 1 kg ms-2
Pressure =
force/ area
Units of Pressure
1 Pascal (Pa)= 1 Nm-2
What is pressure against a box determined by?
Number of collisions over area
Kinetic energy of colliding particles
Ideal Gas Law
PV = nRT
What does pressure in the ocean depend on?
Function of depth
Water:
Is effectively incompressible.
Pressure is proportional to depth as the weight of water above you increases monotonically
Every 10m deeper, pressure rises by 1 atmosphere (atm)
What does pressure in the atmosphere depend on?
Pressure as a function of altitude
Air:
Is compressible
Air at the bottom is compressed by the weight of air above.
Pressure falls exponentially with altitude
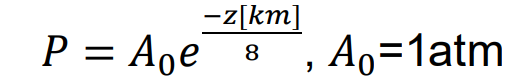
What is the scale height of the earth?
8 km
What is the air pressure at sea level?
1 atm
What is the air pressure at 8 km?
e-1
e-folding height
every 8 km it falls by the same fraction
Most of the mass of the atmosphere is below 8 km