Chemical Systems and Equilibrium -Equilibrium Calculations
1/34
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
reaction quotient (Q)
a numerical value determined by using the same formula as the equilibrium constant (data for a reversible reaction) that may or may not be at equilibrium
using Q to det the dir of a rxn
rxn would move in the dir from Q to K
Q < K
more reactants than products at the current concentrations, therefore the rxn must proceed RIGHT
Q > K
more products than reactants at the current concentrations, therefore the rxn would proceed left
calc eq concentrations from K and initial concentrations steps
balanced chem eq
calc initial concentrations if necessary
CALC Q and det the concen shift dir so you know what is pos and neg on the ICE table
make ICE table
place all eq expressions for each substance in the K expression and isolate for x
three ways to calc eq concens
perfect square (no quad as you sqr root both sides at some point)
small k simplication (100 rule)
quad formula
small k simplication
when k is vv small so you can assume the rxn hardly proceeds and can assume the eq concen is almost the same as initial concens
remove x term associated w the SMALLEST NON-ZERO CONCEN
use 100 rule
small k simplification : 100 rule
is the ratio of [initial concen of reactant'] / K is greater than 100, then x is vv small so it can be removed from that part of the expression
K constant for solubility
look at the coefficients in the BALANCED NIE WITH THE IONS
molar solubility
the value of x in K solubility solving questions
recall the definition of solubility
solubility
the max amount of an ionic compound that will dissolve in a given volume of solvent at a particular temp
(solubility) use Q and K to det if a precipitate forms : Q < K
equilibrium shifts right therefore no precipitate bc less ions are present than what is needed to saturate the solution
(solubility) use Q and K to det if a precipitate forms : Q > K
equil shifts left; more ions are present than what is required to saturate the solution therefore it is supersaturated to produces a precipitate
common ion effect
a DECREASE in solubility of an ionic compound due to the presence of a common ion in solution
consistent with Le Chatelier’s Principle
AgCl(s) ⇌ Ag+ (aq) + Cl- (aq)
• Adding NaCl decreases the solubility (ie. quantity of solute that dissolves) of AgCl because there are now more Cl- (chloride anions) in the solution – reaction shifts to the left
another definition of solubility
quantity of solute that dissolves
the guy who made that one principle
Le Chatelier
eq for K constant
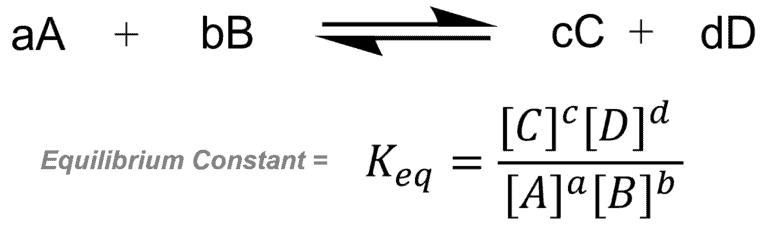
K constant would change according to the
temp of the system
homogeneous equilibria
equilibria where the reactants and products are all in the same phase.
heterogeneous equilibria
reactions and products are in diff phases
concentration of liquids and solids are constant bc
their concentrations are fixed and equal to its density
EX: Since a litre of water at SATP is 1.00kg, which is equal to 55.5 mol, water has a “concentration” of 55.5mol/L
super large Keq would
favour products
very small Keq would
favour reactants
K approx = 0
favours neither products or reactants
Keq’
equilibrium constant for the reverse rxn (1 / Keq)
le chatelier’s principle
“...when a chemical system at equilibrium is disturbed by a change in a property, the system adjusts in a way that OPPOSES the change.”
factors like temp, pressure, and concen can affect equilibrium therefore causing an equilibrium shift
addition of a reactant causes the rxn to
proceed in the direction that consumes the added substance (go towards the prods aka the right, other way if it was the addition of products)
removing a substance causes the rxn to
proceed in the direction that produces the missing substance
when adding a substance
the rate of the forward and reverse rxns are slightly faster than the initial eq bc more particles would be present
when adding a reactant
the forward rxn (rxn shifts right) increases which causes the reverse rxn to also pick up, and the forward rxn will start to slow as the reactants are used up
temp shifting a rxn
if mixture is cooled, rxn goes towards the side that is producing heat (energy)
if mixture is heated, rxn goes towards the side that is NOT producing heat (energy)
le chatelier and gas volume changes
only for eqns that are all gases
decrease in volume (increase in pressure) causes rxn to proceed in the dir of FEWER # OF MOLES
increase in volume causes rxn to proceed in the dir of more moles
IF THERE IS THE SAME # OF MOLES ON BOTH SIDES, the rxn does not shift
if there’s a decrease in V, then both the forward and reverse rxns should
increase in rate due to increased concens on both sides
changes that do not affect the posi of equilibrium sys’s
adding catalysts (would speed up both the reverse and forward rxn)
adding inert gases (noble gases)
less volume
more concen