Module 1 - Practical Techniques
0.0(0)
Card Sorting
1/5
Last updated 9:11 AM on 3/31/25
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
6 Terms
1
New cards
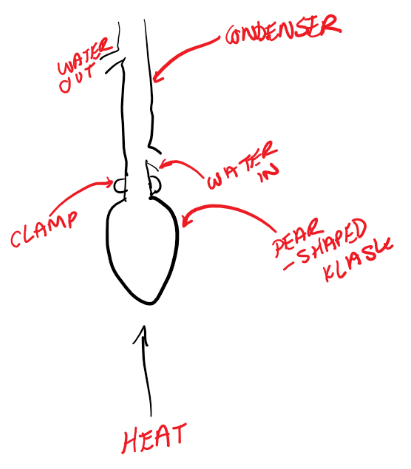
[1.2.2] What is HEATING UNDER REFLUX?
A procedure used to prepare an organic liquid without boiling off the solvent, reactants or products.
Anti-bumping granules are added to ensure that the contents will boil smoothly.
Anti-bumping granules are added to ensure that the contents will boil smoothly.
2
New cards
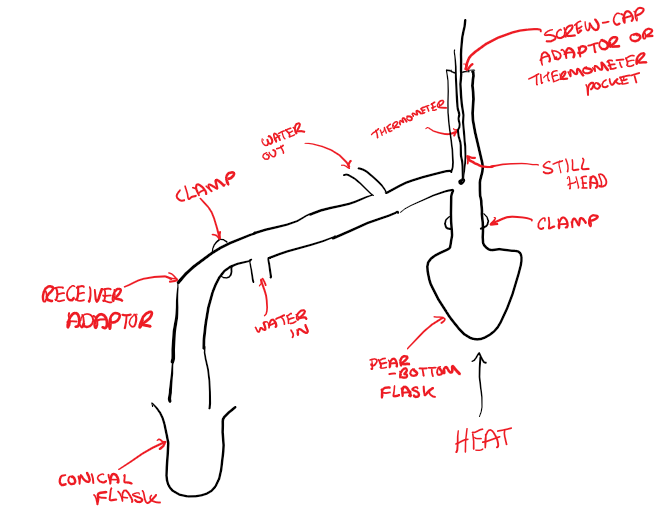
[1.2.2] What is DISTILLATION?
Distillation is a method used to separate a pure liquid from its impurities.
A conical flask is used to collect the distillate so that the distillation apparatus is not completely airtight.
The different liquids in the mixture have different boiling points, meaning that the liquids with the lowest boiling points move first into the condenser.
A conical flask is used to collect the distillate so that the distillation apparatus is not completely airtight.
The different liquids in the mixture have different boiling points, meaning that the liquids with the lowest boiling points move first into the condenser.
3
New cards
[1.2.2] How do we PURIFY ORGANIC PRODUCTS?
Once the organic layer has been identified, the two layers are separated via a separating funnel.
1) Ensure that the tap of the separating funnel is closed.
2) Pour the mixture of liquids into the separating funnel, place a stopper in the top of the funnel and invert it to mix the contents.
3) Allow the layers to settle.
4) Add some water to see which layer increases in volume - this is the aqueous layer.
5) Place a conical flask beneath the separating funnel, remove the stopper and open the tap until the whole of the lower layer has left the funnel.
6) Place a second conical flask beneath the separating funnel to collect the other layer.
7) There will be a conical flask containing the organic layer and another containing the aqueous layer.
1) Ensure that the tap of the separating funnel is closed.
2) Pour the mixture of liquids into the separating funnel, place a stopper in the top of the funnel and invert it to mix the contents.
3) Allow the layers to settle.
4) Add some water to see which layer increases in volume - this is the aqueous layer.
5) Place a conical flask beneath the separating funnel, remove the stopper and open the tap until the whole of the lower layer has left the funnel.
6) Place a second conical flask beneath the separating funnel to collect the other layer.
7) There will be a conical flask containing the organic layer and another containing the aqueous layer.
4
New cards
[1.2.2] How do we DRY ORGANIC PRODUCTS?
1. Add the organic liquid to a conical flask.
2. Using a spatula, add some of the drying agent to the liquid and gently swirl the contents to mix together.
3. Place a stopper on the flask to prevent the product from evaporating away and leave for 10 minutes.
4. If the solid has completely stuck together in a lump, there is still some water present. Add more drying agent until some solid is dispersed into the solution as a fine powder.
5. Decant the liquid from the solid into another flask. If the liquid is dry it should be clear.
2. Using a spatula, add some of the drying agent to the liquid and gently swirl the contents to mix together.
3. Place a stopper on the flask to prevent the product from evaporating away and leave for 10 minutes.
4. If the solid has completely stuck together in a lump, there is still some water present. Add more drying agent until some solid is dispersed into the solution as a fine powder.
5. Decant the liquid from the solid into another flask. If the liquid is dry it should be clear.
5
New cards
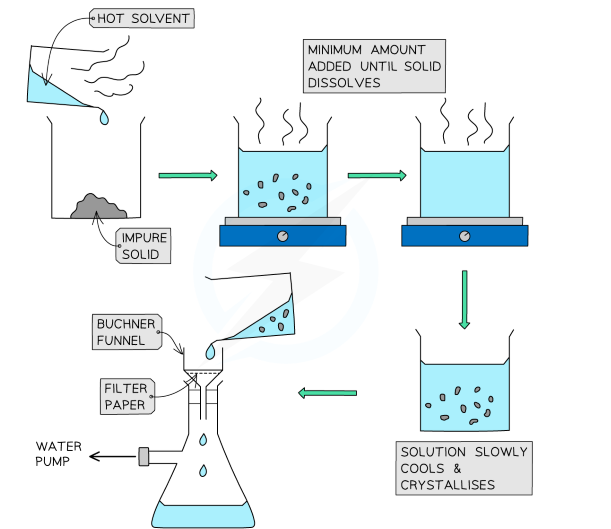
[1.2.2] What is RECRYSTALLISATION?
The principle of recrystallisation is that a hot solvent is used to dissolve both the organic solid and the impurities. As the solution cools, the solid crystallises out and leaves behind any impurities in the solution.
6
New cards
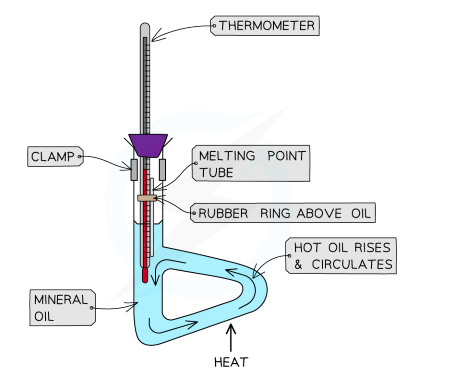
[1.2.2] How can we DETERMINE THE MELTING POINT of a substance?
The melting point of a solid is indicative of its purity and identity.
--> A melting point can be matched to a known substance as a means of identification or confirmation of a desired product.
--> The proximity of a melting point to the actual data book value can express purity. Impurities tend to lower the melting point of a solid.
The melting point range also reveals the degree of purity.
--> Pure substances have sharp, well defined melting points.
--> Impure substances have a broad range of melting points.
We can determine the melting point of a substance, e.g. benzoic acid via a thiele tube:
--> A melting point can be matched to a known substance as a means of identification or confirmation of a desired product.
--> The proximity of a melting point to the actual data book value can express purity. Impurities tend to lower the melting point of a solid.
The melting point range also reveals the degree of purity.
--> Pure substances have sharp, well defined melting points.
--> Impure substances have a broad range of melting points.
We can determine the melting point of a substance, e.g. benzoic acid via a thiele tube: