Chap 1 and 2 - Mole and Redox
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
Define relative atomic mass and relative isotopic mass
Relative atomic mass (Def.) : Defined as the ratio of the average mass of one atom of the element to 1/12 of the mass of an atom of 12C isotope
Average includes relative abundance of ALL possible isotopes of that element
Relative isotopic mass (Def.) : Of a particular isotope is defined as the ratio of the mass of one atom of the isotope to 1/12 of the mass of an atom of 12C isotope
Deals specifically with atomic mass of 1 of the possible isotopes of an element
Define relative molecular mass and relative formula mass
Relative molecular mass (Def.) : Of a molecule is defined as the ratio of the average mass of one molecule of the substance to 1/12 of the mass of an atom of 12C isotope
Relative formula mass (Def.) : Of an ionic compound is defined as the ratio of the average mass of one formula unit of the compound to 1/12 of the mass of an atom of 12C isotope
State the unit for avogadros constant, molar mass, relative molecular mass
Avogadro's constant : /mol , molar mass : g , relative molecular mass : no unit
Define Avogadros constant, molar mass and molar gas volume
Avogradro’s constant : (Def.) : number of particles in 1 mole of a substance (6.02 x 10^23)
Molar mass : (Def.) : mass of one mole of a substance and has a unit of g/mol
Molar gas volume : (Def.) : volume that one mole of a gas occupies at a particular set of temperature and pressure
What is ppm and ppb
Concentrations also given as parts per million (ppm) or parts per billion (ppb) : used as a measure of small level (concentrations) of pollutants in air, water, soil
When a solution is diluted (by adding more solvent), the concentration of the solution decreases but the number of moles of the solute in the diluted solution remains unchanged
State the ideal gas equation
pV = nRT (Ideal Gas Equation)
This equation is the Ideal Gas Law:
p = pressure (in Pascals, Pa) (Eg. p=1atm=101325 Pa (1 atm = 101325 Pa)
V = volume (in m³)
n = number of moles of gas
R = universal gas constant (8.31 J mol⁻¹ K⁻¹)
T = temperature (in Kelvin, K)
Define empirical and molecular formula
Empirical formula (Definition) : of a compound is the simplest formula which shows the ratio of the atoms of the different elements in the compound
Molecular formula (Definition) : of a compound is the formula which shows the actual number of atoms of each element in one molecule of the compound
State the standard combustion equation of a hydrocarbon
CxHy + (x + y/4) O2 -> xCO2 + y/2 H2O
Complete combustion of a hydrocarbon (a compound that contains hydrogen and carbon only) gives 0.352 g of carbon dioxide and 0.072 g of water. Calculate its empirical formula and its molecular formula, given that it has a molar mass of 104 g mol−1
Answer C8H8

A mixture of CuSO4•5H2O and MgSO4•7H2O is heated until a mixture of the anhydrous salt is obtained. If 5.00 g of the mixture gives 3.00 g of the anhydrous salts, what is the percentage by mass of CuSO4•5H2O in the mixture?
73.8%

Define aliquot, standard solution, equivalence point
Aliquot : measured with a pipette and placed in a conical flask
Standard solutions : solution whose concentration is accurately known
Equivalence point : occurs when the reactants in the two solutions react according to the stoichiometry of the reaction
Define titre volume, end point, working range
Titre volume : the volume of solution added from the burette
End point : where indicator changes colour
Working range : pH where indicator changes colour
Describe dilution of a solution
Involves the addition of more solvent to a given solution
The number of moles of solute present does not change -> the solute is dispersed in a larger final volume -> lower concentration
Describe sampling a solution
Collection of a portion from a given solution
The concentration of the sample is the same as the original solution
The number of moles of solute present changes
State when back titration is used
Used when : direct titration not possible
Eg. One of the reactants is an insoluble solid (carbonates), when direct titration will be too slow, or when one of the reagents is volatile (Eg. ammonia)
Explain how back titration is used to find concentration of a solution
A known excess of one reagent A is allowed to react with an unknown amount of B
The amount of unreacted A is then determined by titration with a reagent C of known concentration
From the titration results, the amount of unreacted A and the amount of B can be found by simple stoichiometric calculations
Define redox reaction, reduction, oxidation
Redox : A reaction that involves reduction and oxidation simultaneously
Reduction : A process whereby a substance gains electrons, resulting in a decrease in oxidation number
Oxidation : A process whereby a substance loses electrons, resulting in an increase in oxidation number
Define reducing agent, oxidising agent, disproportionation, oxidation state
Reducing agent : A substance that undergoes oxidation and is an electron doner
Oxidising agent : A substance that undergoes reduction and it is an electron acceptor
Disproportionation : A redox reaction in which the same substance is both oxidised and reduced
Oxidation state : The total number of electrons that an atom either gains or loses in order to form a chemical bond with another atom
Explain how to find the oxidation number
Oxidation number
This oxidation number is average oxidation number of each element (NOT oxidation number is individual atoms) (assuming that all the atoms of the element has the same electron environment)
Oxidation number have no physical or structural significance
For compound with no net charge , all oxidation states must add up to 0
Ion with net charge , OS will add up to that net charge
Ox no of elements in ionic compound is equal to the charge of its ion
For a compound, the more electronegative element is given the negative oxidation number
Very electropositive and electronegative elements always retain the same ON in all their compounds
In PT , electronegativity (ability of atom in a molecule to attract shared electrons in a bond) increases from left to right and down to up
Eg. CIF : F = -1 , CI = +1
Find the oxidation state of oxygen in F2O
O : +2, F : -1
Explain how to find the oxidation number on individual atoms
Pick a bond between 2 atoms.
When the bond is broken , the more electronegative atom will gain one electron from the other atom and have a −1 ON
The less electronegative atom will lose one electron and have a +1 ON
If the bond is a double bond, the oxidation number will be –2 and +2
If 2 elements are the same and bonded together -> there is no difference in electronegativity between the bonded atoms , both atoms are assigned a “0” oxidation number.
Repeat steps 1 to 4 for all the bonds around the atom, and sum up all the oxidation number around an atom to get its overall oxidation number
State some of the common oxidising agents
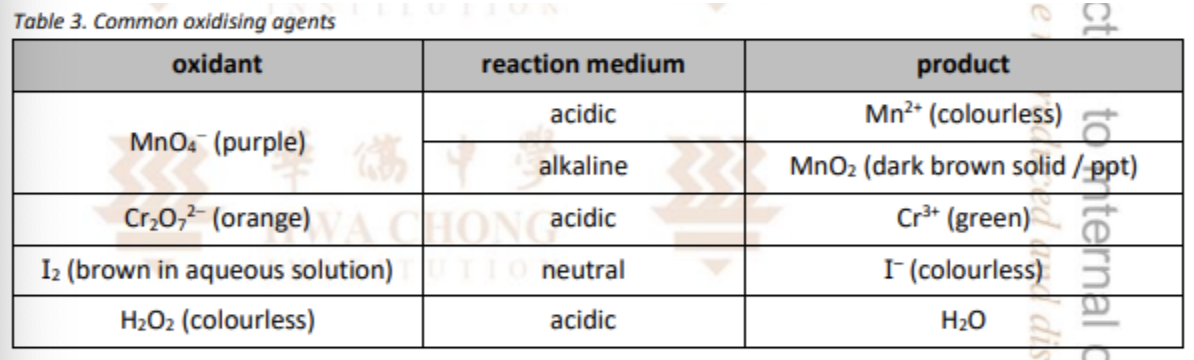
State some of the common reducing agents
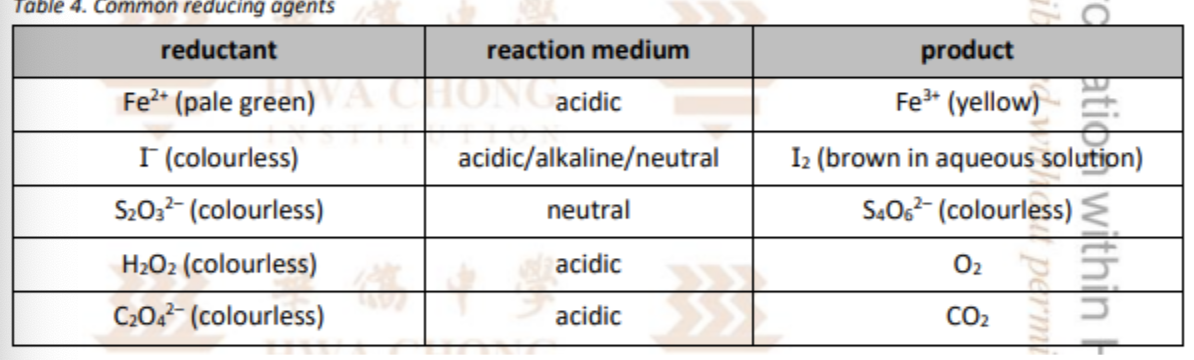
Describe the KMnO4 titration
KMnO4 usually placed in burette and substance to be oxidised + H2SO4 is added to conical flask
Nitric acid and hydrochloric acid are both not suitable as nitric acid is itself an oxidising agent while hydrochloric acid can be oxidised by manganate(VII) ion to give chlorine
MnO4 − + 8H+ + 5e− → Mn2+ + 4H2O
Hydrogen peroxide oxidised to oxygen
When all the reductant is used up, solution turns pink
Explain why volume of H2SO4 used in KMnO4 titration need not be accurate
Dilute H2SO4 is in excess and hence need not be measured accurately.
This is to ensure the solution is sufficiently acidic for the complete reduction of KMnO4
An insufficient acidic medium will result in the formation of a brown precipitate of MnO2 due to incomplete reduction of MnO4– ions leading to a lower titre volume and unreliable results
More acid can be added with vigorous mixing to force the reduction of MnO2 to Mn2+
Describe iodometric titration
2S2O3 2− + I2 → 2I − + S4O6 2−
Only sodium thiosulfate is stable enough to be used as RA in titration as the other RA are easily oxidised by O2 in the air
Only the weak OA, iodine, I2, will oxidise thiosulfate ion, S2O32−, quantitatively to tetrathionate ion, S4O62−
Other stronger oxidising agents would oxidise thiosulfate to a variety of sulfur containing products
The thiosulfate solution is usually placed in the burette and the brown iodine solution in the conical flask
The solution gradually fades to pale yellow
Starch indicator is added into the flask -> Solution turns blue-black
Titration then continues till all iodine is used up (End point : colourless)
Explain why the iodine solution should be titrated as soon as possible once it is prepared
Iodine is volatile and will vaporise easily at room temperature hence titration must be carried out as soon as possible. If this is not possible, cover all flasks containing iodine
Why is the starch indicator added only towards the end of titration when the iodine solution is pale yellow, instead of right at the start?
Starch forms a blue-black water-soluble complex with iodine in which the iodine is trapped within the starch molecules
Hence, starch should not be added at the beginning of the titration when there is a high concentration of iodine since some iodine may remain trapped in the spiral starch molecules -> release of iodine from starch molecules is slow -> even when sufficient _ is added, some iodine is not fully released and mixture still appears blue -> by the time blue colouration disappeared, excess solution has been added to the conical flask -> higher tire values -> inaccurate results
After titration is complete, a slow return of the blue colour is observed. Why is this so?
This is due to atmospheric oxidation of I − back to I2. Ignore any slow return of blue colour after the titration is done