Classification of Matter, Classification of Matter
1/33
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
34 Terms
physical property
characteristic that can be observed without changing the makeup of the substance

chemical property
characteristic that describes how a substance will react to form other substances

physical change
process that does not alter the composition of a substance

chemical change
process involving a new substance being formed

melting
The change of state from a solid to a liquid

freezing
The change of state from a liquid to a solid

evaporation, vaporization, boiling
change from a liquid to a gas

condensation
process of a gas to a liquid

sublimation
process from a solid to a gas without going through the liquid state

melting point
temperature at which a solid changes to a liquid

boiling point
the temperature at which substance changes from a liquid to a gas

element
contains one kind of atom and is found on the Periodic Table
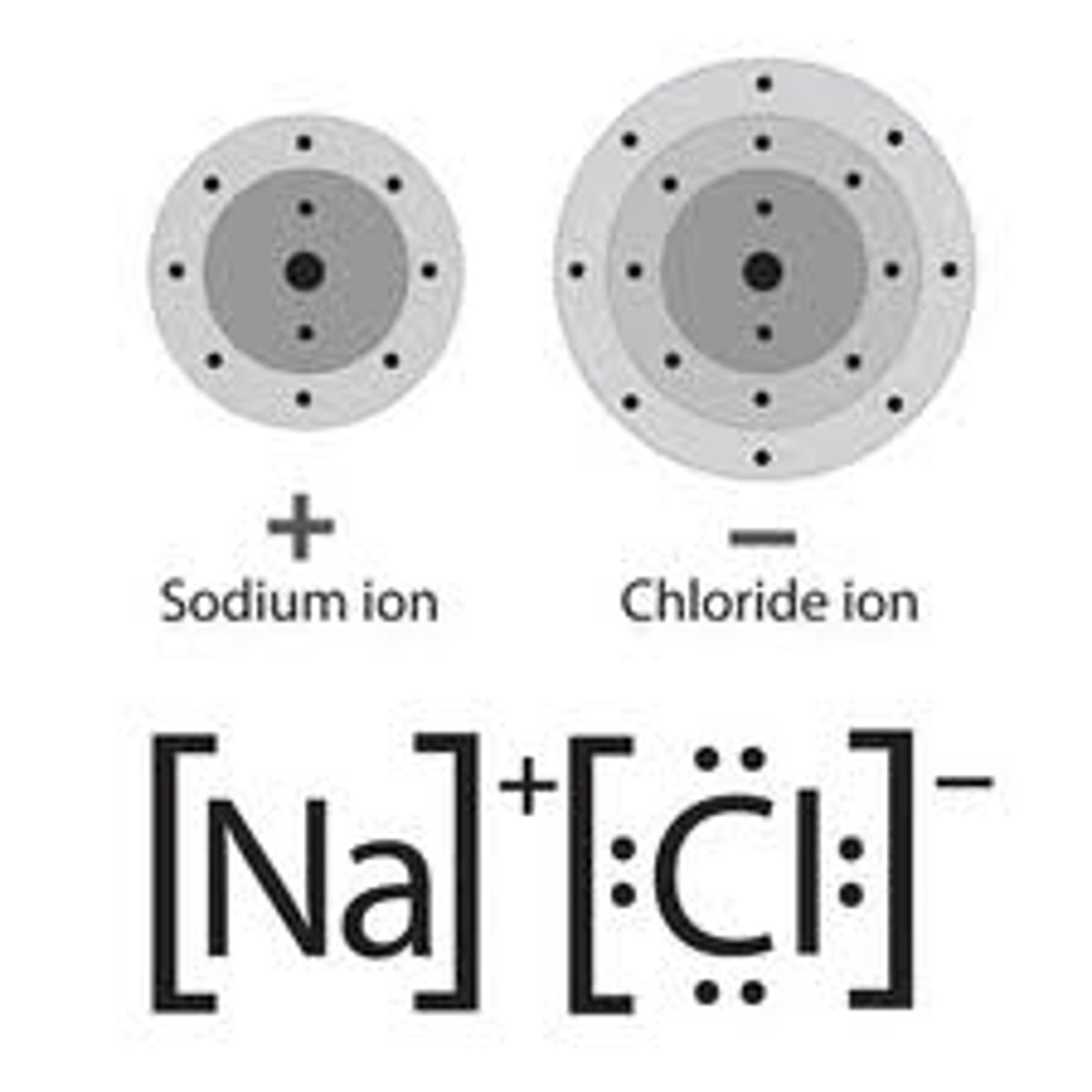
compound
two or more different kinds of atoms bonded together in the same ratio all the time
mixture
two or more substances physically combined so that each retains its identity

homogeneous mixture
2 or more substances evenly mixed together and they appear as one substance

heterogeneous mixture
2 or more substances mixed together and you can see the different parts

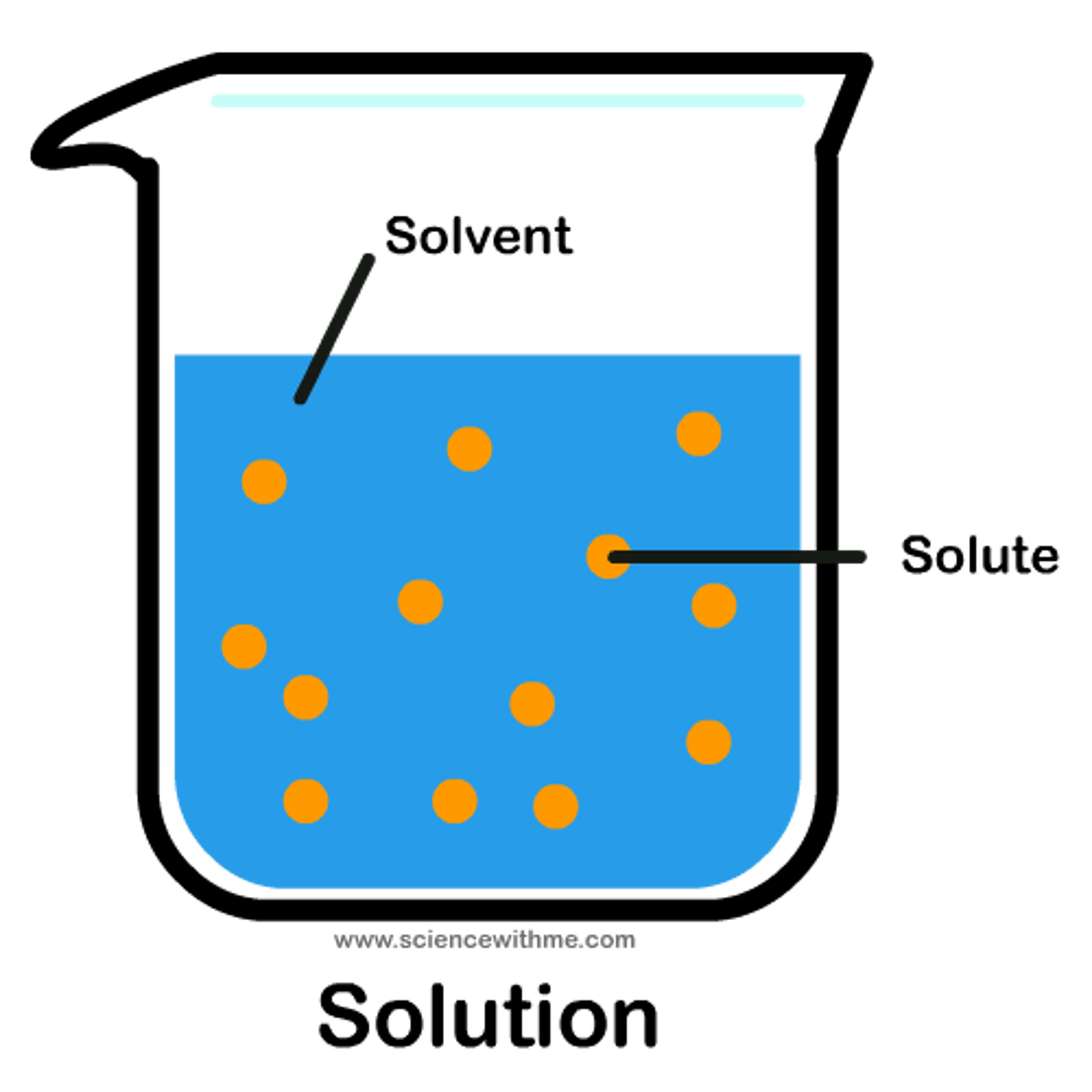
solution
homogeneous mixture with minute particles spread throughout usually liquid form.
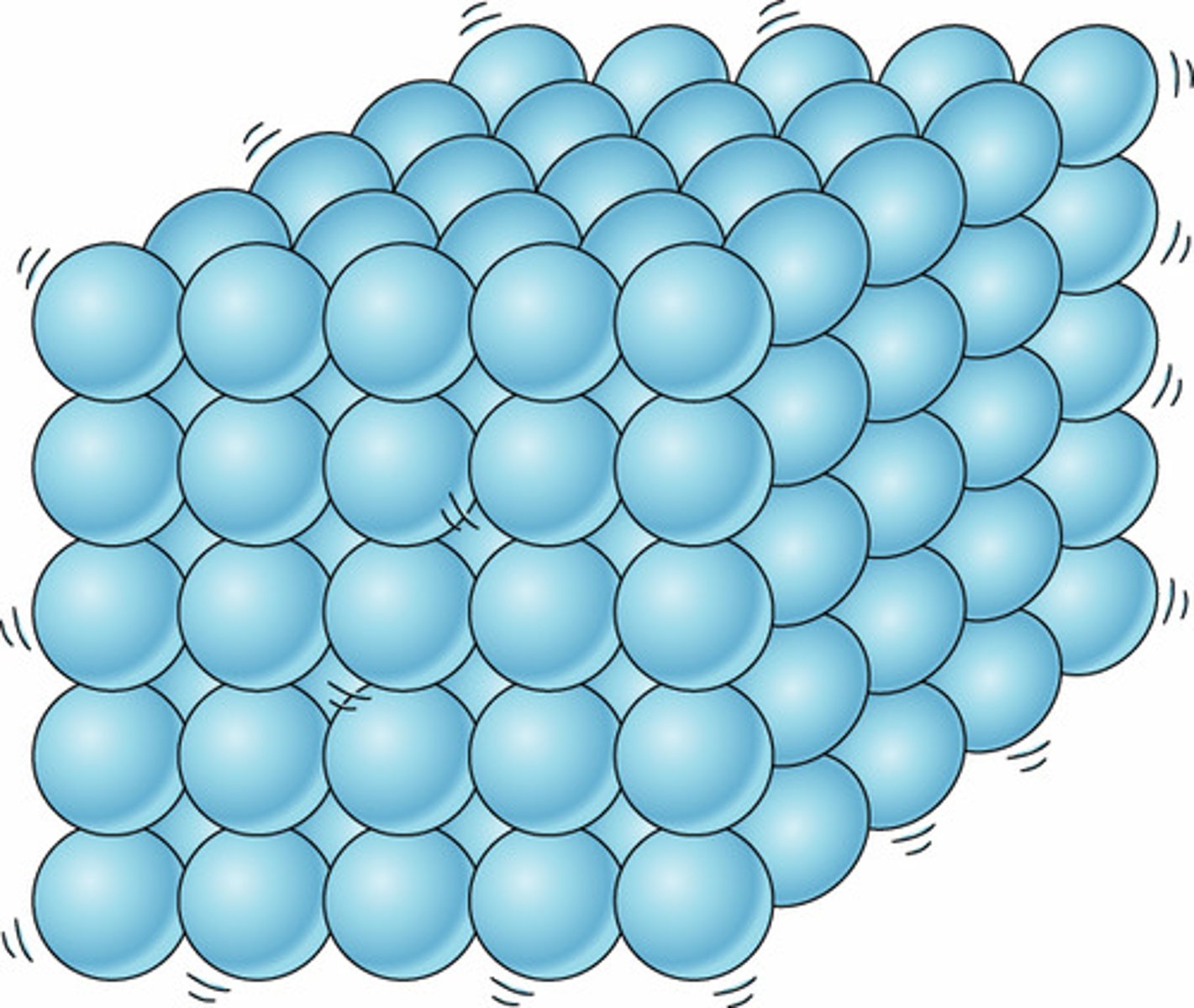
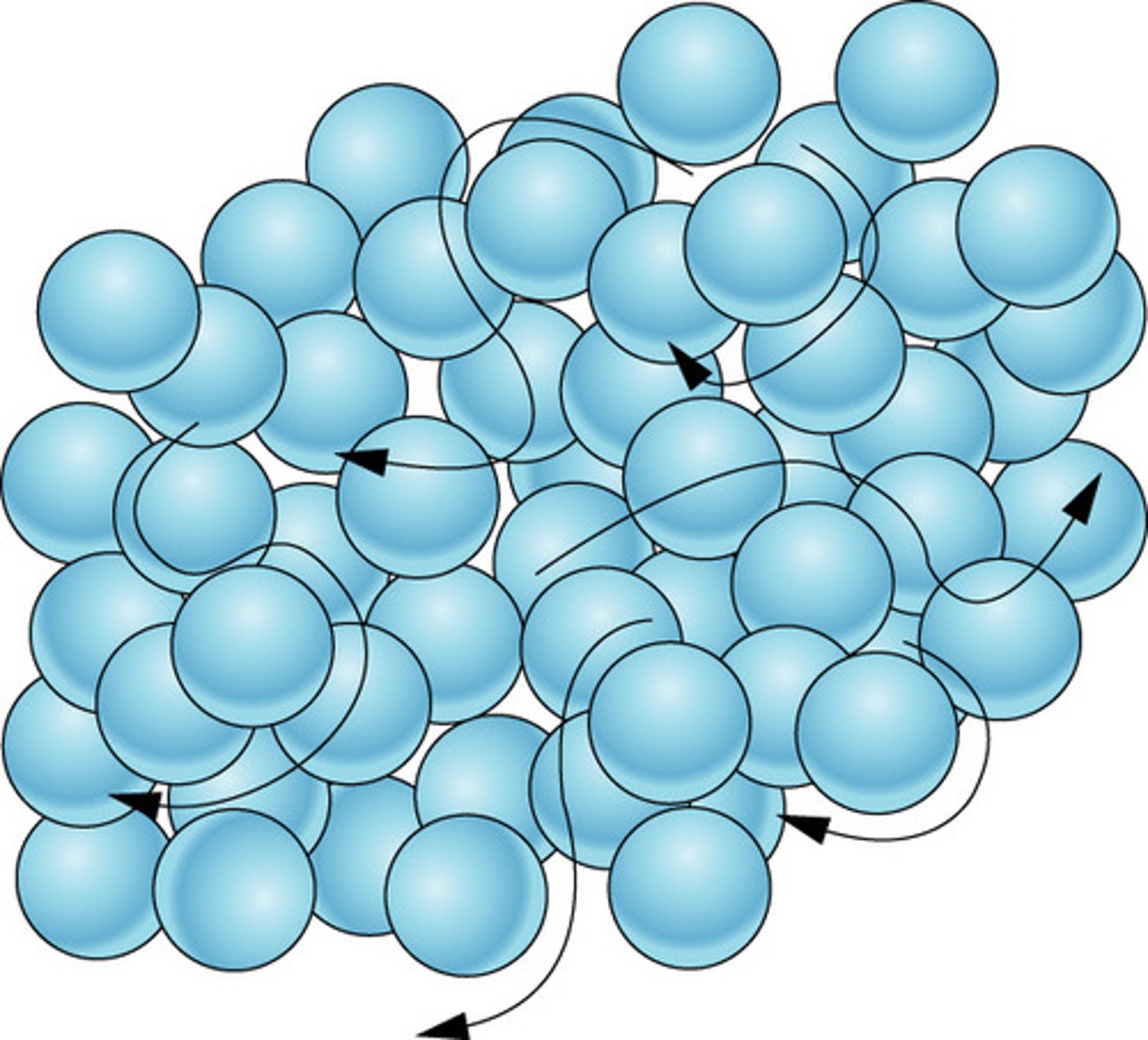
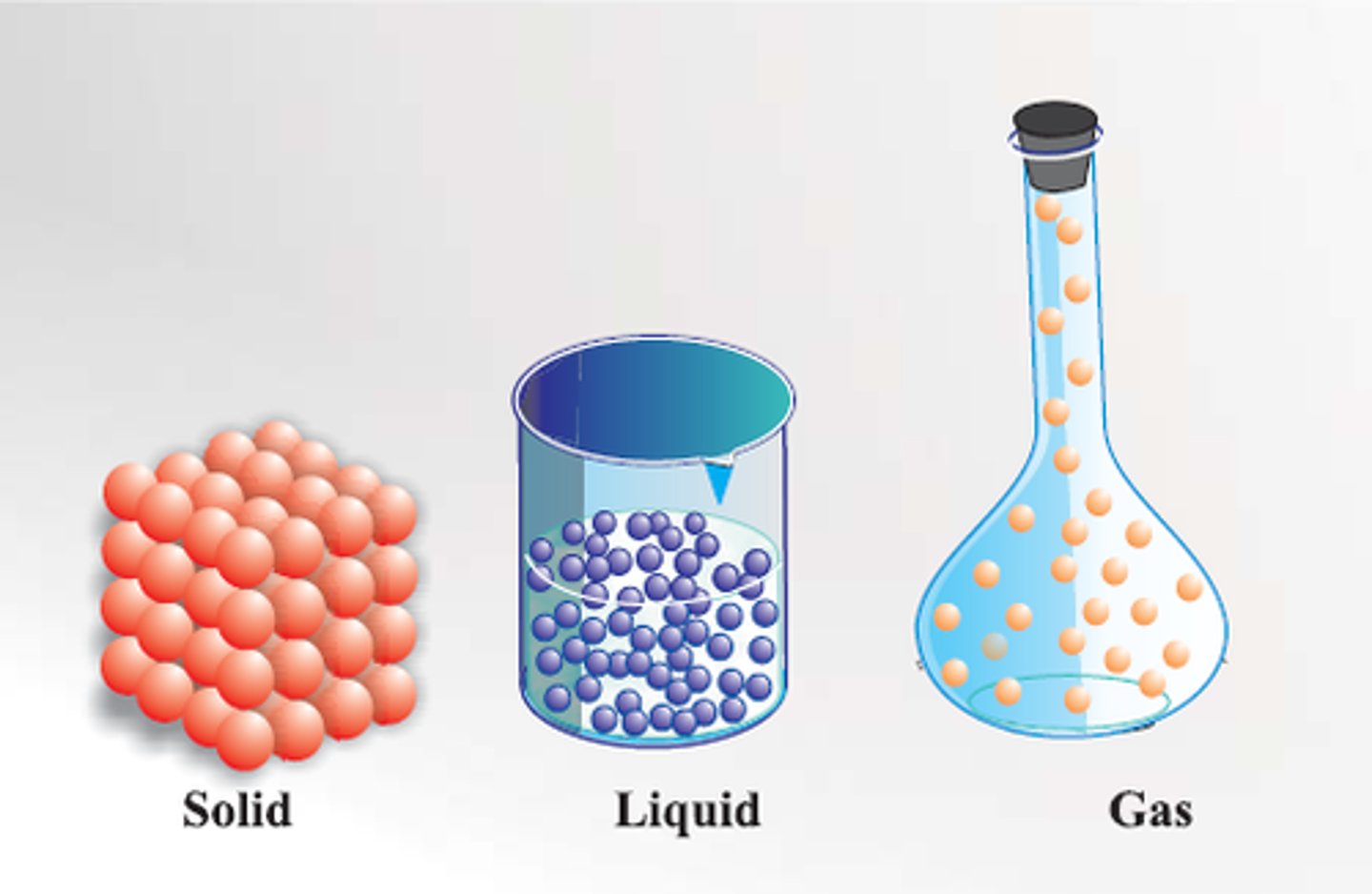
solid
definite shape and volume; close together and little movement
liquid
definite volume but no definite shape; some movement
gas
no definite volume or shape; particles move fast and are far apart
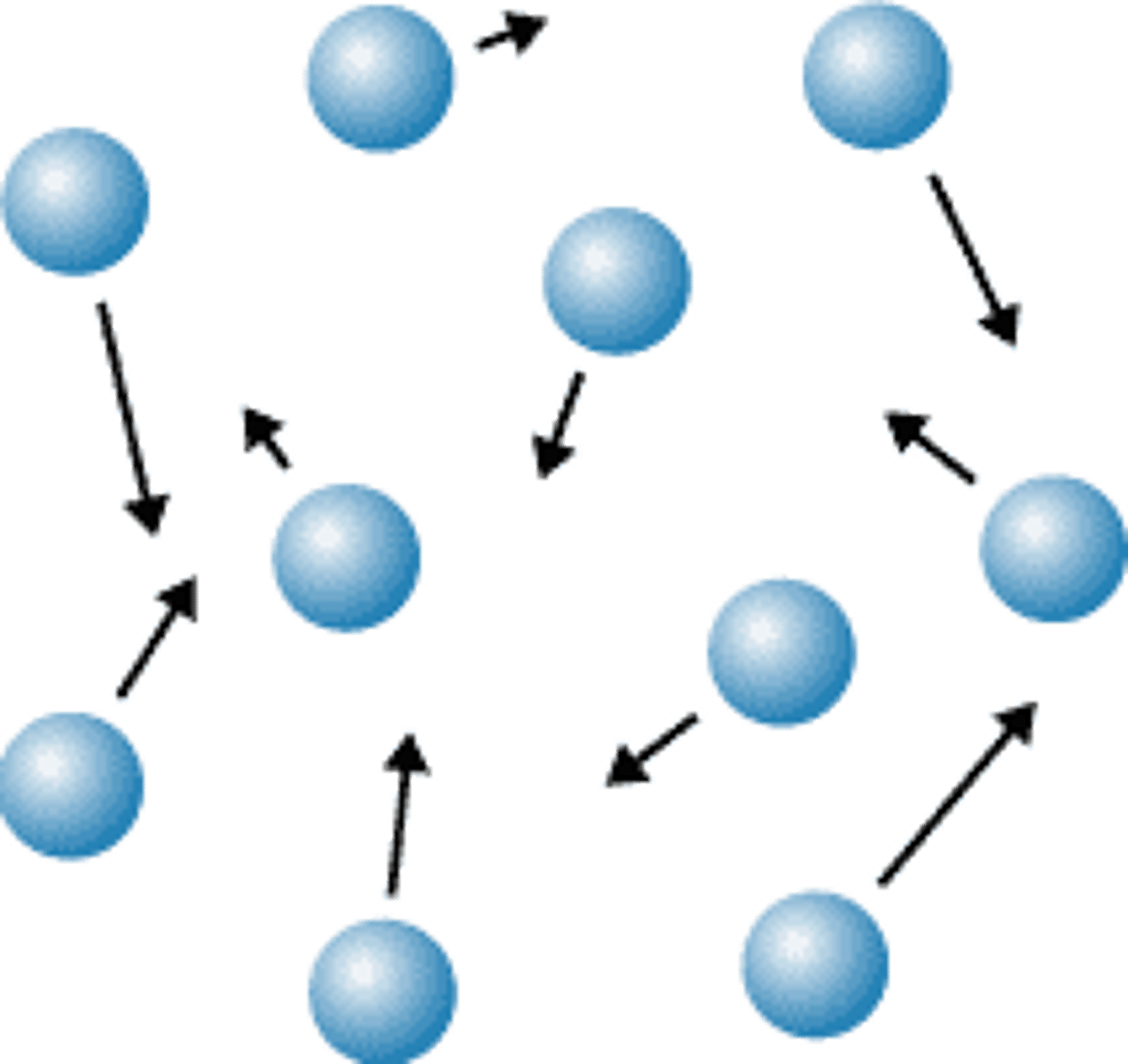
deposition
process from a gas to a solid without going through the liquid state

matter
Anything that has mass and takes up space
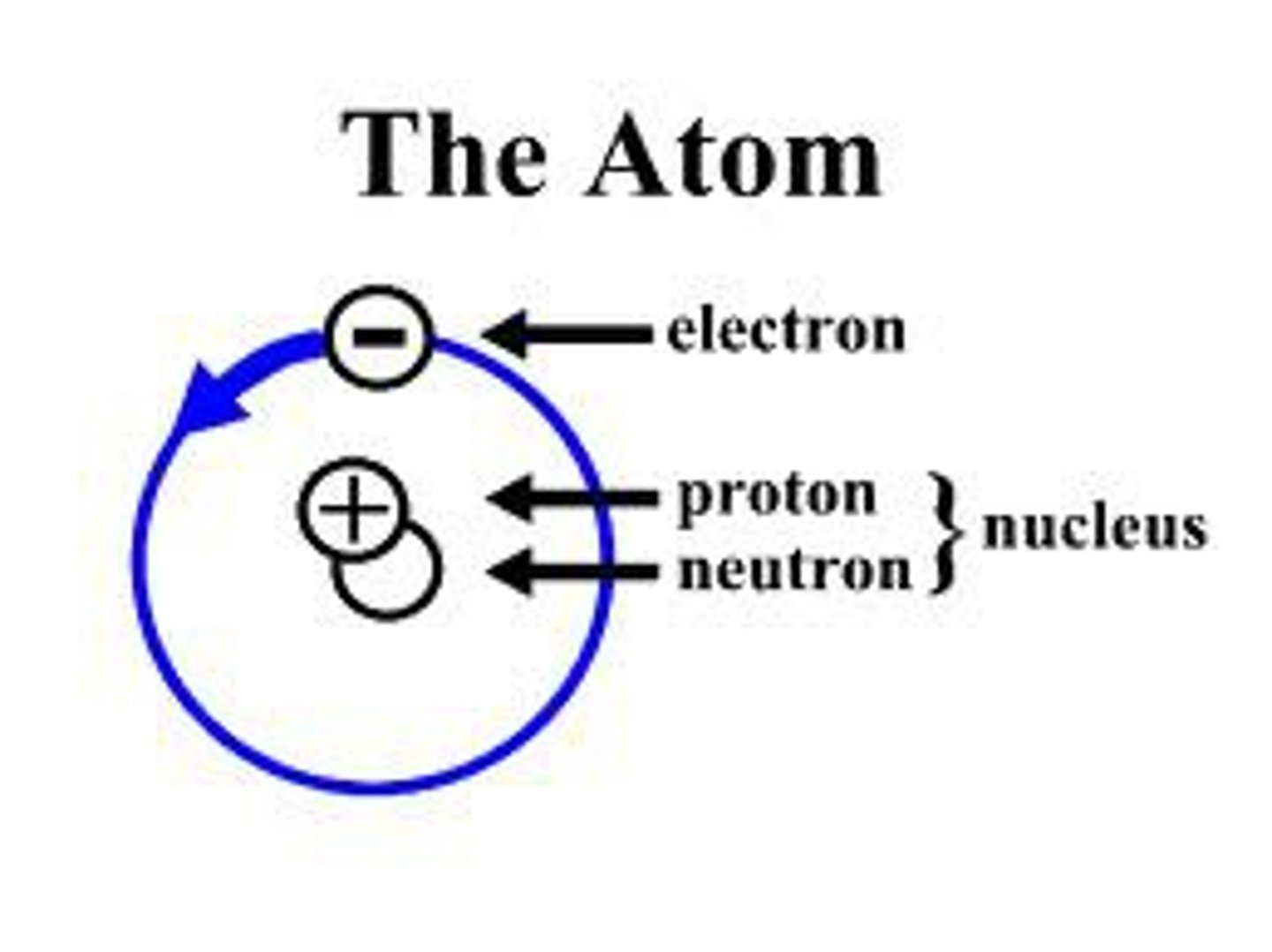
atom
Smallest particle of matter. Smallest particle of an element
molecule
2 or more atoms held together by chemical bonds. Atoms can be of the same kind of element as seen here (nitrogen gas) or can be from different kinds of elements (like H2O).
Jello
Heterogeneous Mixture

Salt Water
Homogeneous Mixture

Brass
Homogeneous Mixture

Iron
Element

Oxygen
Element
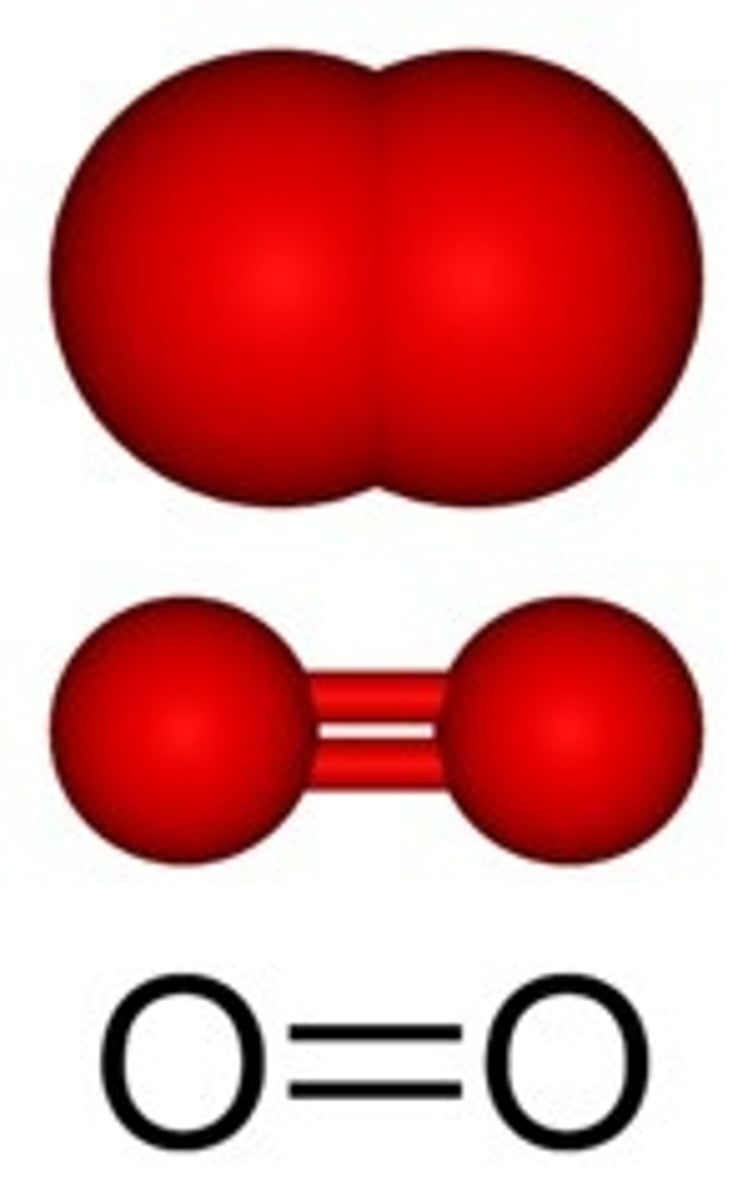
Carbon Dioxide
Compound
Fog
Heterogeneous Mixture

Silicon
Element

Milk
Heterogeneous Mixture

Soda
Homogeneous Mixture
