2.2B VSEPR Theory
1/9
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
What are covalent bonds characterized by?
Every covalent bond is characterized by two values:
Bond length: the distance between bonded nuclei
Bond strength: the amount of energy needed to break the bond. It is also called bond enthalpy.
In general...
Shorter bonds are stronger bonds
In general, shorter bonds are ________ bonds
stronger
Tell me about Multiple Bonds in molecules
Atoms can share more than one pair of electrons to form multiple bonds.
A double bond forms when two pairs of electrons (4e- in total) are shared
A triple bond forms when three pairs of electrons (6e- in total) are shared
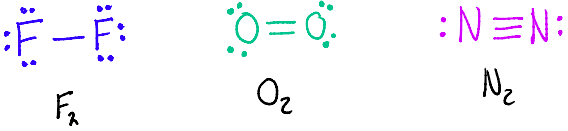 Multiple bonds have a greater number of shared electrons and so have a greater force of electrostatic attraction between the bonded nuclei.
Multiple bonds have a greater number of shared electrons and so have a greater force of electrostatic attraction between the bonded nuclei.
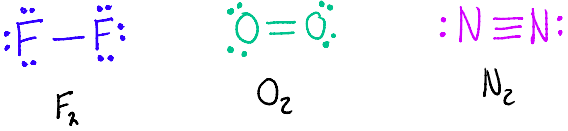
What is the relationship between Bond Length and each quantity of bond?
Shortest: Triple Bonds < Double Bonds < Single Bonds :Longest
What is the relationship between Bond Strength/Enthalpy and each quantity of bond?
Lowest: Single Bonds < Double Bonds < Triple Bond :Highest
What does VSEPR stand for? What does it state?
electrons in covalent bonds and in lone pairs repel one another
Molecules have 3D shapes that place electron pairs on the central atom as far apart as possible.


What are the shapes of molecules determined by?
The number of atoms bound to the central atom
The number of lone pairs of electrons on the central atom
Electron domains can be what types of bonds?
single, double, triple bonding electron pairs or non-bonding pairs of electrons
What determines the geometric arrangements of electron domains?
the total number of electron domains
Why do lone pairs of electrons cause slightly more repulsion than bonding pairs?
Lone pairs of electrons have a higher concentration of charge