Chem IB Exam Structure 1: Models of Particulate Nature of Substances
1/47
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
48 Terms
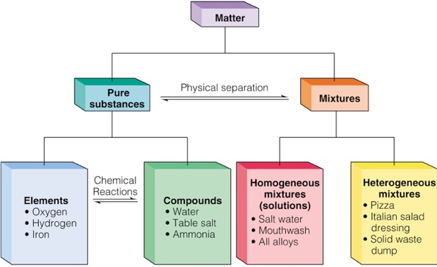
Define Chemical Element
It is Pure Substance made up of only ONE type of atom
Define atom
Smallest particle of an element to show the characteristic properties of that element. Example: Au (gold)
Define Compound
Chemical combinations of different elements
They contain FIXED proportions/ratios of elements, held together by chemical bonds
Ex: NaCl
Define mixture
Composed of Two or MORE substances in which no chemical combination had occurred
It does not have a fixed composition.
Define Homogenous Mixture
Uniform composition and properties. It has the same properties even if sample is taken from anywhere. Ex: saltwater
Define Heterogenous Mixtures
Non-uniform composition and properties are NOT same throughout. Ex: salad
List methods of separating compounds
Filtration: Solid particles in a liquid are removed through the use of a filter that allows only fluid to pass
Recrystallization: A substance is separated by dissolving the mixture in a solvent and then crystallizing desired substance from solution
Distillation: Components of a liquid mixture are separated through selective boiling and condensation
Paper Chromatography: Separates components of a mixture based on absorption to a solid phase.
Understand
Define Temperature
Measure of average kinetic energy of particles of a substance
Fluids
Liquids and gases are fluids which refers to their ability to flow
Define sublimation and deposition
Sublimation: Direct inter-conversion from solid to gas without the liquid phase
Deposition: Change from gas directly to solid. e.g. frosting
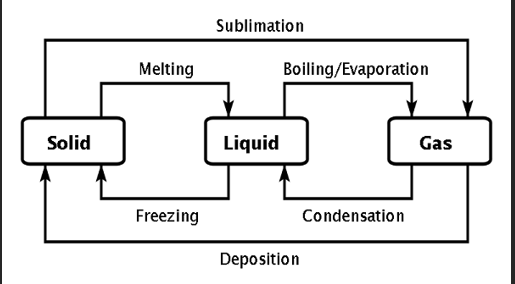
Distinguish between Vaporization and Evaporation
Vaporization: State change of matter from liquid to gas and can be accomplished through evaporation or boiling at FIXED temperature
Evaporation: State change that occurs only at surface of a liquid and at a temperature below the boiling point eg. clothes drying outside
Boiling Point
When all molecules in a liquid have enough kinetic energy to change into a gas
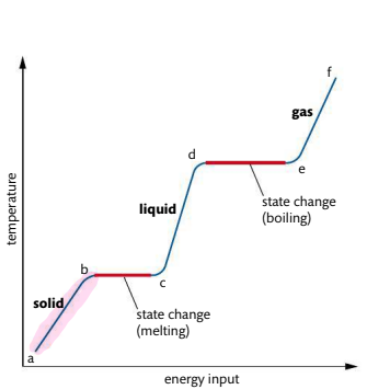
What occurs in heating curve from a-b
The solid is heated and as vibrational energy of particles increases so does the temperature
Note, during a state change there will be no increase or decrease in temperature
Adding temperature only increases the kinetic energy of the molecules, which will eventually break the bonds, then the molecules will change state
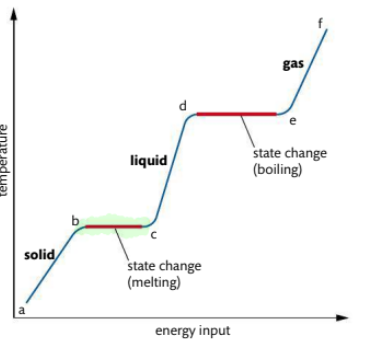
What occurs in heating curve from b-c
It is the melting point. Vibrations are energetic enough for particles to move away from fixed positions and form a liquid Temperature remains constant at this point
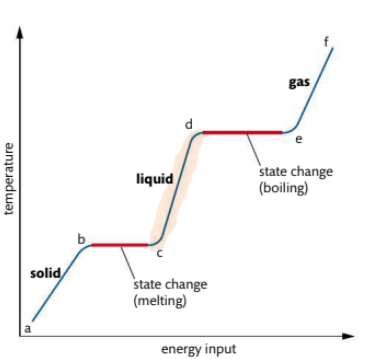
What occurs in heating curve from c-d
Liquid is heated, particles gain energy and temp increases
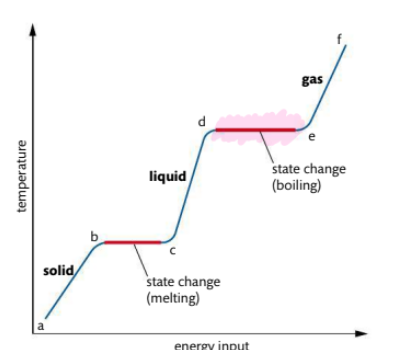
What occurs in heating curve from d-e3
d-e: boiling point, sufficient energy to break all interparticle forces and form a gas.
• Requires more energy than melting
• Temperature remains constant
• Bubbles of gas visible throughout
volume of liquid
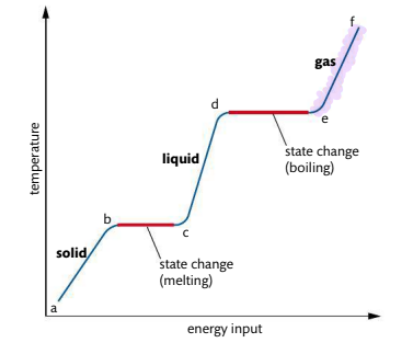
What occurs in heating curve from e-f
gas is heated under pressure, kinetic energy of particles continues to rise, temperature also rises
Conversion of Kelvin to Celsius
Temperature (K) = Temperature (°C) + 273.15
Distinguish between atomic # and Mass #
Atomic #: Number of protons in an atom
Mass #: Number of protons + the number of neutrons in an atom
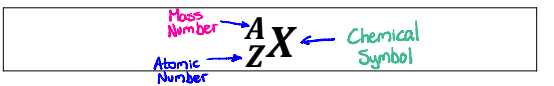
Ions?
When atoms gain or lose electrons
Cations vs. anions
Cations are atoms that are positively-charged ion because they lose electrons
Anions are atoms that are negatively-charged ion because they gain electrons
Define isotopes
They are atoms of an element that have the same # of protons but different # of neutrons
Have very similar chemical characteristics because # of protons and electrons are the same
Have different atomic mass because of different # of neutrons which leads to different physical properties
Atomic radius vs. Ionic radius
Atomic Radius: The distance of the nucleus from its outermost electron
Ionic Radius: The radius of the atom that has gained or lost electron(s) and become negatively or positively charged
Relative Average Mass Formula

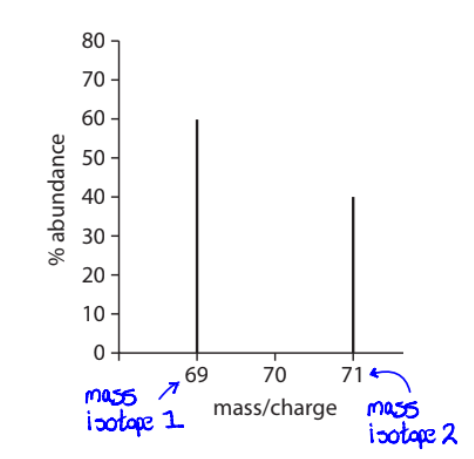
Define Mass Spectra and be able to interpret it
Mass spectrometer is an instrument used to measure mass of individual atoms.
Results are presented as a mass spectrum, where % abundance is plotted against mass/charge ratio of different ions
Relative average Mass can be calculated from this data
Define Electromagnetic radiation
A from of energy that propagates through space at the speed of light as electromagnetic waves AKA photons
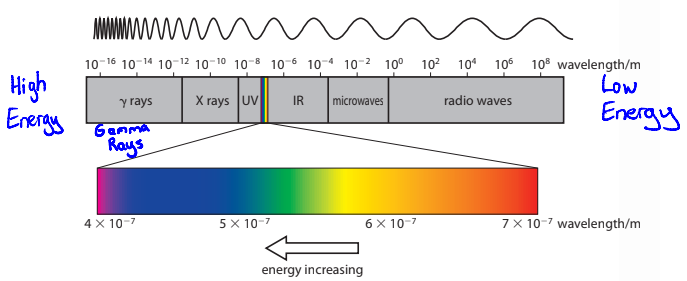
Define Frequency
Number of waves that pass a point in 1 second
Define Emission Spectra
produced by atoms emitting photons when electrons in excited states return to lower energy levels.
Explain Convergence of lines on the hydrogen spectrum
As energy levels increases, they get closer together and converge at high energy.
Distinguish between a continuous and a line spectrum.
When excited electrons ‘fall’ from a higher to a lower energy state, photons with a discrete amount of energy are emitted. The emission spectrum of atoms is a line spectrum: only light of a particular colour (discrete energy) is emitted.
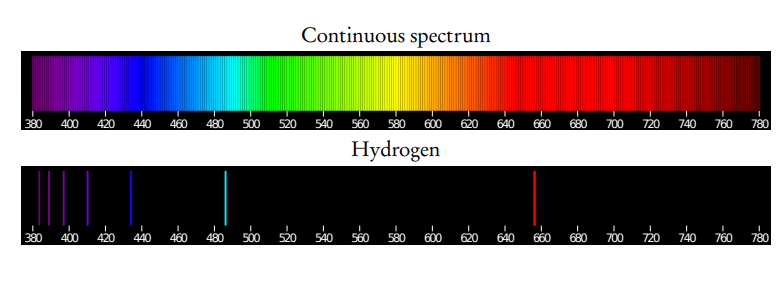
Describe the emission spectrum of the hydrogen atom, including the relationships between the lines and energy transitions to the first, second and third energy levels.
Electrons that ‘fall’ to the groundstate (n = 1) emit photons with the greatest amount of energy (UV radiation). The length of the arrows is proportional to the amount of energy. Electrons that ‘fall’ to n = 2 emit visible light and to n = 3 emit infrared radiation.
line spectra converge at higher energy because the energy levels inside the atom are CLOSER together at higher energy.
When an electron reaches the highest energy (n=infinity), the electron leaves the atom and results in an ion
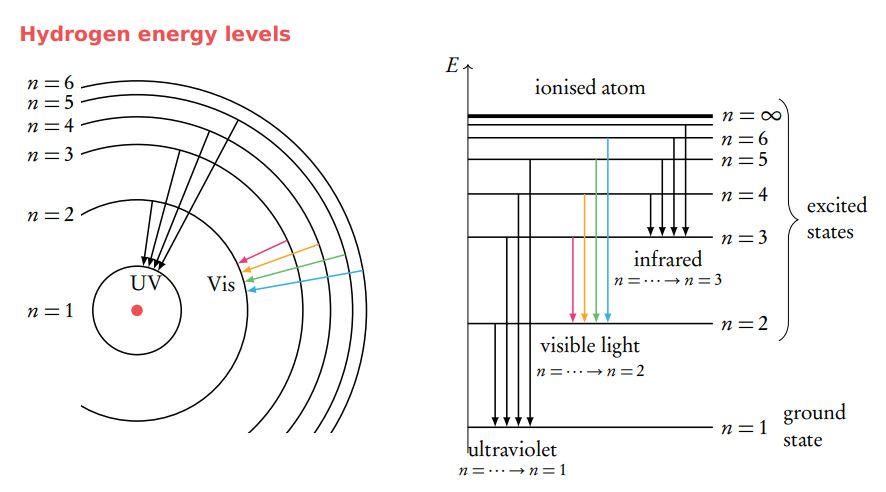
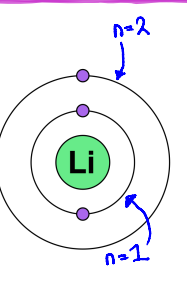
Energy level
It is how far an electron is away from the nucleus and denoted as ‘n”
The maximum number of electrons that can be found in each
energy level can be found using 2n²
Sub-level
Shape of region in which electron can be found. Sublevels contain fixed # of orbitals
Orbitals
Region of space where there is a High probability of finding an electron
o s: 1 orbital, 2 electrons
o p: 3 orbitals, 6 electrons
o d: 5 orbitals: 10 electrons
o f: 7 orbitals, 14 electrons
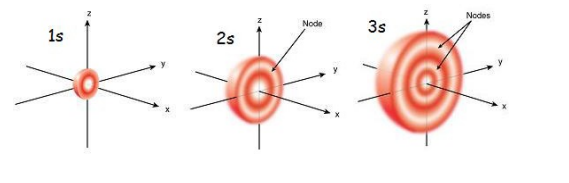
What is the s sub-level
Every s subshell consist of one spherical orbital, which is further away from the nucleus the higher the shell number.

What is a Node
A region with ZERO probability of finding an electron
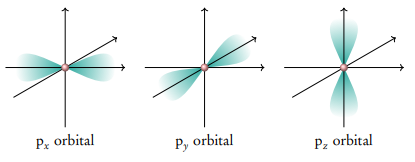
What is the p sub-level
The second energy level, n=2, can have s AND p sub-levels
The p subshell always contains 3 orbitals which are aligned along the x,y, z-axis. Since the second shell (n = 2) contains the 2s 2p subshells which can host 2 and 6 electrons respectively, the maximum total number of electrons in the second shell is 8.
Apply the Aufbau principle, Hund’s rule and the Pauli exclusion principle to deduce electron
configurations for atoms and ions up to Z = 36.
Electrons fill into the lowest energy orbitals first. Each orbital can hold at most 2 electrons (Aufbau principle)
If two electrons occupy the same orbital, they must have a different spin (Pauli Exclusion Principle)
If multiple orbitals have the same energy, they are filled singly first, then doubly. (Hund’s Rule)
Writing electron confi/arrangement. Also what are lectron config for Chromium and Copper
The 4s orbital is filled first before 3d, but is removed first before 3
The electronic configurations of the transition elements copper and chromium do not follow the expected patterns
Chromium has the electron configuration: [Ar] 3d5 4s1
Copper has the electron configuration: [Ar] 3d10 4s1
![<ul><li><p><strong>The 4s orbital is filled first before 3d, but is removed first before 3</strong></p></li><li><p>The electronic configurations of the transition elements copper and chromium do not follow the expected patterns </p><ul><li><p>Chromium has the electron configuration: [Ar] 3d5 4s1 </p></li><li><p>Copper has the electron configuration: [Ar] 3d10 4s1</p></li></ul></li></ul><p></p>](https://knowt-user-attachments.s3.amazonaws.com/f1fceb3c-c099-4e68-8c6f-34dd998268e7.png)
What is the ground state
When an electron is with no addition of energy
What is the infinity level
The highest possible energy level that an electron can have and still be part of the H atom
Define First Ionization Energy
The MINIMUM amount of energy required to remove 1 mole of electrons from 1 mol of gaseous atoms.
X9G) → X+ + e-
What factors influence 1st ionization energy
Size of the nuclear charge
Distance of outer electrons from the nucleus
Shielding effect
How does the size of nuclear charge affect 1st ionization energy
As the # of proton increases, the nuclear charge increase
The larger the positive charge, the GREATER the attractive electrostatic force between the nucleus and electrons (More protons create a stronger electrostatic force, pulling electrons closer to the nucleus.)
SO, a larger amount of energy is needed to overcome these forces and remove an electron
As the proton number increases, ionization energy increases: First ionization energy increases across each period
How does the distance of outer electron affect 1st ionization energy
The electrostatic attraction decrease as distance increases so electrons in shells are more weakly attracted to the nucleus
The further the outer electron is form the nucleus, the LOWER the ionization energy is
Thus, ionization energies tend to decrease down a group of the periodic table
How does shielding effect affect 1st ionization energy
It occurs when the full inner shells of electrons PREVENT electrons in higher levels from the pull of the nucleus
The GREATER the shielding effect, the lower the electrostatic force between outer electrons and the nucleus
Thus, ionization energy is lower as the number of full electron shells between the outer electrons and the nucleus increases
Formula for effective nuclear charge
Zeff = Z - S
Z: atomic number(#of electrons/[protons)
S: shielding electrons