thermal physics + extra
1/95
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
96 Terms
common (not si) unit for temperature
°C
si unit for temperature
K (kelvin)
si unit of energy transferred/work done
J (joules)
si unit of mass
kg (kilograms)
si unit of density
kg/m³
si unit of length
m (metre)
si unit of area
m²
si unit of volume
m³
si unit of speed/velocity
m/s
si unit of acceleration
m/s²
si unit of force
N (newtons)
si unit of pressure
Pa (equivalent to N/m²)
si unit for specific heat capacity
J/kg°C
explain why heating a system will change the energy stored within the system and raise its temperature or produce changes of state
heating a system involves transferring energy to the particles of molecules within the system
this increases the kinetic energy of the particles which means the internal energy of the system increases, which increases the temperature
this can also produce a change of state if the energy the molecules together as opposed to increasing the kinetic energy supplied is used to overcome the bonds between them so the motion and arrangement of particles changes
this does not increase the temp. because no energy goes towards increasing the kinetic energy of the system
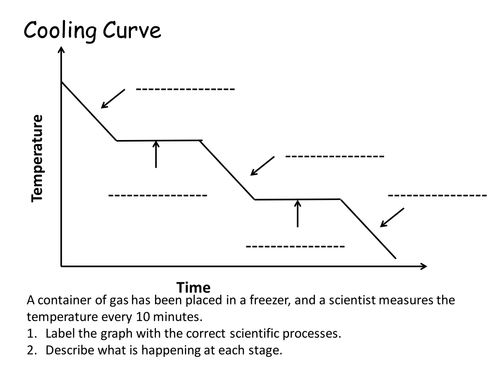
label the points on this cooling graph (the points are the same on a heating graph except they are reversed in order)
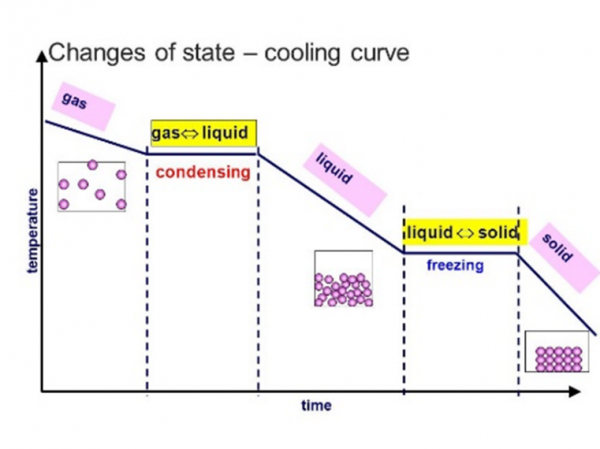
what is it called when a solid heats up into a liquid
melting
what is it called when a solid heats up into a gas
subliming/sublimation
what is it called when a liquid heats up into a gas
boiling/evaporating
what is it called when a gas cools down into a liquid
condensation
what is it called when a liquid cools down into a solid
freezing

is this substance a solid, liquid or gas and how can you tell by its arrangement and presumed motion
solid:
molecules close together in a fixed regular lattice pattern
strong intermolecular forces of attraction
molecules vibrate but can’t move about

is this substance a solid, liquid or gas and how can you tell by its arrangement and presumed motion
liquid:
molecules close together in random arrangement
weaker intermolecular forces than solids
molecules move around each other

is this substance a solid, liquid or gas and how can you tell by its arrangement and presumed motion
gas:
particles far apart in random arrangement
negligible/very weak intermolecular forces
particles are constantly moving with random motion
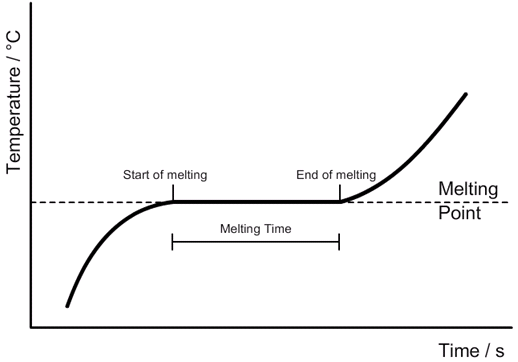
describe an experiment to show constant temperature during a change of state
fill a beaker with boiling water
put ice in the beaker and record the temperature of the ice every 10s as it melts
the results should be a straight line on a temperature/time graph as the energy in the ice goes towards breaking the bonds in the ice rather than increasing its kinetic energy
how is specific heat capacity defined and what is its si unit
specific heat capacity is the amount of energy required to increase the temperature of 1kg of a substance by 1°C
it is measured in J/kg°C
what is the equation for change in thermal energy
ΔQ = m × c × ΔT
Change in thermal energy [J] = Mass [kg] x Specific heat capacity [J/kg 0C] x Change in temperature [0C]
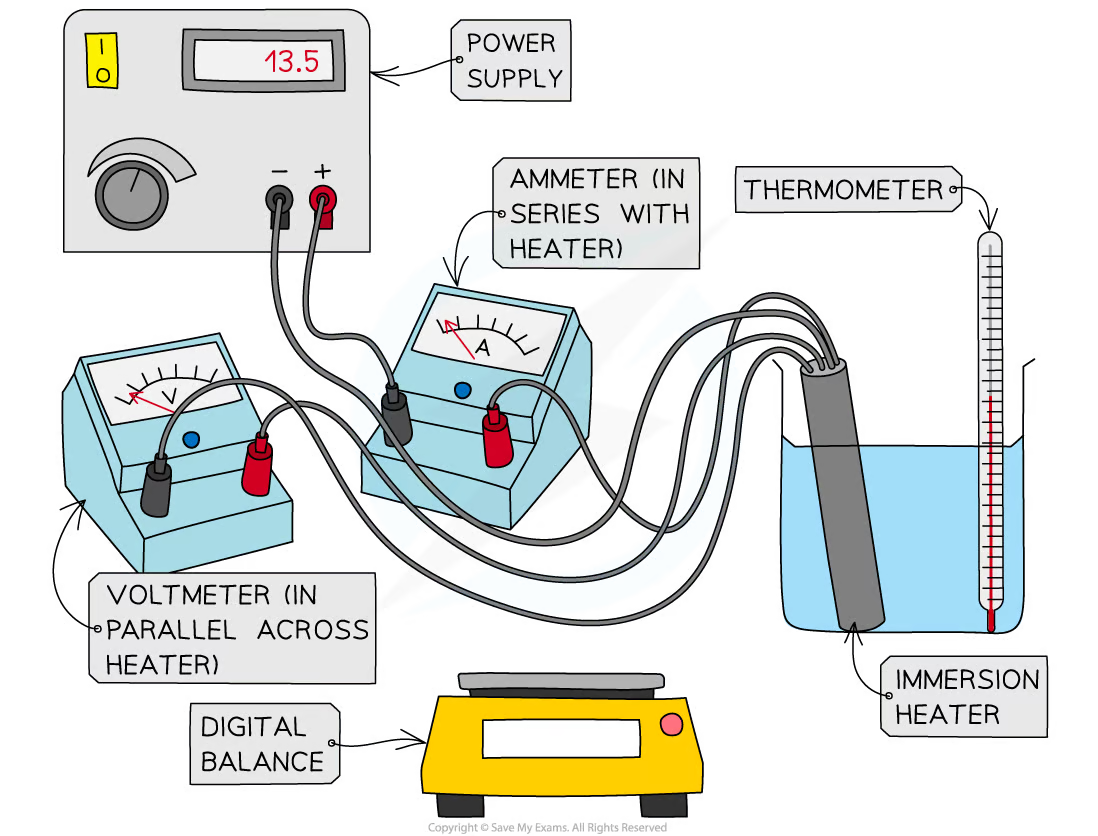
5.14) describe an experiment to investigate the specific heat capacity of materials including water and some solids
measure the mass of an insulating container, fill it with 200ml water and then measure the mass again (the difference between these numbers is the mass of the water)
measure the temperature of water and turn on power which is connected to the water by a immersion heater and connect it to voltmeter and ammeter
wait 1 minute and then measure the water temperature and take voltmeter and ammeter measurements
calculate energy supplied using equation: energy supplied = voltage x current x time
substitute the answer as Q in the equation Q=mcΔT to find specific heat capacity
repeat 3 times to find an average
plot graph
5.7) which changes occur to evaporate/ boil a liquid into a gas
liquids have some kinetic energy
as they are heated, particles vibrate more so their kinetic energy increases
kinetic energy increases → particles vibrate more → frequency of collisions increases → particles get further away from eachother
liquid reaches boiling point when particles are far away enough that their intermolecular forces break and they become gases
5.7) which changes occur to melt a solid into a liquid
solids can’t move so they have no net kinetic energy
as they are heated, the particles vibrate so kinetic energy is gained
kinetic energy increases → particles vibrate more → frequency of collisions increases → particles get further away from eachother and become liquid because they break free of their previous bonds
how do molecules in a gas exert a pressure on the walls of a container
gas molecules have rapid and random motion
when they hit the walls of the container, they exert a force
pressure = force/area and the force exerted from the gas molecules is spread out over the area of the container’s walls
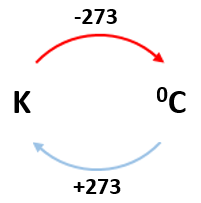
why is 0K absolute zero
at absolute zero the particles have no thermal energy or kinetic energy, so they stop moving
this temperature is called 0K, which is equivalent to -273°C
how to convert between Kelvin and Celsius scales
C = K - 273
K = C + 273
K is always bigger than C
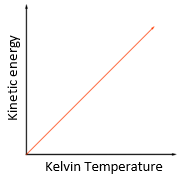
why does an increase in temperature result in an increase in the average speed of gas molecules
as you increase the temperature of a gas
the kinetic energy of the gas particles increases proportionally to temperature
so particles move around with a greater average velocity/speed
what is the relationship between the Kelvin temperature of a gas and the average kinetic energy of its molecules
kelvin temperature is directly proportional to the average kinetic energy of its molecules
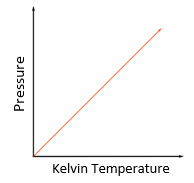
explain the relationship between Kelvin temperature and pressure at constant volume (pressure law)
as a gas is heated, kinetic energy of the particles increases so their average speeds increase
this means there are more collisions per second with the wall, so they exert a greater average force on the wall
this means that the total pressure exerted by the particles increases:
pressure is force / area, and the force here increases while the area stays the same
therefore the relationship between pressure and temperature is directly proportional (as pressure increases, temperature increases and vice versa)
this assumes volume + mass are constant
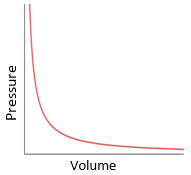
explain the relationship between pressure and volume at constant temperature (boyle’s law)
if temperature is constant, average particle speed is constant
as volume decreases in a container, particle collision frequency per second increases
this is because the same amount of particles are moving around in a smaller volume so they are more likely to hit the sides
more collisions exerted per second on the sides means that the particles exert a greater force on the wall over a given time, so the average force exerted on the walls increases
this means that the total pressure exerted by the particles increases:
pressure is force / area, and the force here increases while the area stays the same
this means that the relationship between pressure and volume is inversely proportional: as volume decreases, pressure increases and vice versa
assumes constant temperature + mass
equation for the relationship between the pressure and Kelvin temperature of a fixed mass of gas at constant volume
P₁/T₁ = P₂/T₂
temperature must always be in kelvin
P₁ = (P₂*T₁)/T₂
P₂ = (P₁*T₂)/T₁
T₁ = (T₂*P₁)/P₂
P₂ = (P₁*T₂)/T₁
if youre arranging for P₁, you need to divide by the opposite letter and the opposite number so you need to divide by T₂. then put the other values (P₂*T₁) over the T₂
equation for the relationship between the pressure and volume of a fixed mass of gas at constant temperature
P₁V₁ = P₂V₂
if youre arranging for P₁, put the value with the same number but the opposite letter underneath the dividing sign (so you put T₁ under the division sign), and then multiply the other numbers over the division sign
so you get (T₂ * P₂)/V₁
5.3) equation for density
density (kg/m3)= mass (kg) / volume (m3)
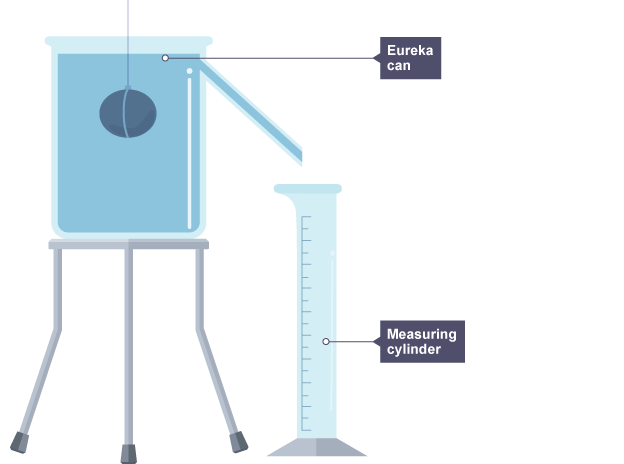
5.4) how can you find the density of a regular or irregular object
regular objects
measure the sides of the object using a ruler
calculate the volume
do mass/volume to find the density
irregular objects
submerge in eureka can and measure the volume of water displaced
do mass/volume to find the density
5.5) equation for pressure
pressure (Pa or N/m2)= force (N) / area (m2)
5.6) describe how pressure moves in fluids at rest
pressure moves equally in all directions
explain why heating a system will change the energy stored within the system and raise its temperature or produce changes of state
heating a system involves transferring energy to the particles of molecules within the system
this increases the kinetic energy of the particles which means the internal energy of the system increases, which increases the temperature
this can also produce a change of state if the energy the molecules together as opposed to increasing the kinetic energy supplied is used to overcome the bonds between them so the motion and arrangement of particles changes
this does not increase the temp. because no energy goes towards increasing the kinetic energy of the system

label the points on this cooling graph (the points are the same on a heating graph except they are reversed in order)

what is it called when a solid heats up into a liquid
melting
what is it called when a solid heats up into a gas
subliming/sublimation
what is it called when a liquid heats up into a gas
boiling/evaporating
what is it called when a gas cools down into a liquid
condensation
what is it called when a liquid cools down into a solid
freezing

is this substance a solid, liquid or gas and how can you tell by its arrangement and presumed motion
solid:
molecules close together in a fixed regular lattice pattern
strong intermolecular forces of attraction
molecules vibrate but can’t move about

is this substance a solid, liquid or gas and how can you tell by its arrangement and presumed motion
liquid:
molecules close together in random arrangement
weaker intermolecular forces than solids
molecules move around each other

is this substance a solid, liquid or gas and how can you tell by its arrangement and presumed motion
gas:
particles far apart in random arrangement
negligible/very weak intermolecular forces
particles are constantly moving with random motion
describe an experiment to show constant temperature during a change of state
fill a beaker with boiling water
put ice in the beaker and record the temperature of the ice every 10s as it melts
the results should be a straight line on a temperature/time graph as the energy in the ice goes towards breaking the bonds in the ice rather than increasing its kinetic energy

how is specific heat capacity defined and what is its si unit
specific heat capacity is the amount of energy required to increase the temperature of 1kg of a substance by 1°C
it is measured in J/kg°C
what is the equation for change in thermal energy
ΔQ = m × c × ΔT
Change in thermal energy [J] = Mass [kg] x Specific heat capacity [J/kg 0C] x Change in temperature [0C]
5.14) describe an experiment to investigate the specific heat capacity of materials including water and some solids
measure the mass of an insulating container, fill it with 200ml water and then measure the mass again (the difference between these numbers is the mass of the water)
measure the temperature of water and turn on power which is connected to the water by a immersion heater and connect it to voltmeter and ammeter
wait 1 minute and then measure the water temperature and take voltmeter and ammeter measurements
calculate energy supplied using equation: energy supplied = voltage x current x time
substitute the answer as Q in the equation Q=mcΔT to find specific heat capacity
repeat 3 times to find an average
plot graph

5.7) which changes occur to evaporate/ boil a liquid into a gas
liquids have some kinetic energy
as they are heated, particles vibrate more so their kinetic energy increases
kinetic energy increases → particles vibrate more → frequency of collisions increases → particles get further away from eachother
liquid reaches boiling point when particles are far away enough that their intermolecular forces break and they become gases
5.7) which changes occur to melt a solid into a liquid
solids can’t move so they have no net kinetic energy
as they are heated, the particles vibrate so kinetic energy is gained
kinetic energy increases → particles vibrate more → frequency of collisions increases → particles get further away from eachother and become liquid because they break free of their previous bonds
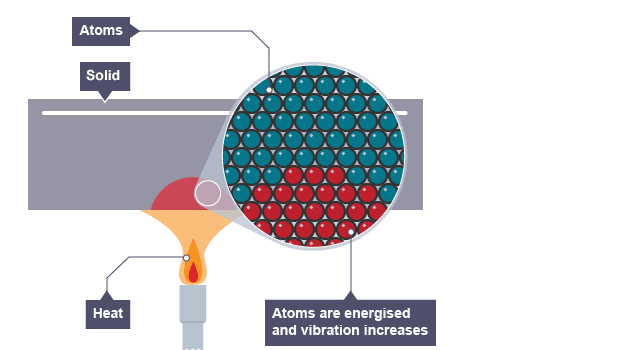
how does conduction work in non-metal solids or liquids (insulators)
solid is heated up
the heat energises the molecules so vibration (kinetic energy) increases
the vibration (high kinetic energy) slowly moves throughout the solid because as the molecules vibrate more they hit adjacent molecules, making them vibrate more too and increasing their kinetic energy
this transfers heat energy from hotter to cooler parts of the non-metal
how does conduction work in metals and how does it compare to that of insulator solids or liquids
some electrons in a metal can leave their atoms and move around the atom as a free (delocalised) electron, leaving metal ions behind
metals are already good conductors
this means when the free electrons absorb heat energy, they move much faster so they are more likely to crash into metal ions
some of the kinetic energy is absorbed by the ions so they vibrate faster and with greater amplitude, transferring heat energy from hotter to colder parts of the metal
this is faster than conduction in non-metals/insulators, where conduction is caused by vibrations passed between atoms because they dont have free electrons

what is conduction
the transferral of thermal energy in solids or liquids by the vibration of particles
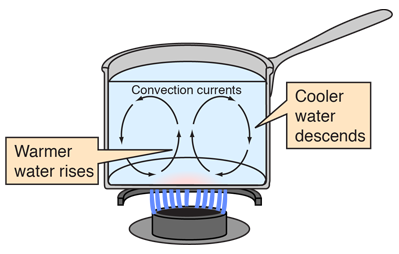
how do convection currents work in fluids
energy is transferred from heat source to air
particles move further apart so the fluid expands
it then rises up because it’s less dense
as it rises, it cools because it transfers heat to its surroundings so it becomes less dense and sinks
cooler air becomes denser and sinks
the corners of a beaker or room which aren’t affected by the heating can push the fluid around
this process repeats
how does radiation happen and where can it happen
all bodies emit infrared radiation
the hotter an object, the more infrared radiation it radiates in a given time
radiation can operate in a vacuum because it doesn’t require particles to transfer energy
rank the absorbing and emitting abilities of thermal radiation of:
shiny, dull/dark, black, white surfaces
from best to worst
black
best emitter/absorber
thermal energy is radiated,
dull/dark, white, shiny
(shiny surfaces reflect light, not absorb it)
worst emitter/absorber
thermal energy is reflected
what are some examples of convection in everyday phenomena
radiators
fridges
weather
air conditioning (uses reverse convection currents, i.e cooling current)
why does convection work only in fluids
in gases and liquids the particles can move past each other but in solids they can’t
how can you stop unwanted convection (currents)
stop the free flow of fluids by containing/reducing area of the convection current
for example, wearing a blanket reduces air movement, so it reduces the convection current
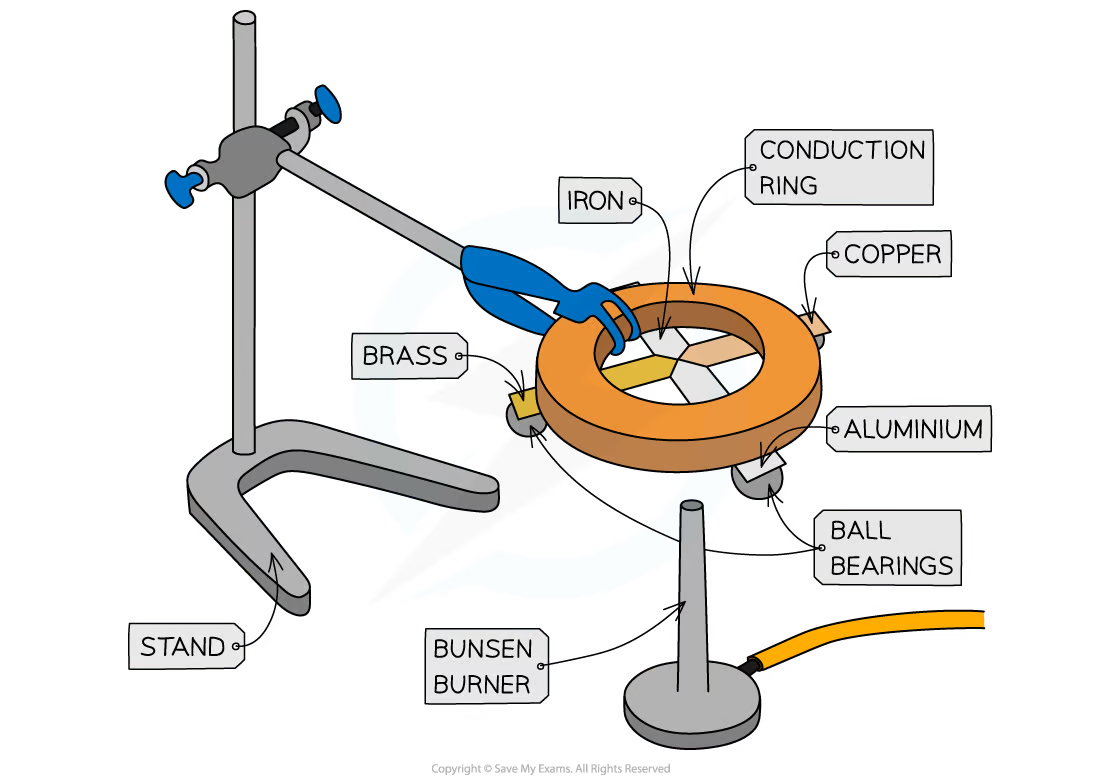
describe an experiment to investigate conduction
use a clamp and stand to secure a conduction ring over a bunsen burner
attach ball bearings to the ends of the metal strips using wax
turn the strips upside down and heat the centre so each of the strips is heated at the central point where they meet
when heat is conducted along to the ball bearing the wax melts and the ball bearing drops
time how long this takes for the strips and record in a table
repeat and find an average
independent var: type of metal
dependent var: rate of conduction
control var: size and thickness of metal strips
describe an experiment to investigate convection
fill a beaker with cold water and put it on top of a tripod and heatproof mat
pick up the potassium permanganate crystals and drop them into the centre of the beaker
heat the beaker with a bunsen burner and record observations
repeat experiment with hot water and record observations
convection currents will be formed in both beakers but the current is faster in hot water
independent var: temp of water
dependent var: rate of convection
control vars: Amount of water in beaker, Size of bunsen burner flame, Size of potassium permanganate crystal
describe an experiment to investigate radiation (6)
make sure theres an equal volume of water in each bottle
place both bottles an equal distance away from an infrared heater
the bottles should have the same heater output
make sure the starting temperature of the water is the same
measure the temperature of water in the bottles after a given time
repeat the investigation more than once
calculate a mean
take care to avoid burns from heater/clean up water spillages
how do molecules in a gas exert a pressure on the walls of a container
gas molecules have rapid and random motion
when they hit the walls of the container, they exert a force
pressure = force/area and the force exerted from the gas molecules is spread out over the area of the container’s walls
why is 0K absolute zero
at absolute zero the particles have no thermal energy or kinetic energy, so they stop moving
this temperature is called 0K, which is equivalent to -273°C
how to convert between Kelvin and Celsius scales
C = K - 273
K = C + 273
K is always bigger than C

why does an increase in temperature result in an increase in the average speed of gas molecules
as you increase the temperature of a gas
the kinetic energy of the gas particles increases proportionally to temperature
so particles move around with a greater average velocity/speed
what is the relationship between the Kelvin temperature of a gas and the average kinetic energy of its molecules
kelvin temperature is directly proportional to the average kinetic energy of its molecules

explain the relationship between Kelvin temperature and pressure at constant volume (pressure law)
as a gas is heated, kinetic energy of the particles increases so their average speeds increase
this means there are more collisions per second with the wall, so they exert a greater average force on the wall
this means that the total pressure exerted by the particles increases:
pressure is force / area, and the force here increases while the area stays the same
therefore the relationship between pressure and temperature is directly proportional (as pressure increases, temperature increases and vice versa)
this assumes volume + mass are constant
explain the relationship between pressure and volume at constant temperature (boyle’s law)
if temperature is constant, average particle speed is constant
as volume decreases in a container, particle collision frequency per second increases
this is because the same amount of particles are moving around in a smaller volume so they are more likely to hit the sides
more collisions exerted per second on the sides means that the particles exert a greater force on the wall over a given time, so the average force exerted on the walls increases
this means that the total pressure exerted by the particles increases:
pressure is force / area, and the force here increases while the area stays the same
this means that the relationship between pressure and volume is inversely proportional: as volume decreases, pressure increases and vice versa
assumes constant temperature + mass
equation for the relationship between the pressure and Kelvin temperature of a fixed mass of gas at constant volume
P₁/T₁ = P₂/T₂
temperature must always be in kelvin
P₁ = (P₂*T₁)/T₂
P₂ = (P₁*T₂)/T₁
T₁ = (T₂*P₁)/P₂
P₂ = (P₁*T₂)/T₁
if youre arranging for P₁, you need to divide by the opposite letter and the opposite number so you need to divide by T₂. then put the other values (P₂*T₁) over the T₂

equation for the relationship between the pressure and volume of a fixed mass of gas at constant temperature
P₁V₁ = P₂V₂
if youre arranging for P₁, put the value with the same number but the opposite letter underneath the dividing sign (so you put T₁ under the division sign), and then multiply the other numbers over the division sign
so you get (T₂ * P₂)/V₁

how does conduction work in non-metal solids or liquids (insulators)
solid is heated up
the heat energises the molecules so vibration (kinetic energy) increases
the vibration (high kinetic energy) slowly moves throughout the solid because as the molecules vibrate more they hit adjacent molecules, making them vibrate more too and increasing their kinetic energy
this transfers heat energy from hotter to cooler parts of the non-metal

how does conduction work in metals and how does it compare to that of insulator solids or liquids
some electrons in a metal can leave their atoms and move around the atom as a free (delocalised) electron, leaving metal ions behind
metals are already good conductors
this means when the free electrons absorb heat energy, they move much faster so they are more likely to crash into metal ions
some of the kinetic energy is absorbed by the ions so they vibrate faster and with greater amplitude, transferring heat energy from hotter to colder parts of the metal
this is faster than conduction in non-metals/insulators, where conduction is caused by vibrations passed between atoms because they dont have free electrons

what is conduction
the transferral of thermal energy in solids or liquids by the vibration of particles
how do convection currents work in fluids
energy is transferred from heat source to air
particles move further apart so the fluid expands
it then rises up because it’s less dense
as it rises, it cools because it transfers heat to its surroundings so it becomes less dense and sinks
cooler air becomes denser and sinks
the corners of a beaker or room which aren’t affected by the heating can push the fluid around
this process repeats

how does radiation happen and where can it happen
all bodies emit infrared radiation
the hotter an object, the more infrared radiation it radiates in a given time
radiation can operate in a vacuum because it doesn’t require particles to transfer energy
rank the absorbing and emitting abilities of thermal radiation of:
shiny, dull/dark, black, white surfaces
from best to worst
black
best emitter/absorber
thermal energy is radiated,
dull/dark, white, shiny
(shiny surfaces reflect light, not absorb it)
worst emitter/absorber
thermal energy is reflected
what are some examples of convection in everyday phenomena
radiators
fridges
weather
air conditioning (uses reverse convection currents, i.e cooling current)
why does convection work only in fluids
in gases and liquids the particles can move past each other but in solids they can’t
how can you stop unwanted convection (currents)
stop the free flow of fluids by containing/reducing area of the convection current
for example, wearing a blanket reduces air movement, so it reduces the convection current
describe an experiment to investigate conduction
use a clamp and stand to secure a conduction ring over a bunsen burner
attach ball bearings to the ends of the metal strips using wax
turn the strips upside down and heat the centre so each of the strips is heated at the central point where they meet
when heat is conducted along to the ball bearing the wax melts and the ball bearing drops
time how long this takes for the strips and record in a table
repeat and find an average
independent var: type of metal
dependent var: rate of conduction
control var: size and thickness of metal strips

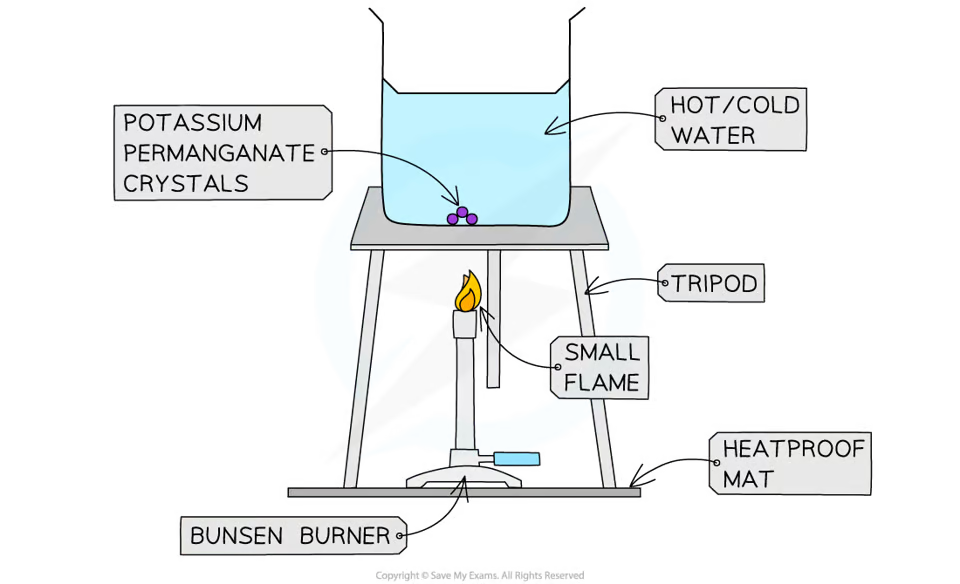
describe an experiment to investigate convection
fill a beaker with cold water and put it on top of a tripod and heatproof mat
pick up the potassium permanganate crystals and drop them into the centre of the beaker
heat the beaker with a bunsen burner and record observations
repeat experiment with hot water and record observations
convection currents will be formed in both beakers but the current is faster in hot water
independent var: temp of water
dependent var: rate of convection
control vars: Amount of water in beaker, Size of bunsen burner flame, Size of potassium permanganate crystal

describe an experiment to investigate radiation (6)
make sure theres an equal volume of water in each bottle
place both bottles an equal distance away from an infrared heater
the bottles should have the same heater output
make sure the starting temperature of the water is the same
measure the temperature of water in the bottles after a given time
repeat the investigation more than once
calculate a mean
take care to avoid burns from heater/clean up water spillages

what are some situations where you want to reduce energy transfer
Keeping a house warm
Keeping a hot drink hot or cold
Dressing to stay warm in cold weather
how to reduce conduction
use materials with a low thermal conductivity: insulators
how does insulation keep something warm
The insulator contains the trapped air, which is a poor thermal conductor
Trapping the air also prevents it from transferring energy by convection
This reduces the rate of energy transfer from the object, meaning that it will stay warmer for longer
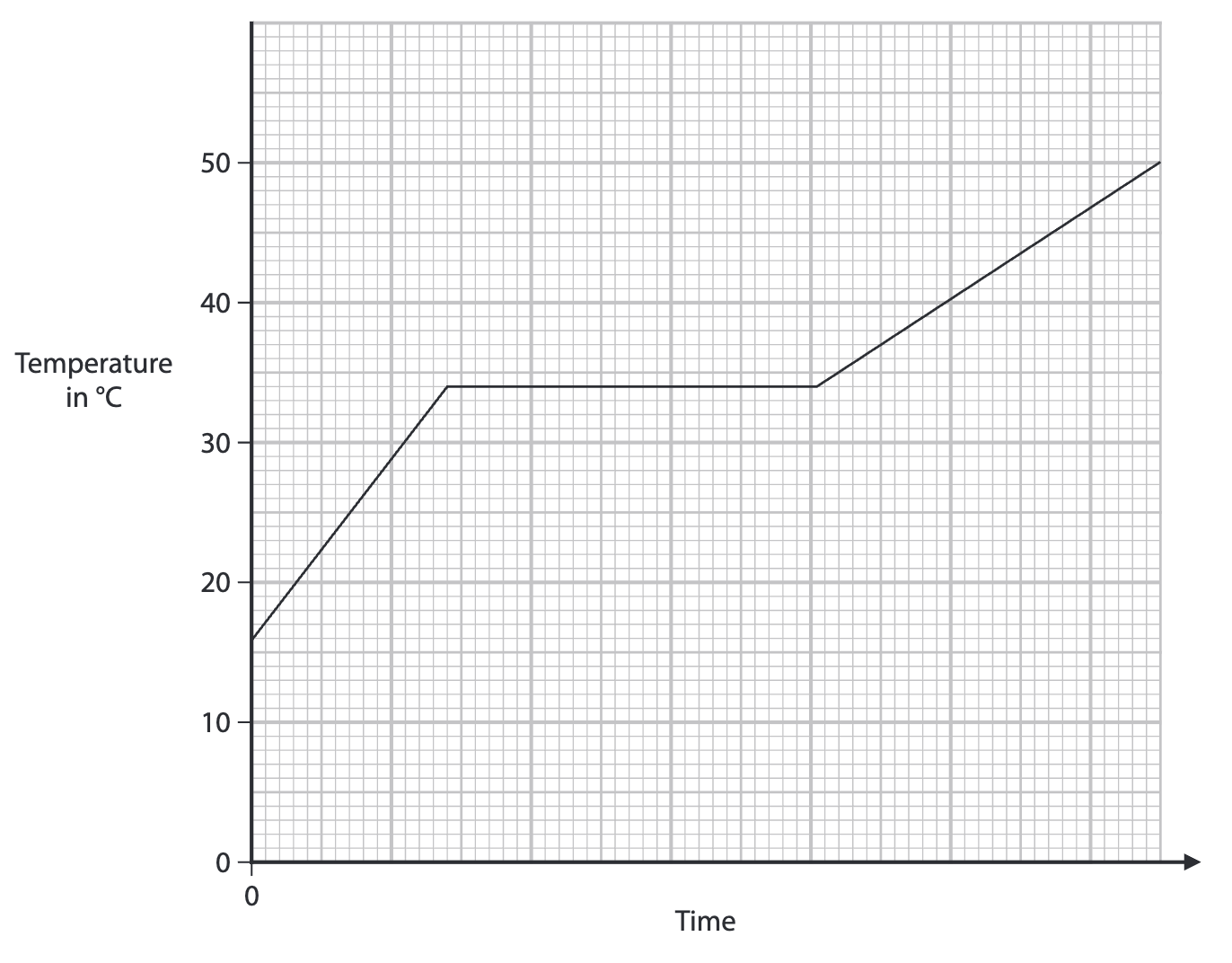
on a heating graph why do the two lines have different gradients
the substance is at different states here
so they have different specific heat capacities
so one has a faster rate of reaction than the other
what do you need for conduction/convection/radiation to occur
conduction: physical contact between two things
convection: fluids (particles that have net movement) and a gap between objects, i.e not next to each other
radiation: very different temperatures