Ch. 9 to 11 - atomic spectroscopy
1/16
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
17 Terms
What are flame AAS and AES methods used for?
To analyze atomic metals and metal ions
What are the advantages of ICP methods (ICP-AES and ICP-MS)?
They’re able to analyze some nonmetals and have increased sensitivity compared to older flame methods
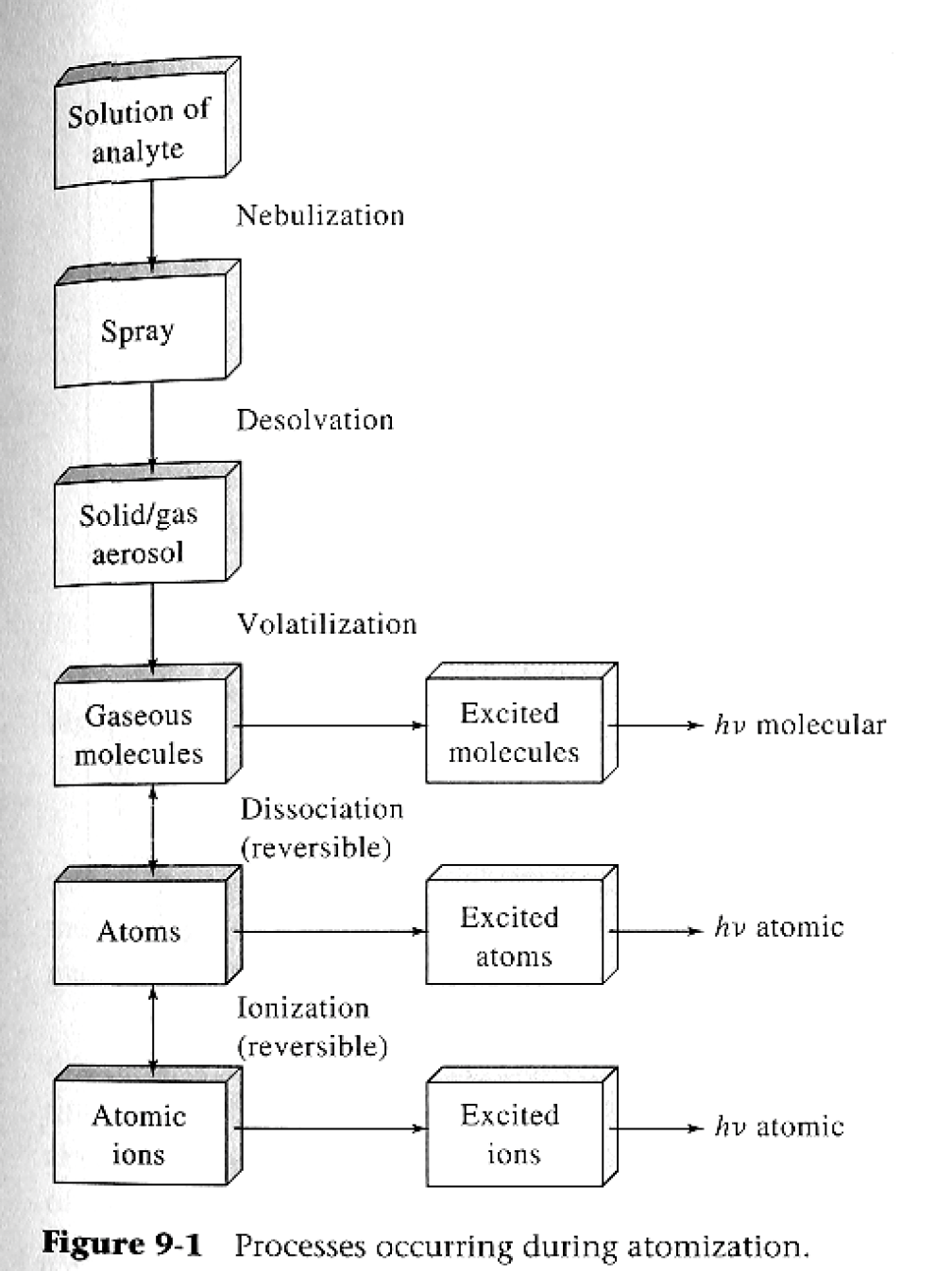
What are the steps of flame AAS?
Nebulization (solution of analyte to spray)
Desolvation (spray to aerosol)
Volatilization (aerosol to gas) (goes from gaseous to excited molecules)
Dissociation (reversible) (atoms to excited atoms)
Ionization (irreversible) (ions to excited ions)
What is Bohr’s theory?
Energy in each steady state can be determined by principle quantum number, where E = ( 2.18 × 10^(-18) J ) / n²
What is atomic spectra?
Refers to the spectrum of discrete line frequencies emitted by excited atoms
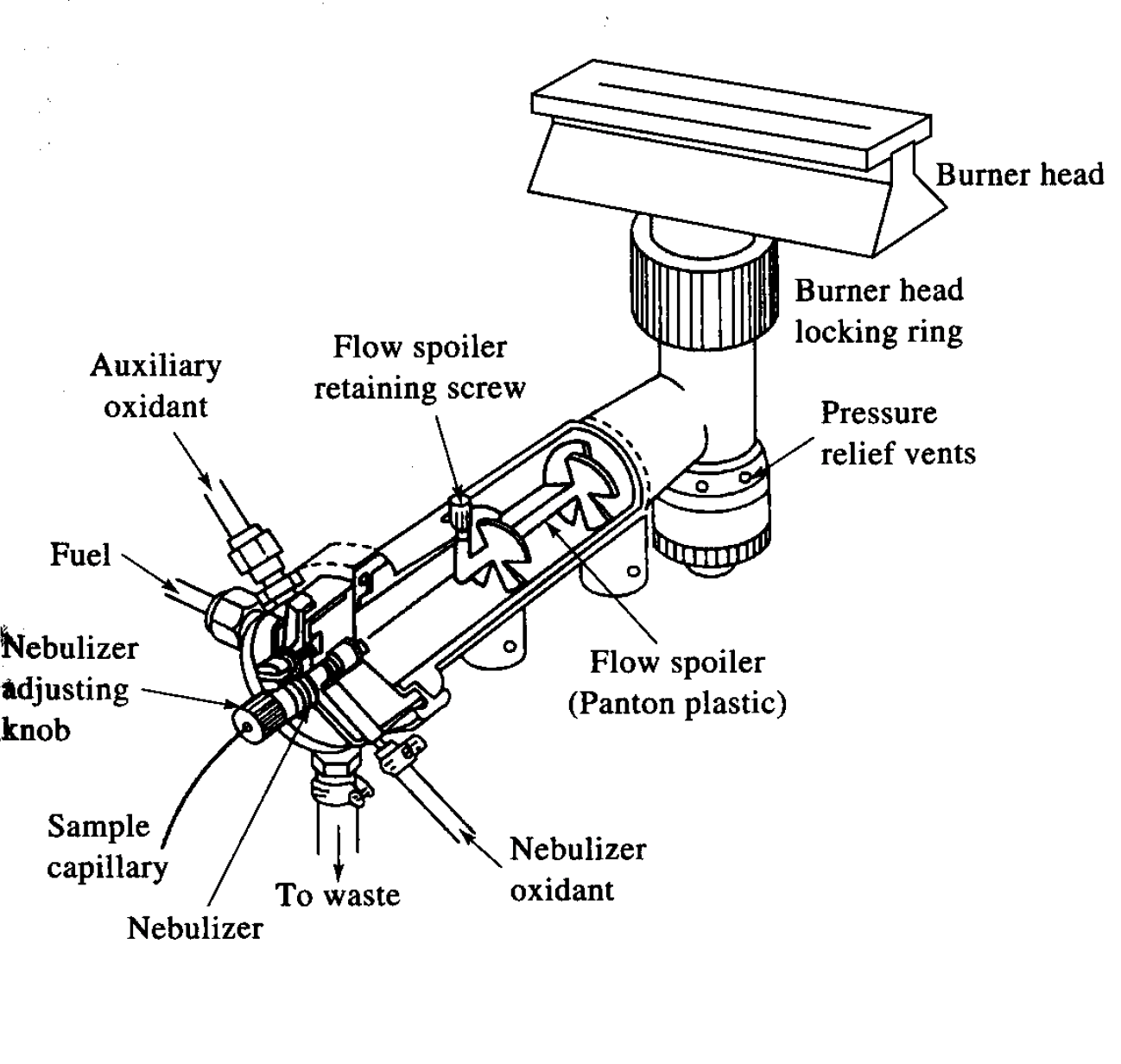
What is a laminar flow burner?
Flame or torch used during flame AAS/AES that oxidizes sample by flow of oxidant and fuel, providing relatively quiet flame and a long path length
What instrumentation is used for AAS/AES?
Radiation source (for AAS)
Sample holder (flame or torch)
Wavelength selector
Detector (PMT or MS)
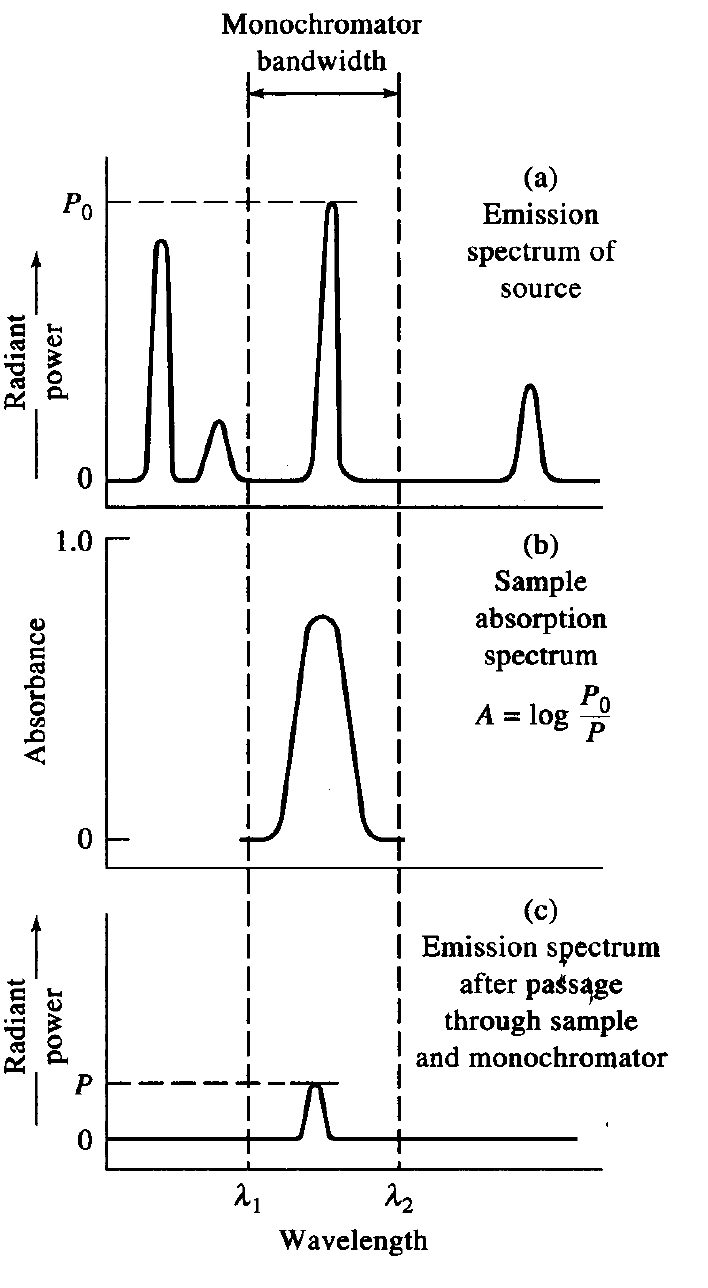
What conditions must be present for Beer’s law to be obeyed in flame AAS/AES?
Line width of source must be less than the absorption band line width
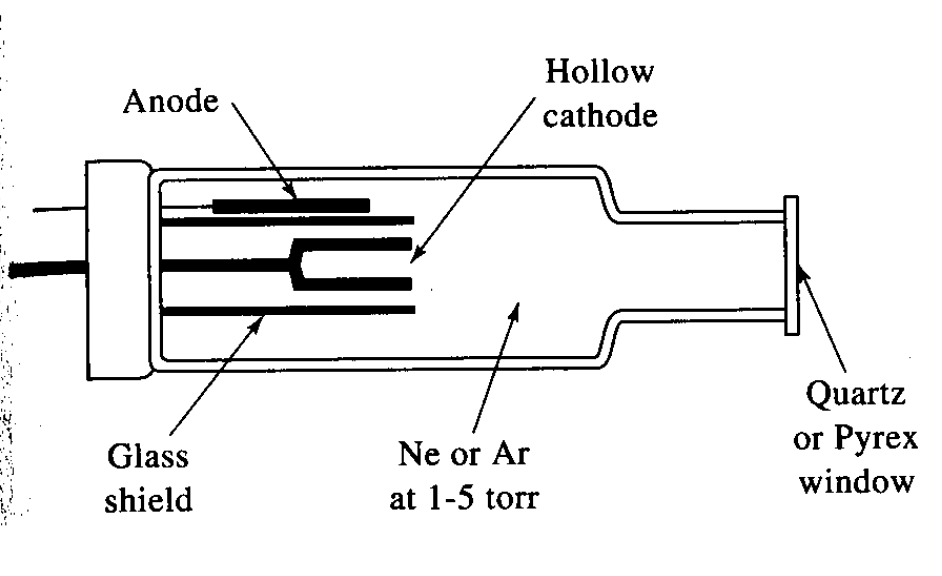
How are narrow band widths achieved for flame AAS/AES?
Using a line emitter source, most common example is a hollow cathode lamp containing a tungsten anode in a glass tube with neon or argon
What is spluttering?
Refers to when gaseous cations acquire enough kinetic energy to dislodge some of the metal atoms from the cathode surface and produce an atomic cloud. Some of these atoms emit photons with characteristic frequencies which provide emission spectra
How can one differentiate between absorption and emission radiation that occur simultaneously during flame AAS/AES?
Through use of either of a chopper or a pulsed power supply
What is ICP spectroscopy?
Uses plasma torch as its higher temperature limits the amount of chemical and spectral interference and also permits determination of some nonmetals (Cl, Br, S)
What is plasma?
Electrically conducting gaseous mixture of equal amounts of cations and electrons to maintain conductance. Only a small amount of plasma is sampled for use in ICP, current carrying ionized argon stream has the same temperature as the sun
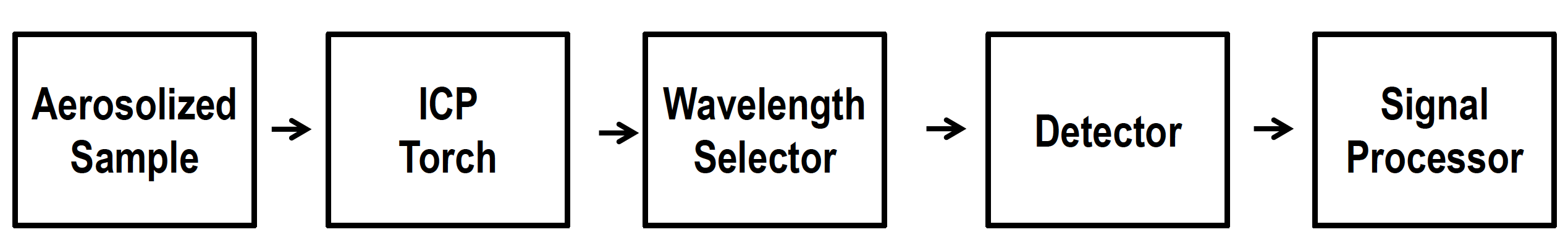
What is the general construction of ICP methods?
Aerosolized sample
ICP torch
Wavelength selector
Detector
Signal processor
What range do emission spectrometers display?
The entire UV-vis spectrum, 170nm to 800nm. Few extend into the UV, 150nm, where they can analyze for P, S, and C
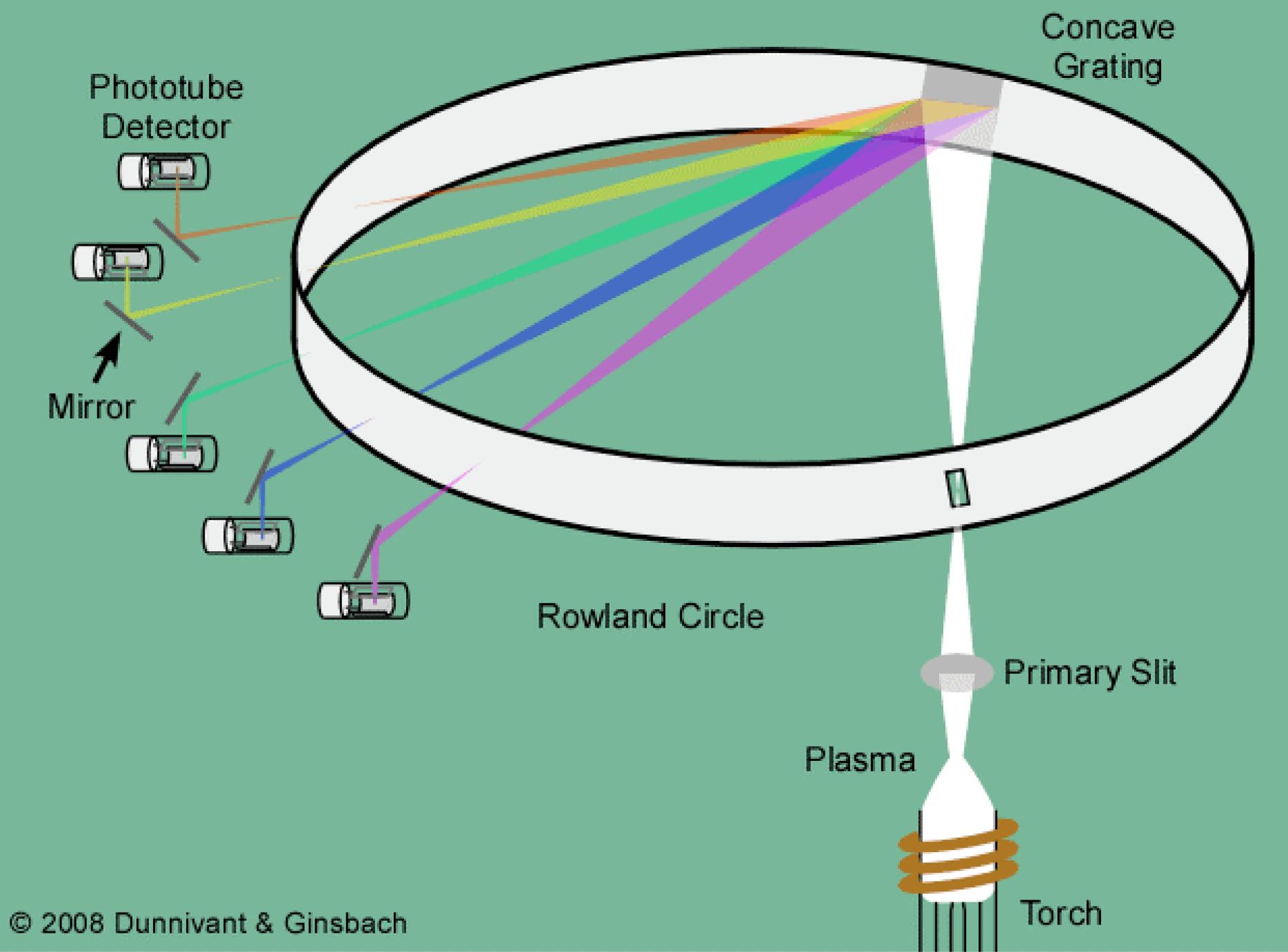
What are the 3 types of emission monochromators?
Sequential type: sample from one wavelength to the next
Multichannel AES: sample all at once, can rotate grating to detect specific elements
Fourier transform
What are the benefits of ICP-MS?
Detects all the elements at once, and has a very low limit of detection ranging from 0.1 to 10 ppb. This is nearly 1000 times more sensitive than atomic absorption/emission