2.2B VSEPR Theory
Covalent Bonds
Every covalent bond is characterized by two values:
Bond length: the distance between bonded nuclei
Bond strength: the amount of energy needed to break the bond. It is also called bond enthalpy.
In general...
Shorter bonds are stronger bonds
Multiple Bonds
Atoms can share more than one pair of electrons to form multiple bonds.
A double bond forms when two pairs of electrons (4e- in total) are shared
A triple bond forms when three pairs of electrons (6e- in total) are shared
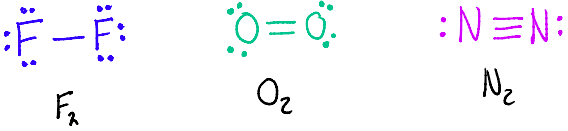 Multiple bonds have a greater number of shared electrons and so have a greater force of electrostatic attraction between the bonded nuclei.
Multiple bonds have a greater number of shared electrons and so have a greater force of electrostatic attraction between the bonded nuclei.
Bond Length
Shortest: Triple Bonds < Double Bonds < Single Bonds :Longest
Bond Enthalpy
Lowest: Single Bonds < Double Bonds < Triple Bond :Highest
Molecular Shapes: Valence Shell Electron Pair Repulsion Theory (VSEPR) Theory
electrons in covalent bonds and in lone pairs repel one another
Molecules have 3D shapes that place electron pairs on the central atom as far apart as possible.

Shapes of molecules are determined by...
1. The number of atoms bound to the central atom
2. The number oflone pairs of electrons on the central atom
Electron domains can be single, double, or triple bonding electron pairs or non-bonding pairs of electrons
Total number of electron domains determine the geometric arrangements of electron domains
Lone pairs of electrons have a higher concentration of charge and so cause slightly more repulsion than bonding pairs