C1.1 Enzymes and Metabolism Review (IB Bio SL)
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
IB definition of metabolism
The complex network of interdependent and interacting chemical reactions occurring in living organisms through the use of enzymes (Simplified: All enzyme catalyzed reactions
Enzyme
A substance produced by a living organism that acts as a catalyst to bring about a specific biochemical reaction
Catalyst
A substance that increases the rate of a chemical reaction without itself undergoing any permanent chemical change
Why are enzymes necessary?
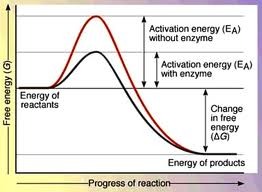
All chemical reactions require some amount of activation energy to react the activation energy without enzymes would be more than the organism can supply enzymes work to lower this activation energy to one that the organism can provide
Need to be able to interpret this graph
Catalysis
The acceleration of a chemical reaction by a catalyst
What type of protein are enzymes?
Globular protein (meaning that is is roughly globe-shaped)
Structure of enzymes
Enzymes are globular proteins with an active site for catalysis. The active site is a small handful of amino acids that directly bind to the substrate (the overall structure affects binding though)
Substrate
A specific reactant (what is put in) acted upon by an enzyme
Induced fit model
Both the enzyme and the substrate change shape when binding occurs
Anabolic reaction
Any condensation reaction that forms polymers
Examples of anabolic reactions
Protein synthesis, glycogen formation, photosynthesis
Catabolic reaction
Any hydrolysis reaction that breaks down macromolecules into monomers
Examples of catabolic reactions
Digestion, oxidation in respiration
What must happen in order for a reaction to occur between a substrate and enzyme?
They must collide
How does temperature affect the likelihood of a reaction between an enzyme and substrate?
Temperature is the speed of molecules, so the higher the temperature, the faster molecules are moving. The faster the molecules are moving, the more likely they are to collide
How can the likelihood of a collision between an enzyme and substrate increase?
Increase the temperature
Embed either the enzyme or substrate into a membrane and lock it in place
How to measure the rate of reaction in an enzyme-catalyzed reaction
Use the rate of reaction equation: Rate of reaction = change in concentration of substance over time Rate = ΔB/Δt = -ΔA/Δt
Reactants go down, products go up
Factors that affect enzyme activity
Substrate concentration, pH, and temperature
Temperature vs. enzyme activity
As temperature increases, enzyme activity linearly increases, then sharply declines as enzymes denature past the optimum temperature (Only higher temperatures cause denaturation, lower temperatures simply cause the enzyme to move slower)
pH vs. enzyme activity
Enzymes denature on both sides of the optimum
Substrate concentration on enzyme activity
As substrate concentration increases, enzyme activity rapidly increases, then plateaus once all enzymes are being used
Relationship between structure of active site, enzyme-substrate specificity, and denaturation
Enzymes are shaped specifically for their substrate, so if an enzyme denatures (changes shape), it no longer works