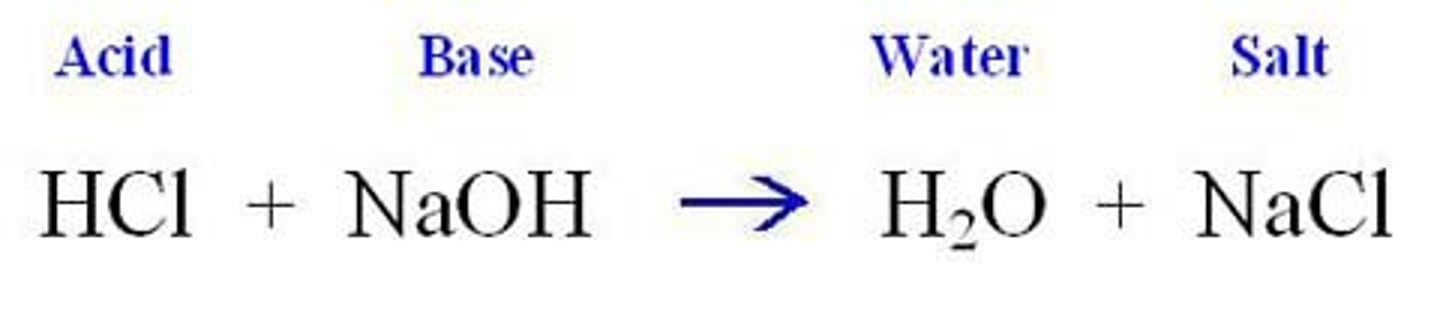SNC2DY-a Types of Reactions
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
32 Terms
synthesis
two reactants (elements or compounds) react to produce one new compound.
metal oxide + water=
metal hydroxide or base
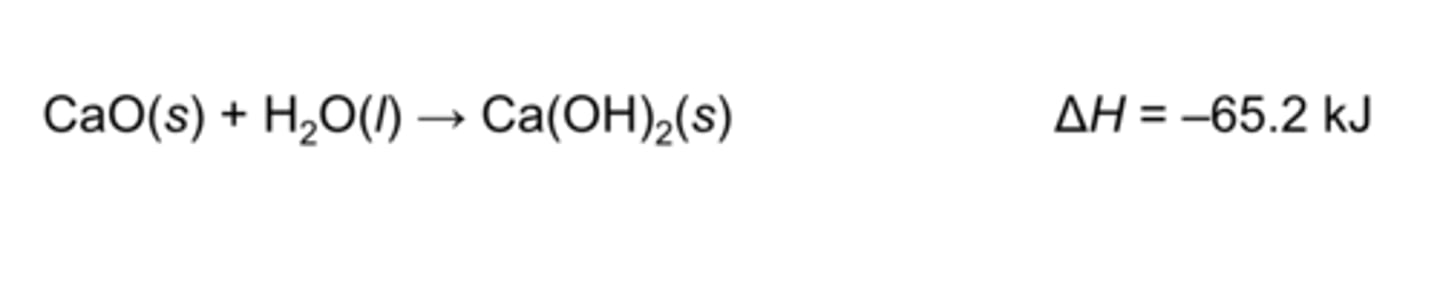
nonmetal oxide + water
acid
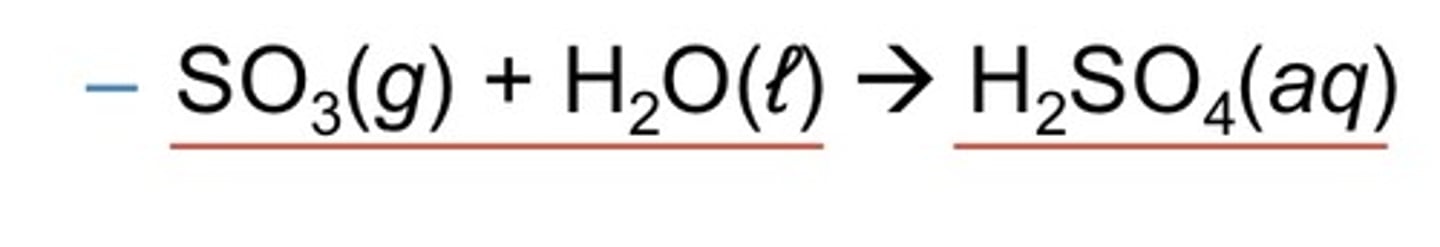
metal oxide + carbon dioxide
metal carbonate

metal halides + oxygen
metal halates
MgCl2(s) +3O2(g)---> Mg(ClO3)2(s)
decomposition
AB->A+B
complete combustion
hydrocarbon + oxygen --> carbon dioxide + water
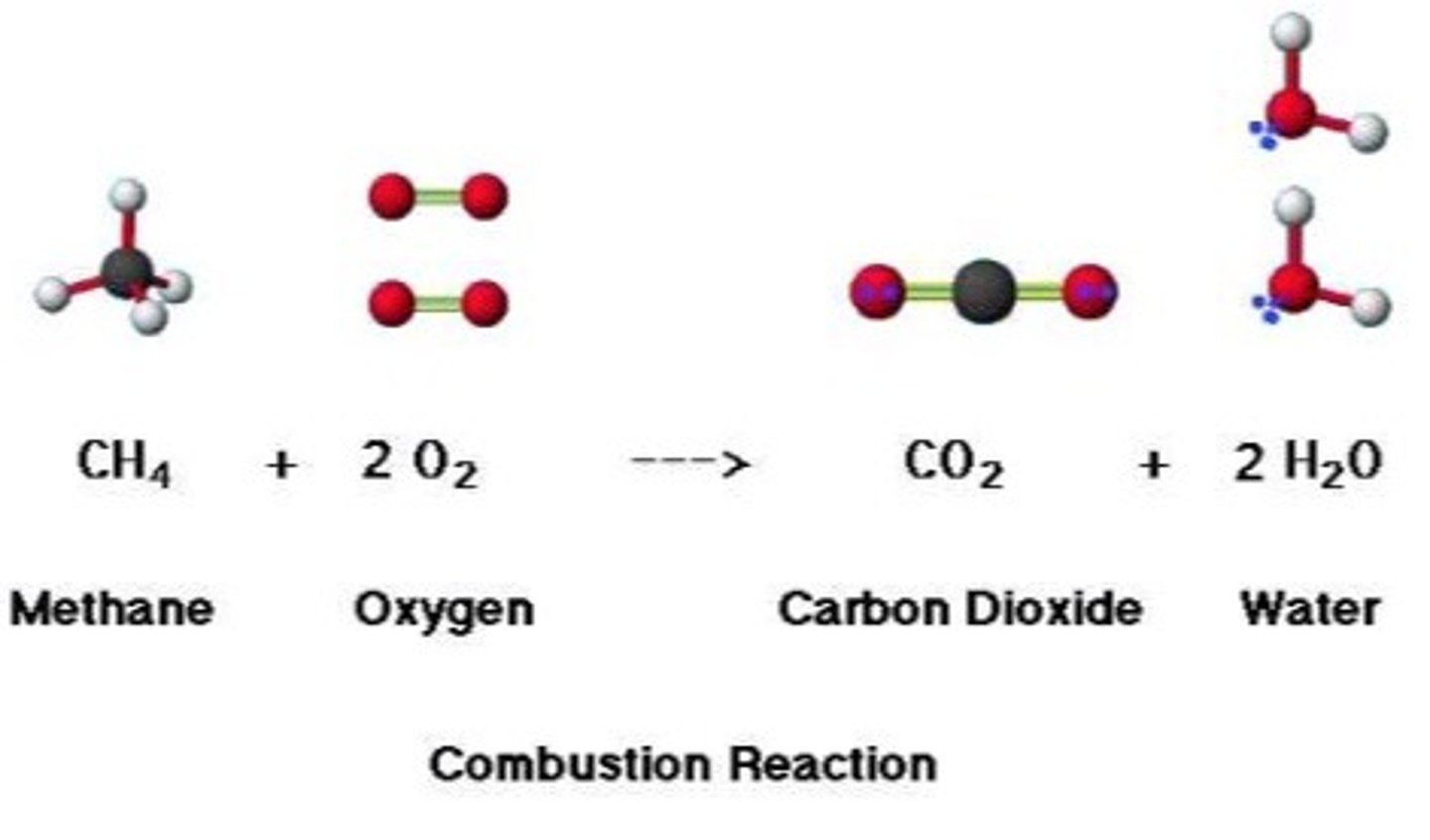
incomplete combustion
insufficent oxygen, produces: carbon dioxide, carbon monoxide, water and soot (solid carbon)

vehicles burn gasoline
incompletely (low engine temp)
CO binds w hemoglobin
metals + oxygen
metal oxide or peroxides

nonmetal + oxygen
nonmetal oxide
diatomic elements with states
HOFBrINCl
P4S8
all the solids: I2, P4, S8
liquid: Br2
law of conservation of mass
Matter is neither created nor destroyed
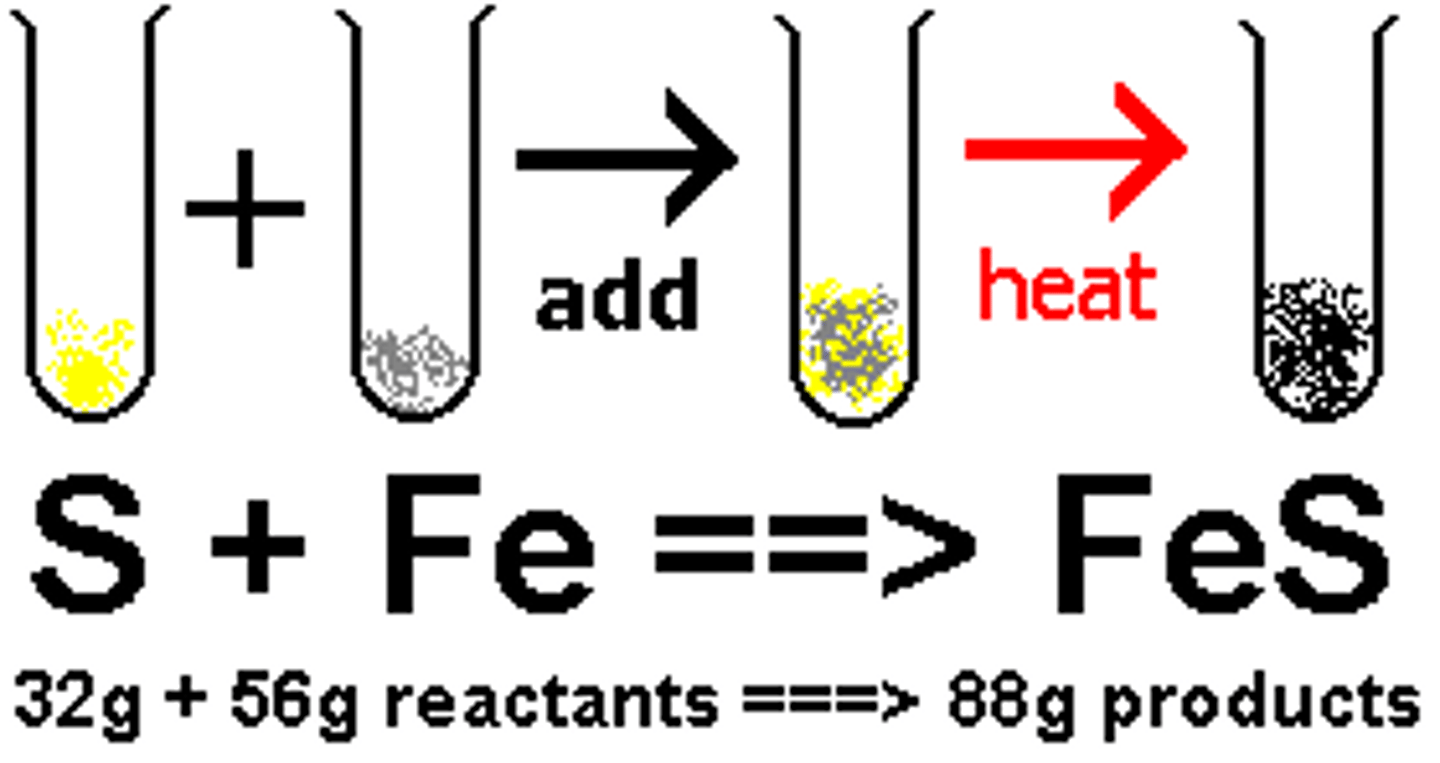
single-displacement reaction
chemical reaction in which one element replaces another element in a compound
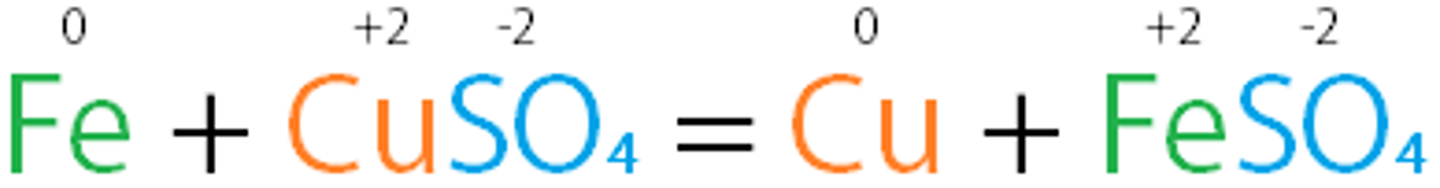
Metals from Li - Na
react w water to form metal hydroxide and hydrogen gas
Metals from Mg--->Cd
react w steam to form metal oxide n hydrogen gas
metals above hydrogen
react w acids to form ionic compound and hydrogen gas
NR meaning
non-reactive (cant dispace)
Metals from Li - Na w O2
forms metal peroxides and metal oxides
metals from Mg-Hg
reacts w oxygen to form metal oxide
what is corrosion
Corrosion is the destruction of materials by chemical reactions with substances in the environment.
metals that dont corrode
gold n platinum
where is corrosion beneficial
when reacting w oxygen aluminum oxide forms a protective layer coating the aluminum
and copper forms the green patina (weatherproof 75 years)
what is rust? diff between rust n corrosion
reddish brown flaky iron (III) oxide
rust doesnt stick to the metal unlike other metals
5 factors of rust
air, h20 electrolytes, acidity, mech.stress
more humid air=more rust
dry air=less rust
salt
speeds up rusting
protective coating for rust
cover metal w rust inhabiting paint, chrome or plastic
corrosion resistant materials
stainless steel (fe, ni, cr, c)
plastic
galvanizing
Coating steel with zinc to prevent corrosion forming ZnO sticking to steel n zinc
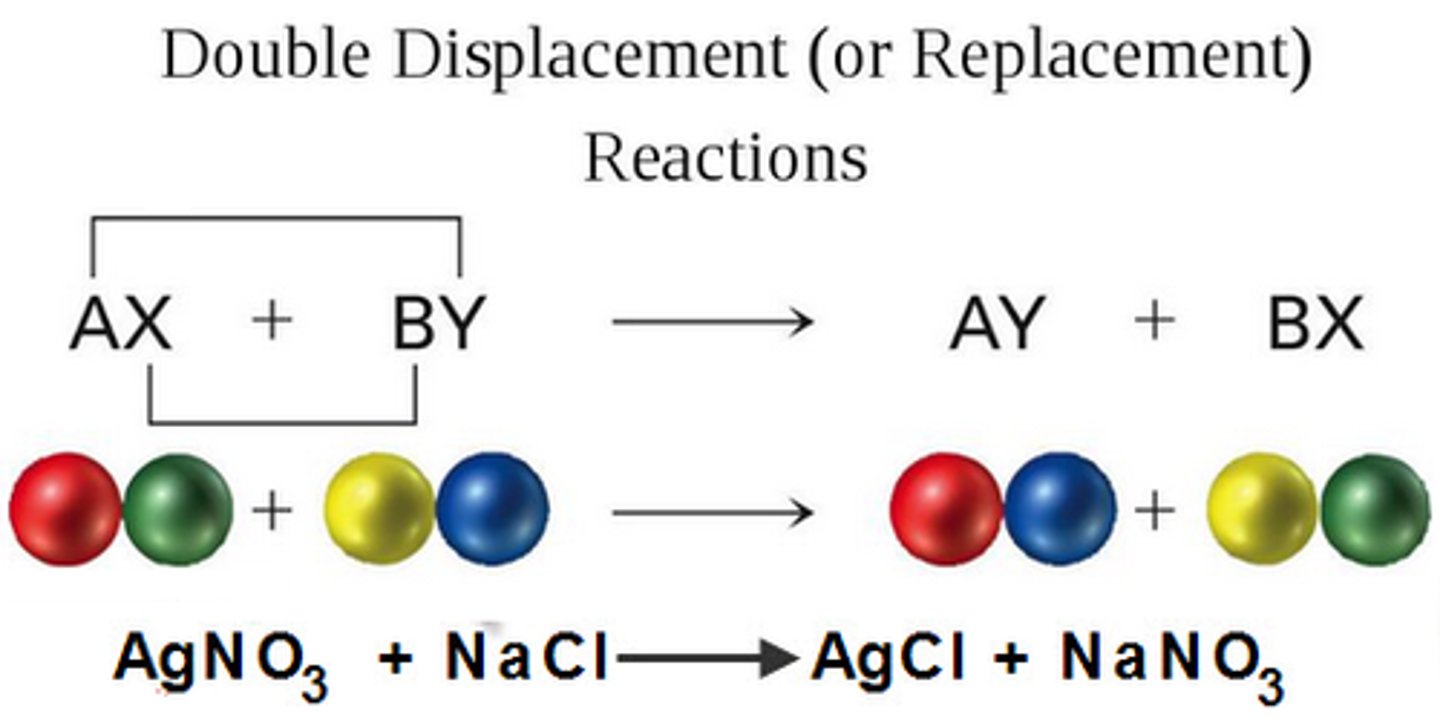
double-displacement reaction
a reaction in which a gas, a solid precipitate, or a molecular compound forms from the apparent exchange of atoms or ions between two compounds
precipiation reactions
2 ionic=2 new solids where 1 is precipitate (insol) and other is aq

acid base reaction
a reaction where an acid reacts with a base to produce water and a salt (ionic compound)
when base is CO2 a carbonate also forms