CHEM 1030 Chapter E: Units and Measurements
1/48
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
49 Terms
Qualitative observations
Descriptive in nature
Quantitative observations
Numerical values obtained from different measuring devices
Numerical value
Reflects the precision of the instrument or piece of glassware used to make the measurement
Systematic/Determinate Error
Error is in the same direction
Higher or lower than should be
Random/Indeterminate Error
Equal probability of measure being lower or higher than should be
Difficult to correct or find source
Length
meter (m)
Mass
kilogram (k)
Time
seconds (s)
Temperature
kelvin (k)
Amount of substance
mole (mol) 6.02 × 1023 units
Electric Current
ampere (A)
Luminous Intensity
candela (cd)
tera (T)
1012
giga (G)
109
mega (M)
106
kilo (K)
103
hecto (h)
102
deka (da)
101
deci (d)
10-1
centi (c)
10-2
milli (m)
10-3
micro (μ)
10-6
nano (n)
10-9
pico (p)
10-12
What is the formula used to convert from Fahrenheit to Celsius?
°F = 9/5 (°C) + 32
What is the formula used to convert from Kelvin to Celsius?
K = °C + 273.15
Precision
Agreement or closeness of a series of measured values
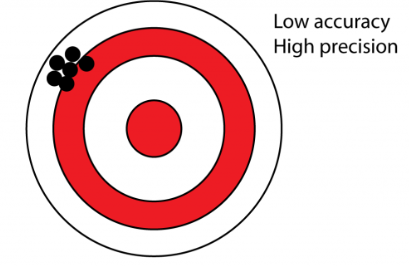
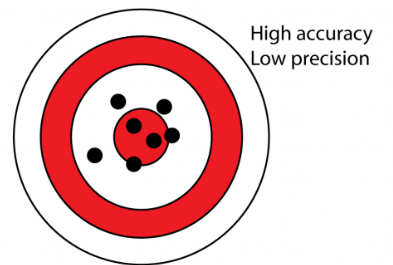
Accuracy
Agreement or closeness of measured value/s to actual known value
Reporting of significant figures in an answer depends on the _________________ of the MEASURED values given in the problem.
precision
Conversion factors, number used to change one set of units to another, are treated as ______ values.
exact
All nonzero values _________ significant
are
Zeroes between nonzero digits __________ significant
are
Leading zeroes to the left of a nonzero digit _________ significant
are not
Zeroes to the right after a nonzero digit _______ significant
are not
Zeroes to the right after a decimal place _____ significant
are
When multiplying or dividing, the answer should have the same number of sig fig as the number with the _____________.
least amount of sig figs
When adding or subtracting, the answer should have the same number of decimal places as the number with the __________.
least amount of decimal places
50,003 has ____________ sig figs
5
0.9785 has ____________ sig figs
4
Intensive properties are ____________ of size
independent
Extensive properties are ______________ on size
dependent
Density = _____/______
Mass/Volume
Density is an ____________ property
intensive
Energy is
the capacity to do work
Work
the action of a force applied across a distance
Finish the formula: KE = ___________
1/2m(v2)
True or False: m = kg
True
Energy units
kg*m2/s2
Energy is measured in _______________
Joules (J)