Chemistry Unit 1 (Term 1&2) Flashcards VCE Year 11
1/120
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
121 Terms
metallic bonding
A type of chemical bonding that occurs between metal atoms, characterised by a sea of delocalised electrons
ionic bonding
A type of chemical bonding that occurs between metal and nonmetal atoms, characterised by an attraction between oppositely charged ions
co-valent bonding
A type of chemical bonding that involves the sharing of electron pairs between atoms, typically between nonmetals.
cations
Positively charged ions formed when an atom loses one or more electrons.
anions
Negatively charged ions formed when an atom gains one or more electrons.
nuetrons
Subatomic particles with no electric charge (neutral), found in the nucleus of an atom, along with protons.
protons
Subatomic particles with a positive charge, found in the nucleus of an atom, along with neutrons
electrons
Subatomic particles with a negative charge, found in the electron cloud surrounding the nucleus of an atom.
non-metals
Elements that are generally poor conductors of heat and electricity, they often can gain electrons during chemical reactions.
metals
Elements that are good conductors of heat and electricity, they can often lose electrons during chemical reactions
element
A pure substance that consists of only one type of atom, distinguished by its atomic number.
atom
The smallest unit of an element, consisting of a nucleus surrounded by electrons.
nucleus
The central part of an atom, containing protons and neutrons, and is positively charged.
atomic number
The number of protons in the nucleus of an atom
relative atomic mass
The weighted average mass of an atom of an element
molecule
when two or more atoms bond by sharing electrons to form a stable unit (example: hydrogen + hydrogen = h2) - they can be the same or different element
compound
a substance formed when two or more different elements chemically bond together. (example NH3)
Isotopes
atoms of the same number of protons with different numbers of neutrons
isotopic notation
mass number (protons + neutrons)
——————————————— element symbol
atomic number (number of protons)
polyatomic ions
ions made up of two or more atoms that are covalently bonded, carrying a net charge.
subshells
The divisions of electron shells in an atom
orbitals
shells within the subshells
the equation for metal reacting with an acid
metal + acid ⇨ salt + hydrogen gas
the equation for metal reacting with water
metal + water ⇨ metal hydroxide + hydrogen gas
the equation of metal reacting with oxygen
metal + oxygen ⇨ metal oxide
periods
rows in the periodic table (horizontal)
groups
the columns in the periodic table (vertical)
noble gases
a group of chemical elements in the periodic table that are characterized by their low chemical reactivity.
electronic configuration
the distribution of electrons of an atom or molecule
valence electrons
the electrons located at the outermost shell of an atom
valence shell
outermost energy shell where valence electrons are found
reactivity
the tendency of an atom to loose or gain electrons
what are the exceptions to the electronic configuration rules
chromium and copper (when in shell order, add 1, minus 1) - more stable
what are the energy levels found within shells
s < p < d < f
how many orbitals and electrons does each subshell have
s subshell: 1 orbital and 2 electrons
p subshell: 3 orbital and 6 electrons
d subshell: 5 orbital and 10 electrons
f subshell: 7 orbital and 14 electrons
electronic configuration order
1s
2s 2p
3s 3p 3d
4s 4p 4d 4f
5s 5p 5d 5f
6s 6p 6d 6f
What must you do after finding the electronic configuration of an element?
Ensure that you have ordered it in shell order and that the valence shell sits last
What is the rule for finding the effective nuclear charge?
Zeff = Z - S, where Z is the atomic number/amount of protons and S is the number of inner shell electrons
effective nuclear charge/core charge
effective nuclear charge is the measure of attracive force felt by the valence shell electrons towards the nucleus
effective nuclear charge down a group
When moving down a group, the effective nuclear charge remains constant
effective nuclear charge across a period (left to right)
When moving across a period (left to right), the effective nuclear charge increases
effective nuclear charge: general trends (chart)
group: 1=+1, group: 2=+2, group: 13=+3, group: 14=+4, group: 15=+5, group: 16=+6, group: 17=+7, and group: 18=+8
electronegativity
the ability of the atom to attract “shared” electrons towards itself during a chemical bond.
electronegatively moving across a period (left to right)
moving accross a period (left to right), electronegativity increases
electronegativity moving down a group
As you move down a group, electronegativity decreases
atomic radius
the measure of the size of an atom, it’s typically the distance from the center of the nucleus to the outermost isolated electron
atomic radius going down a group
As you move down a group, the atomic radius increases
atomic radius going across a period (left to right)
As you move across a period from left to right on the periodic table, the atomic radius generally decreases.
First ionization energy
the energy required to remove the outermost electron from a neutral atom. It's a physical property that affects an atom's chemical behavior.
ionisation energy down a group
When you move down a group in the periodic table, the ionisation energy decreases.
ionisation across a period (left to right)
When moving across a period (left to right), ionisation energy increases.
what is the reactivity of metals
how readily a metal can participate in chemical reactions with other substances, essentially, how easily it can lose electrons to form positive ions
reactivity of metals down a group
As you move down a group, the reactivity of metals increases.
reactivity of metals across a period (left to right)
As you move across a period (left to right), the reactivity of metals decreases.
what is the reactivity of non-metals
how readily a non-metal can participate in chemical reactions with other substances, essentially, how easily it can gain electrons to form negative ions
reactivity of non-metals down a group
When moving down a group, the reactivity of non-metals decreases.
reactivity of non-metals across a period (left to right)
When moving across a period (left to right), the reactivity of non-metals increases.
What are critical elements?
elements that are in high demand but have limited supply. They are important to businesses and economies, and can include: copper, lithium, nickel etc
lewis dot diagram
bonding of electrons shown via dots
lewis structure
a bond shown between lines (2 electrons for each line/bond)
VSEPR acronym
valence, shell, electrons, protons, and repulsion
VSEPR meaning
The VSEPR model predicts 3-D molecular geometry based on electron pair repulsion.
pyramdial shape

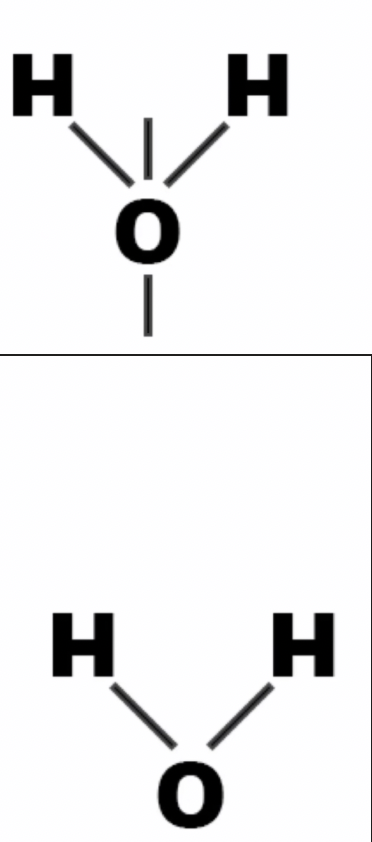
v shape or bent shape
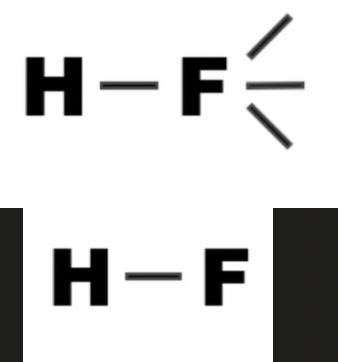
linear shape
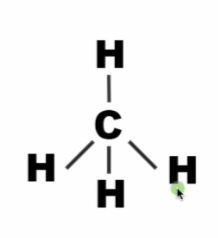
tetrahedral shape
intra-molecular bonds
hold atoms together within a molecule, they are very strong bonds and don’t get broken expect when in a chemical reaction
inter-molecular bonds
hold molecules together, they are generally weaker bonds and often break when heat is applied
polar molecules
a molecule in which one end of the molecule is slightly positive, while the other end is slightly negative
non-polar molecules
molecules that do not have any split electrical charges or split partial charges
how to know if a molecule is polar?
if the electronegativity difference/net dipole is larger than 0.4 and it is unsymmetrical then it is, if not then it isnt polar
states
liquid (l), gas (g) ,aqueous (aq), solid (s)
do metals have high or low electronegativity levels?
low
do non-metals have high or low electronegativity levels?
high
when drawing inter-molecular bonds, ensure…
that your bonds are between delta positive and negatitve atoms, depicitng the strike in polarity
excited state in an atom
When an electron gains energy, it may temporarily occupy a higher state—for example, jumping to 3d instead of 4s.
dispersion forces
the weakest intermolecular forces between atoms and molecules
dipole-dipole attraction
between polar molecules, caused by attraction between their positive and negative ends; second strongest intermolecular force.
hydrogen bonding
only occurs when hydrogen is bonding with N, O or F (about 10 times stronger than the dipole-dipole attraction)
lattice
The three-dimensional arrangement of atoms, ions, or molecules
how many covalent bonds are formed by each carbon atom in graphite and diamond
Diamond: four covalent bonds. Graphite: three covalent bonds
diamond
it is bonded in a 3 dimenshional covalent network lattice — it is a strong network lattice as it is an intramolecular bond
properties of diamond
high melting point
very hard
britlle
does not conduct electricity
high thermal conductivity
insoluable (unable to be dissolved)
graphite
a strcutured covalent layer lattice with layers of 2 dimensional carbon lattices held by weak dispersion forces
properties of graphite
high melting point
high thermal conductivity
soft, slippery feeling
less dense than diamonds
insoluable
the equation for acid reacting with a reactive metal
acid + reactive metal → ionic salt + hydrogen gas
the equation for water reacting with a reactive metal
water + reactive metal → metal hydroxide + hydrogen gas
list the uses of diamonds
jewelry, industrial applications, medicine, and electronics.
list the uses of graphite
pencils, lubricants, batteries, and nuclear reactors.
soluble (aq)
spectator ions
insoluable (s)
ppt
properties of metal
malleable: Flexible
ductile: stretched without breaking
high electrical conductivity
high melting and boiling point
lustre
solvent + solute
solution
solvent
a liquid in which a substance is dissolved
when drawing substances, hydrating means
you cover the whole entire atom in h2o or whatever the atom may be
solute
substance being dissolved
properties of ionic compounds
high melting and boiling points: Resistant to heat
brittle: breaks under stress
hardness: Resistance strength
varying electrical conductivity: Semiconducting behavior
why are ionic compounds brittle?
because of the strong electrostatic forces between their positive and negative ions due to the attraction of opposite charges
when drawing equations with h2o in it when depicitng the intermolecular bonds
make sure you arent adding h2o to the molecule but rather the h20 is above the arrow
metal + hydrogen gas
→ solid metal oxide (o2)