Bacteriology and Oral Diseases; pptx 10
1/44
Earn XP
Description and Tags
Dr. cugini slides
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
45 Terms
what are things to think about and keep in mind?
current evidence is challenging the idea that we are born sterile
when we emerge form the womb we become further inoculated
microbes outnumber human cells at the level of 100:1 to 1:1(if ur including platelets)
we live in harmony with out (commensal) flora-generally
Disease, may times is a chance event
A good pathogen does not kill its host
bacteria are our friends
where does our microbiome come from?
acquisition form our mother and primary caregivers
window of infectivity
~3/4 of our microbiome maternally-derived
Evidence of transmission along family and social networks
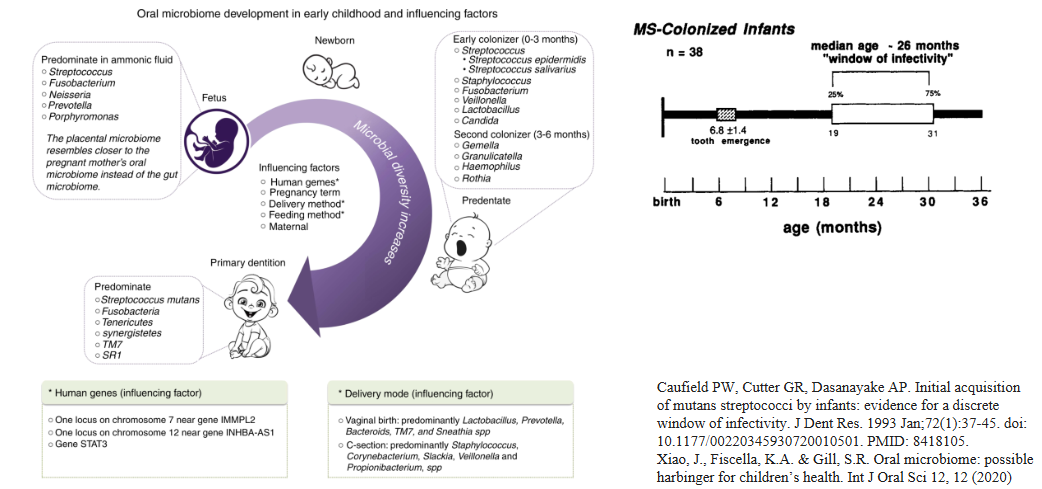
infant microbiome shaped by external factors beginning in utero
mode of delivery
maternal and pediatric diet composition and diversity
breast milk vs cow’s milk formula
antibiotic exposure
societal pressures can impact microbiome health
children born in industrialized nations present fewer species diversity in their gut flora
The infant microbiome matures until ~2-3 years of age
gut microbiota takes on adult properties
*look at the images and diagrams in pictures and read their descriptions
Consequences of microbiome alteration (Dysbiosis)
Dysbiosis
a fundamental shift in the microbial flora generally from a population in homeostasis and health to one that is pathogenic
Many times low abundance organisms (pathobiont?) shift the microbiome by altering the metabolism thus changing the ecology
causes of dysbiosis:
Host genetics (diabetes; immune dysfunction; polymorphisms)
Mechanical (anatomy; overhanging margins in then mouth)
antibiotic use (removal of commensal flora)
Pregnancy (generally reversable)\
Diet
Age
Chicken and egg conundrum
*still trying to understand what is it about this shift that goes from healthy to diseased microbiome (particularly for periodontal disease)
what does this picture table tell you?
It tells you the oral microbiome implications and children’s diseases (diseases of dysbiosis, shift in the flora present) the and it tables (from left to right)
the health condition in the child
the oral microorganisms with increased relative abundance
usually pathogenic bacteria
oral microorganisms with decrease relive abundance
study type from which association identified

Dysbiosis result in:
Diseases and syndromes associated with dysbiosis
Disruption or dysfunctional colonization during infancy linked to immune dysregulation and increased risk of disease into adulthood
Allergies
Diabetes
Leukocyte adhesion deficiency
Irritable bowel disease
Autism spectrum disorder
Heart disease
Rheumatoid arthritis
Periodontal disease....
Why does our microbiome fail us?
This question is an active area of research.
Researchers are actively trying to understand why the microbiome changes
Can we then predict diseases of dysbiosis if we understand pre-dysbiotic events and microbial shifts?
Indicator species
Indicator host cytokines, other proteins, metabolites
Biomarker analysis
Periodontitis as a model for understanding the shift
Inflammatory condition of infection of the gum (gingival mucosa) resulting in bone and tooth loss
Periodontitis and statistics about it
Periodontal diseases are highly prevalent and costly
Effects up to 90% of the worldwide population
Effects half of the US population
$108 billion dental services in the US
$18 billion in Europe
5-10% of medical expenses
~50% of adults living in the USA have gum disease
Most common infectious disease!!!
Eight classifications of Periodontal disease
Gingivitis
Dental plaque-induced gingival diseases
Nonplaque-induced gingival lesions
Chronic periodontitis - the focus for this lecture
Localized
Generalized
Aggressive periodontitis
Localized
Generalized
Periodontitis as a manifestation of systemic diseases
Necrotizing periodontal diseases
Necrotizing ulcerative gingivitis
Necrotizing ulcerative periodontitis
Abscesses of the periodontium
Gingival abscess
Periodontal abscess
Pericoronal abscess
Periodontitis associated with endodontic lesions
Developmental or acquired deformities and conditions
only really focusing on the first 3, chronic periodontitis for this lecture, aggressive periodontitis for next lecture. we are sticking with this classification structure for this class but make note of the new classification scheme
New Classification Scheme for periodontal disease:
Necrotizing periodontitis
Periodontitis as a direct manifestation of systemic diseases
Periodontitis (stage and grade designations)
very controversial, basically split opinion on this change, shift to more clinical classifications, presented in 2017
Chronic Periodontitis
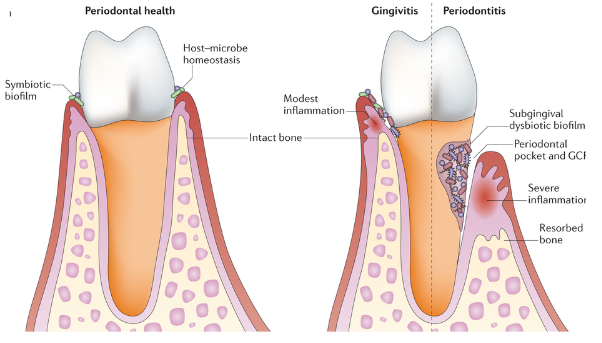
Periodontal disease is a disease resulting from an inflammatory response to a microbial challenge
The challenge is initiated by bacteria that accumulate on the tooth surface above the gum forming plaque
There is a distinct progression to disease
The bacteria is known to be inflammophilic
The host responds it the dysbiotic biofilm through an inflammatory response, this leads to tissue and osseous support destruction
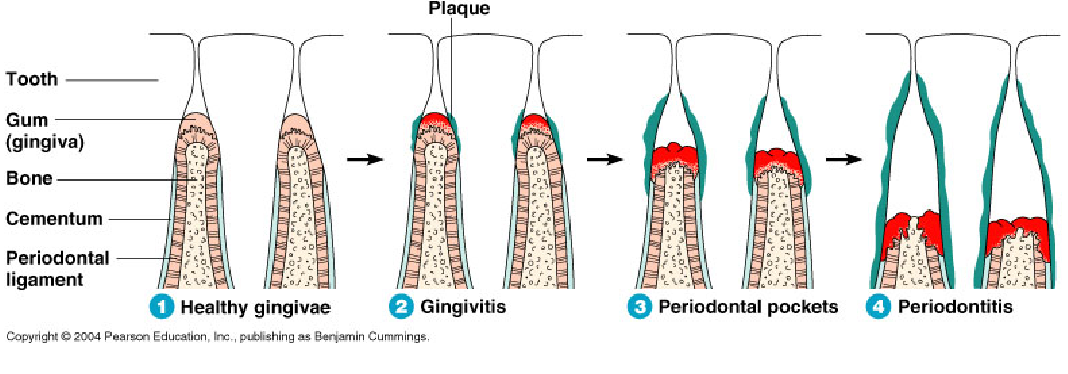
*look at picture to see the difference between normal health, gingivitis, periodontitis
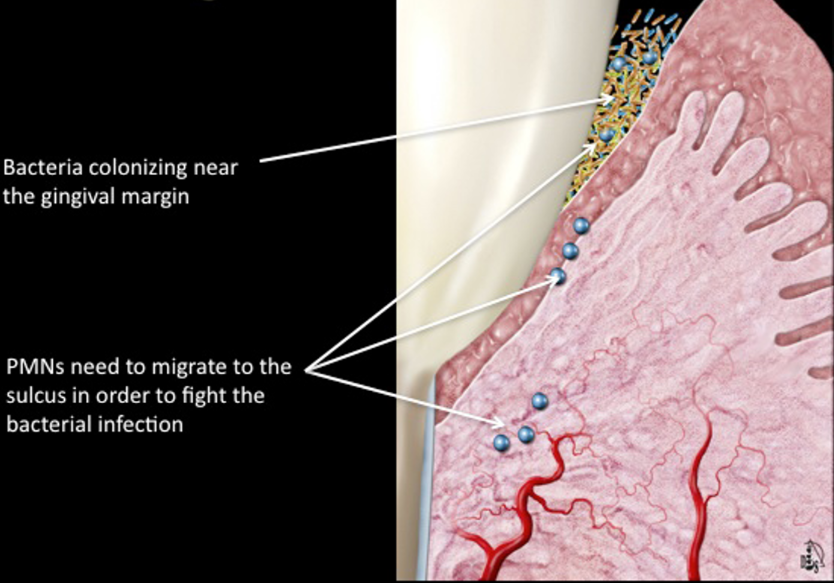
subclinical gingivitis
bacteria that colonize near the gingival margin
PMNs need to migrate to the sulcus in order to fight the bacterial infection
usually okay as long as the body can maintain the bacteria or if you can remove the bacteria
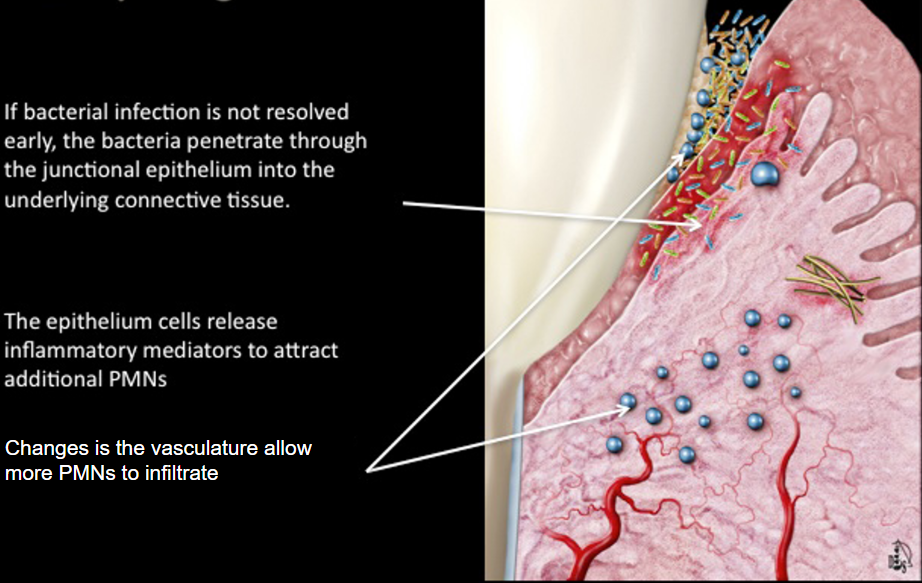
early gingivitis
If bacterial infection is not revolved early, the bacteria penetrate through the junctional epithelium into the underlying connective tissue
the epithelium cells release inflammatory mediators to attract additional PMNs
changes in the vasculature allow more PMNs to infiltrate and the loosening of the tissues allows more bacteria to come in
still reversable
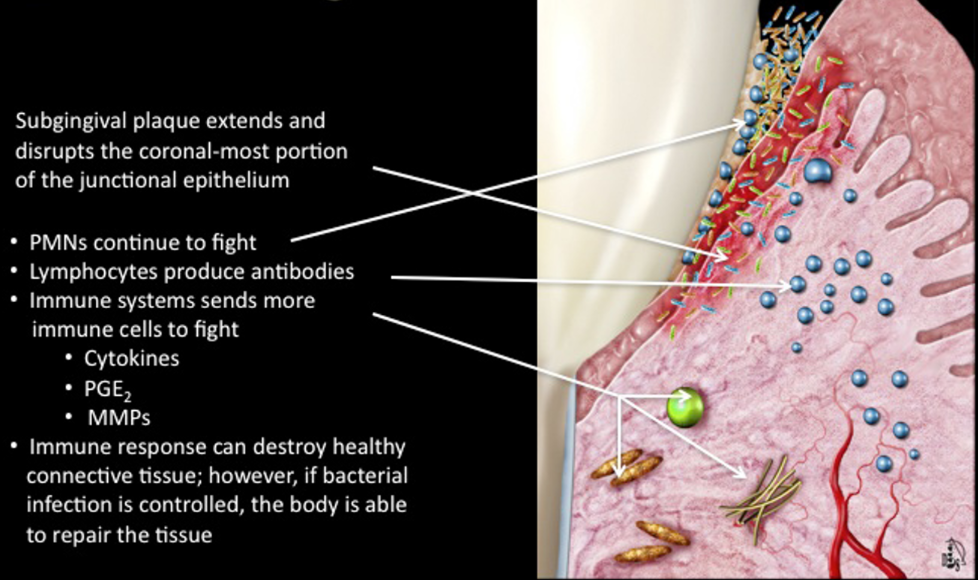
Established Gingivitis
subgingival plaque extends and disrupts the coronal-most portion of the junctional epithelium
immune response can destroy healthy connective tissue; however, if the bacterial infection is controlled, the body is able to repair the tissue
the destroyed connective tissue also act as an additional food source for the bacteria
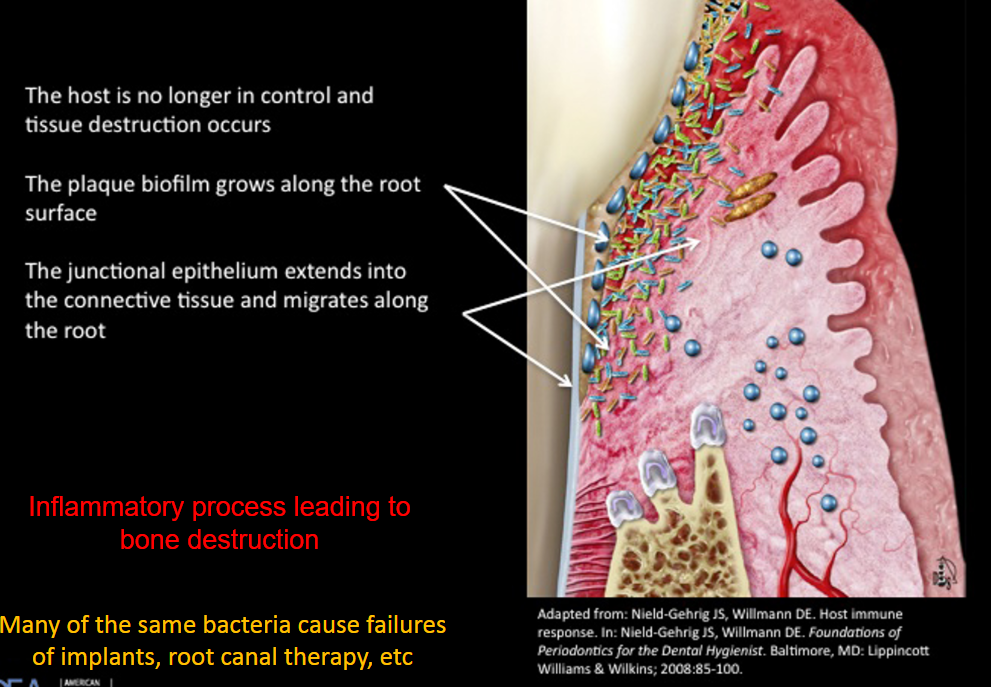
Periodontitis
the host is no longer in control and tissue destruction occurs
the plaque biofilm flows along the root surface
the junctional epithelium extends into the connective tissue and migrates along the root
inflammatory process leading to bone destruction
Many of the same bacteria cause failures of implants, root canal therapy, etc
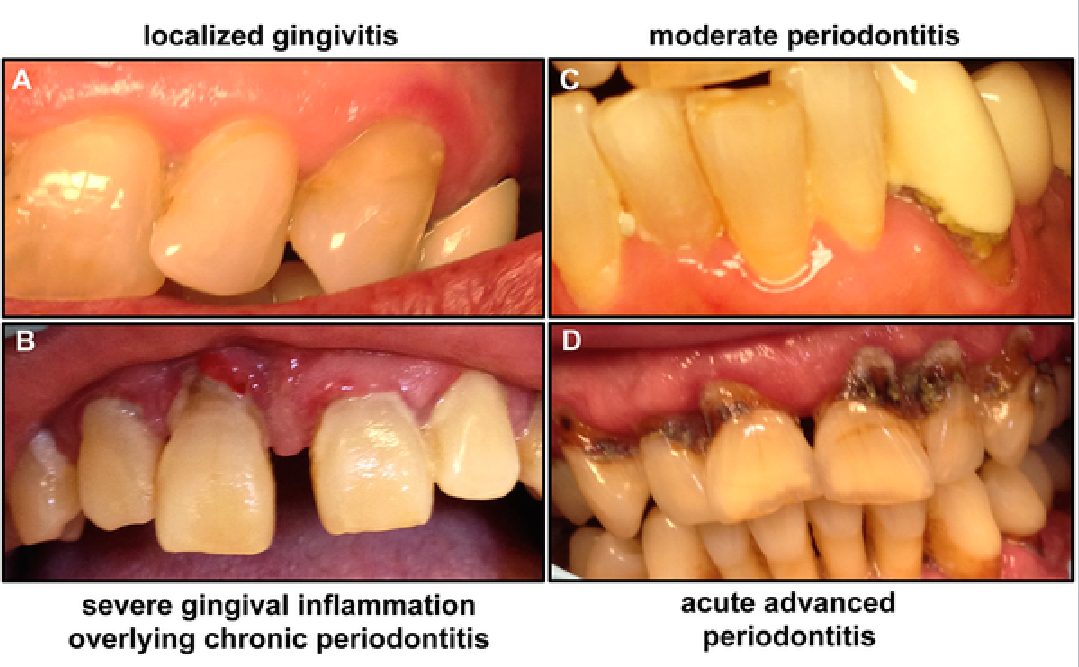
Periodontitis porgression
localized gingivitis
severe gingival inflammation overlying chronic periodontitis
moderate periodontitis
acute advanced periodontitis
The balance between bacteria and host in periodontal disease
Inflammation drives selection of obligate anaerobes
Inflammation is an important source of nutrient:
Degraded collagen and heme containing products, used by proteolytic & asaccharolytic bacteria
GCF: rich serum exudate
Inflammophilic bacteria or pathobionts have a survival advantage
resistance factors
specifics in pic
Risk factors
specifics in pic
*LOOK AT PICTURE
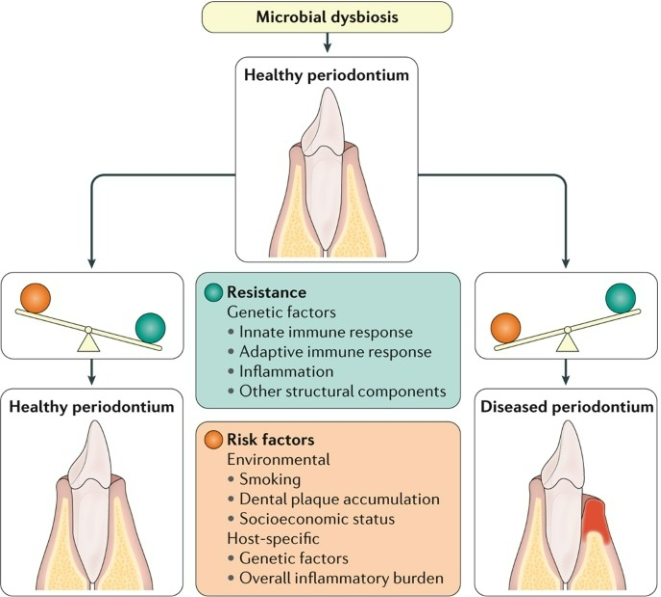
what is this picture basically representing
its representing a summery of periodontitis
chronic periodontitis
A disease of dysbiosis
Microbial imbalance on or inside the body
causes fundamental shift in the bacterial population
outgrowth of pathobionts
gram positive, Aerobic, predominately streptococci ——> Gram negative, anaerobic
whare are changes that occur within the plaque
Population increases
Number of bacteria
Eventual increase in species
Change nutrient availability
Increase and decrease of different nutrients
Shift in the metabolism
Plaque becomes more anaerobic
Drives shift in population and nutrients
Host response to irritation
Also leads to nutrient changes for the bacteria
Bacterial changes that allow for more growth, more virulence factor production, and better attachment
what are late colonizers can be referred to as
periodontopathogens or periodontopathogenic bacteria
Dr. cugini calls them pathobionts
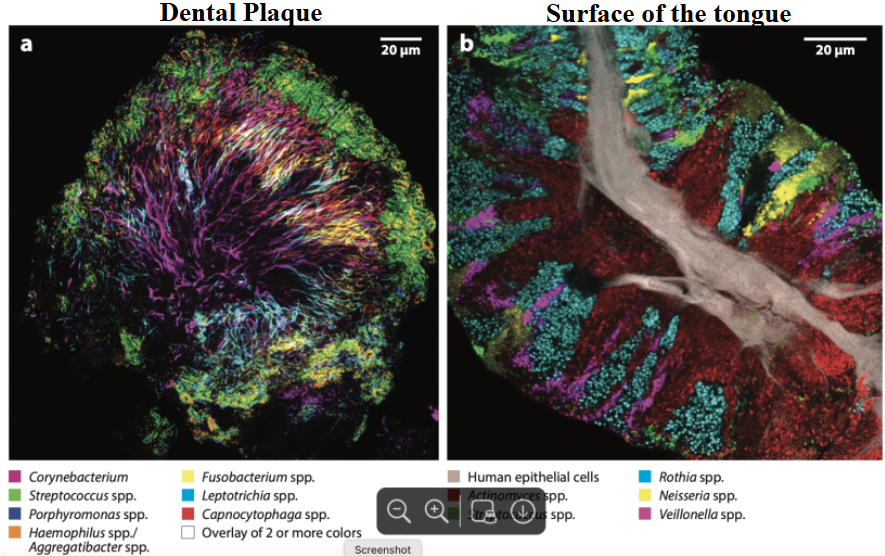
colonized sites are not homogenous and each site have a different set of bacteria colonizing them, why?
it is mainly because of the metabolic interactions present in the plaque
the prevalence of virulence factors
adherence factors on the bacteria
not all can bind to a specific surface or each other
*when testing for identification of present bacteria, will get different signatures depending on site, see image for examples
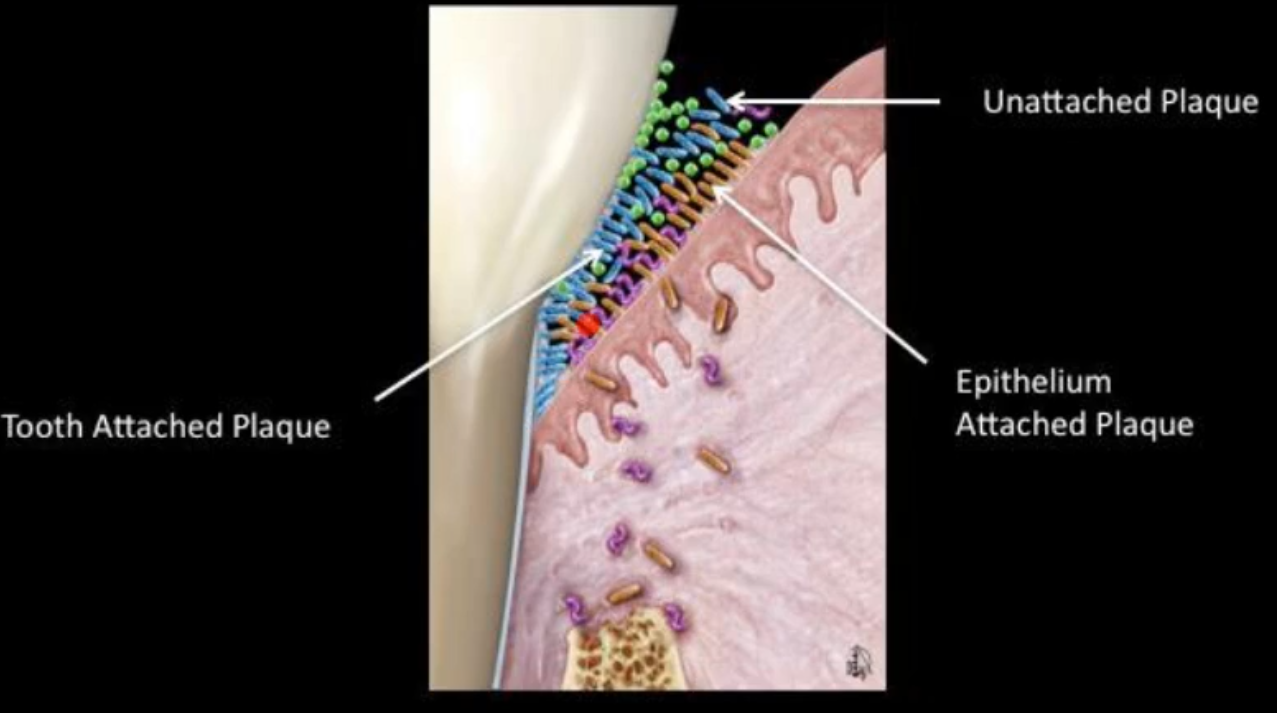
what are the 3 zones of subgingival plaque biofilm?
Unattached plaque
epithelium attached plaque
tooth attached plaque

this is a picture of bacteria that commonly associated with periodontal disease
Red - the main?
orange - can also be associated with disease
green - early colonizers
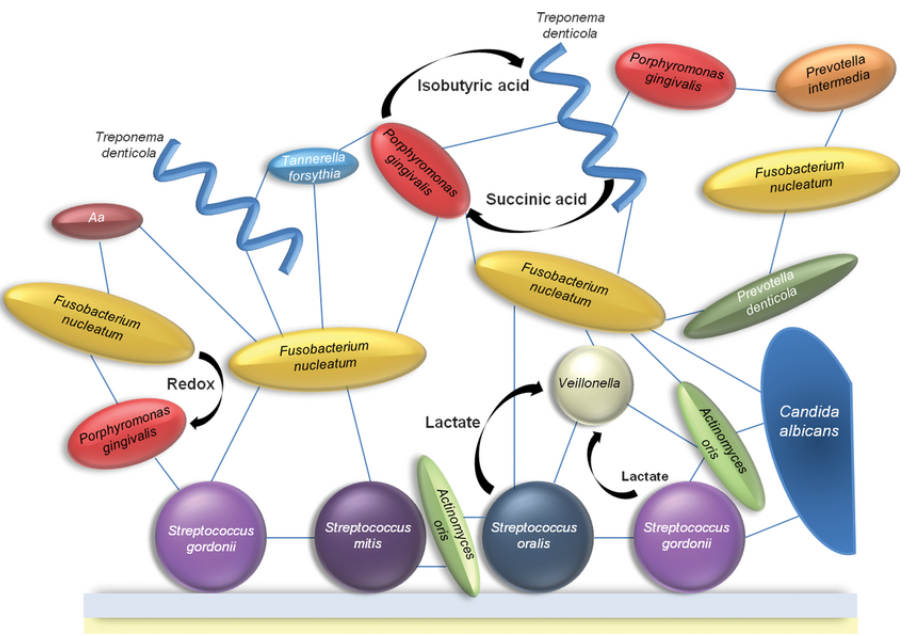
what does this picture show u?
it shows the known interactions within the plaque biofilm and how they can metabolically help each other
What are the qualities for a good periodontal pathogen?
Most are pathobionts- Bacteria that are present in healthy people but increase in number in the plaque because of the environment or loss of host defense.
Their pathogenic qualities are attributed to the following:
• their ability to colonize subgingivally
• their invasive capacity
• their armamentarium of proteases and exotoxins
• their capacity to orchestrate destructive immune responses
Most do not grow well in oxygen-they are anaerobes etc.
what is promoting the shift of the population in chronic perio?
dysbiosis, the shift in the biofilm
If the biofilm is allowed to form on the tooth and it goes below the gingival crevice, what happens?
As the bacteria migrate down in to the subgingival crevice, there is the creation of unique environment that allows for out growth of low-abundance Gram-, anaerobic bacteria
Red complex can thrive
These are highly proteolytic
The center of the biofilm is anaerobic, get a shift in the bacteria’s metabolism
GCF fluid is nutrient rich, good source of “food” for the bacteria
There is protection from the host immune defenses
since we have all these bacteria in our mouths anyway, why don’t we all have chronic periodontitis
mechanical factors
how well u brush
Host genetics
non-permissive environment
microbe-microbe interactions
T/F only people that have poor OH can develop periodontitis
False, you can be healthy or have poor OH and can develop periodontitis; the reason for this is not clear yet
what are the facts about periodontitis
it involves in a rise of the G- bacteria
probably LPS initiation to trigger inflammatory responses
from inflammatory response and tissue breakdown get the increase
more nutrients
allowing more to grow and an increase in bacteria that can grow deeper into the gingiva due to the presents of that nutrient
we know what organism rise in perio:
T. denticola
T. forsythia
P. gingivalis
focusing on this for this lecture because it is the easiest to grow/study
Porphyromonas gingivalis
gram-negative, nonspore forming, anaerobe
distinct black pigment
is a protective mechanism
takes 2 heme groups and combines them(forms bisheme) and then layers itself with bisheme which protects the bacteria from oxygen radicals
Asaccharoyltic
does not use sugars
prefers peptides, dipeptides, proteins
associated with chronic periodontitis
Natural member of the oral cavity...not only there with disease!
was confirmed by detection in children so we know it colonizes early
At low levels in health
Increase detection in a disease state
Not clonal in the oral cavity
Pathobiont
what is the purpose of Socransky’s revision of Koch’s Postulates
koch’s postulates work for acute disease but don’t work for disease for dysbiosis or diseases in a microbiome so socransky revised them
Socransky’s revision of Koch’s Postulates
Isolate organism from disease
Show that organism has virulent traits that are associated with pathogenesis of disease
Show that the organism can produce disease in a healthy animal following Koch’s postulates
Show that the host is responding to the organism
Eliminate the organism and show that the disease resolves
Show that if the organism returns the disease also returns
Does P. gingivalis fulfills Socransky’s revision of Koch’s Postulates?
yes
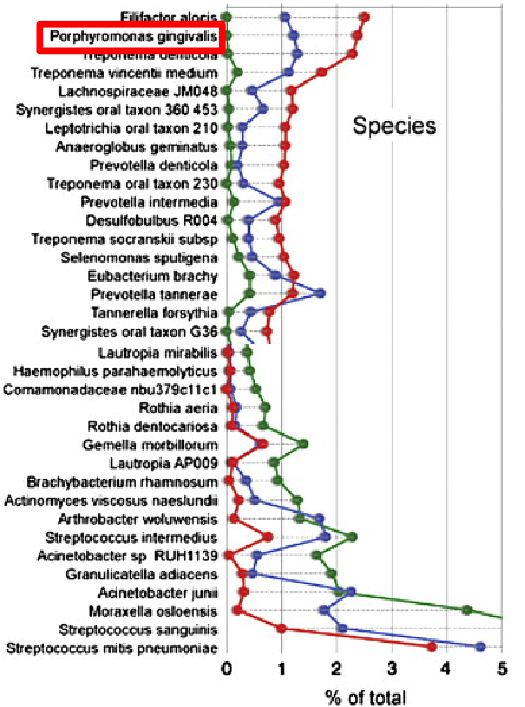
what does this chart show you?
it is a good example of the first postulate, also shows how porphyromonas gingivalis passes this postulate:
found in high percentage of total plaque from patients shallow and deep pockets
contrast with lower level in health
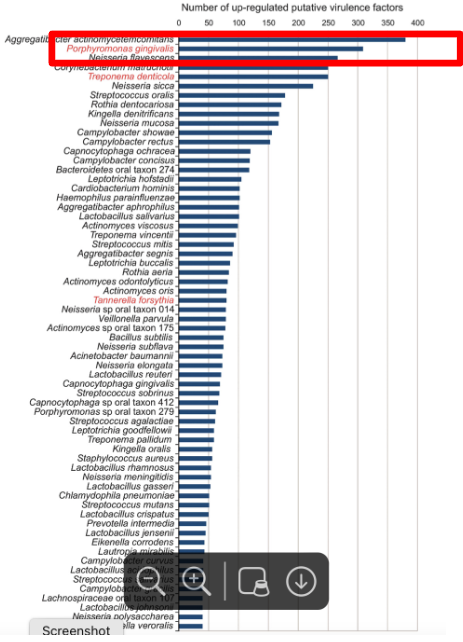
what does this chart show you? (healthy vs diseased)
it is a good example of the second postulate, also shows how porphyromonas gingivalis passes this postulate:
subgingival plaque samples from 7 healthy vs 6 periodontitis patients
metatranscriptome (mRNA sequencing)
Asked what virulence factors were found to be increased in the perio plaque
what are P. gingivalis virulence factors
Proteases- i.e. gingipains, collagenase, deiminase
Protein breakdown and nutrient acquisition
Immune system modulation and evasion
Biofilms, cell-cell attachment, adhesion
Surface attached, secreted, or in blebs/vesicles
Capsule
Immune evasion
Hemolysin
RBC lysis
Hemin acquisition
Fimbria, hemagglutinin
Cell-cell aggregation
Biofilm formation
*has a very unstable genome; Virulence factors of P. gingivalis are focused on finding food and shelter!
p. gingivalis biofilm formation
very similar to other bacterial biofilm formation but can change its gene expression for capsule and fimbria depending on the surface it is in contact with and to or not to form a biofilm
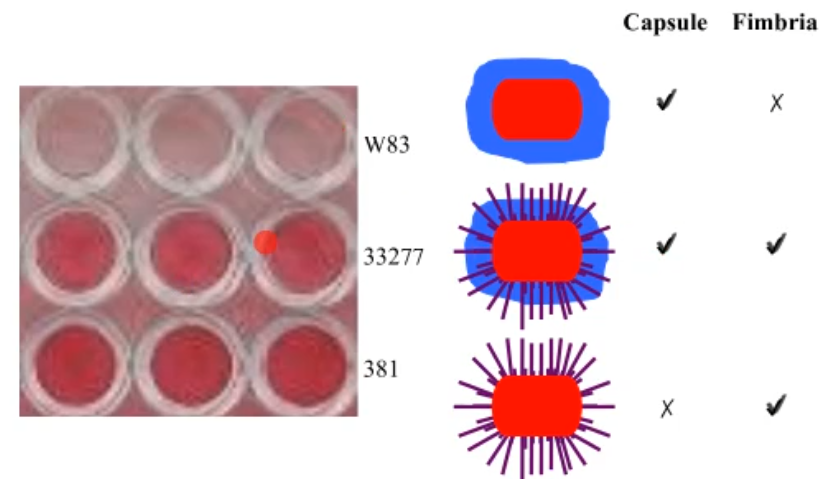
what promote biofilm formation? the gene expression of what bacterial strcutre?
the fimbriae, it facilitates aggregation, surface attachment and biofilm formation; this results in antimicrobial resistance, agent cannot get to the site!
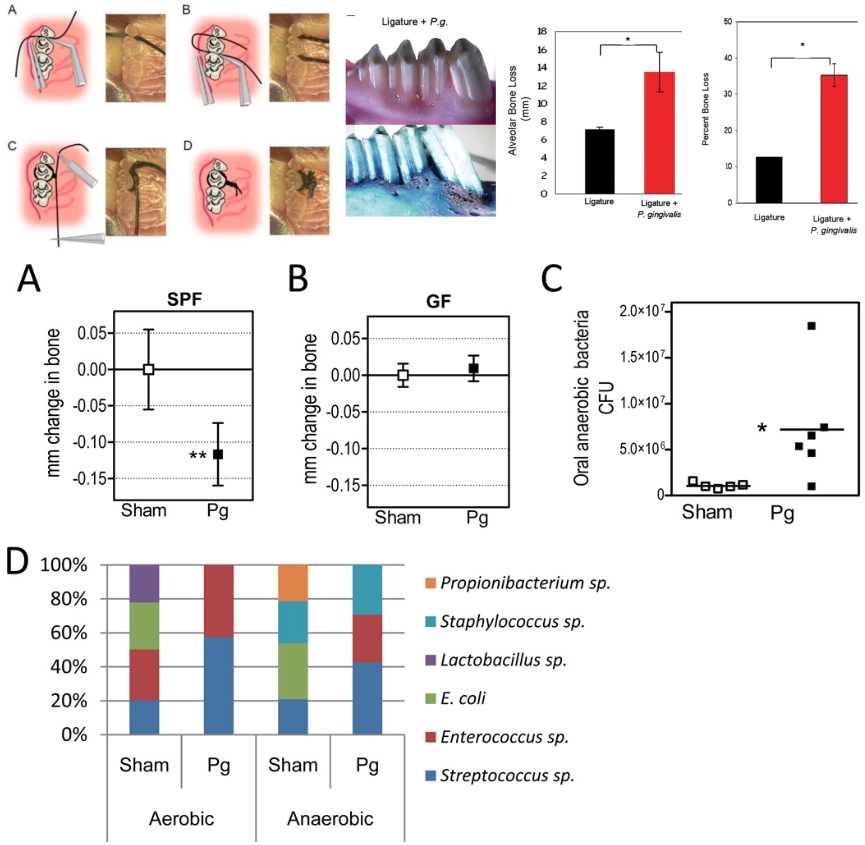
what does these images show you?
it is a good example of the third postulate, also shows how porphyromonas gingivalis passes this postulate:
Shows a test showing how P. gingivalis can cause disease in mice and rabbits in a ligature-induced model(top) and how P. gingivalis can cause bone loss and a change in the microbial flora in mice (bottom half)
these tests basically show how the presents of P. gingivalis can cause bone loss and a shift in the CFUs for oral anaerobic bacteria
*for the study done in mice SPF - specific pathogen free mice (means they were never exposed to PG until the study was performed), GF - germ free mice (means after the mice were born were put in a bacteria free environment; never encountered bacteria, immune system never adapted to encounter bacteria)
Does P. gingivalis make the host respond?
The host produces antibodies against P. gingivalis virulence factors and other surface proteins including antibodies against:
Fimbria
Peptidyl arginine deiminase
Heat shock proteins
Gingipains
The host produces cytokines when P. gingivalis is present (these cytokines are related to inflammation)
IL8
IL6
TNFα
Neutrophils are recruited
*these show how porphyromonas gingivalis passes the fourth postulate
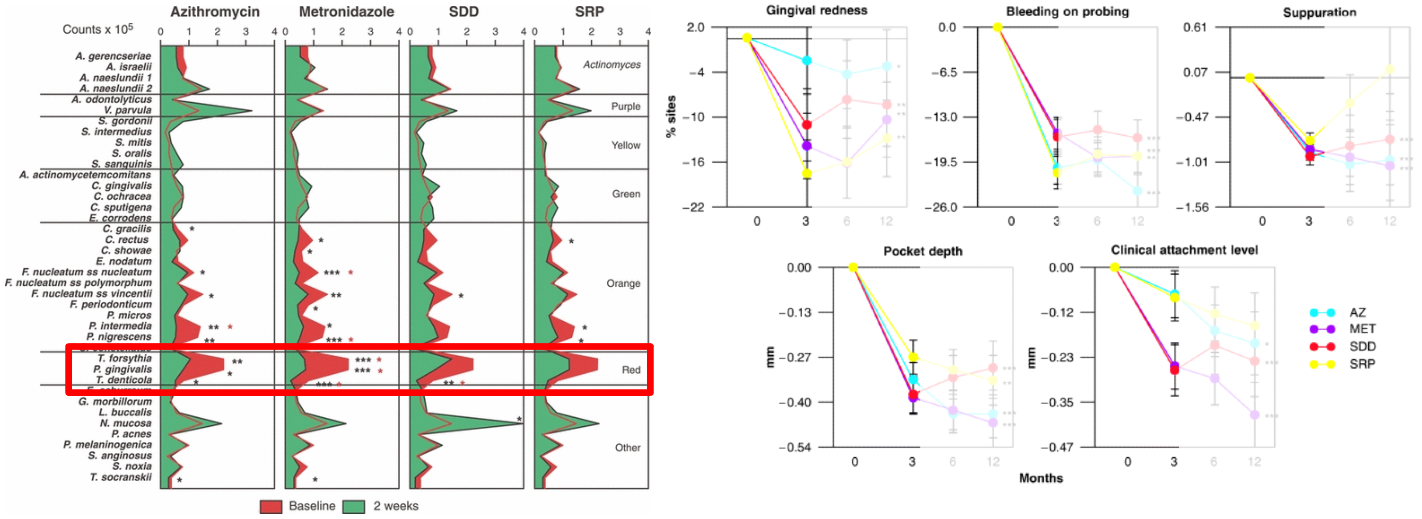
what do these pictures show you?
The pictures show how porphyromonas gingivalis passes the fifth postulate:
Upon antibiotic therapy and SRP treatment P. gingivalis is reduced(left) and how upon treatment symptoms resolve (right)
*for the left picture, the P. gingivalis was treated with Azithromycin metronisazole, SDD (sub-antibacterial dose doxycycline) and SRP (scaling and root planning)
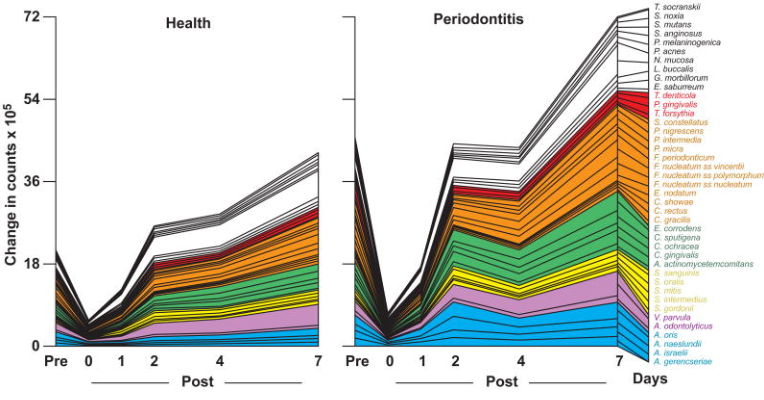
what do these pictures show you?
The picture show how porphyromonas gingivalis passes the sixth postulate:
P. gingivalis increases over time in the absence of dental hygiene
At the entry visit, after the initial monitoring and sampling, all periodontitis subjects received full mouth scaling and root planning (SRP) at a single visit, using manual curettes and ultrasonic devices, followed by polishing and flossing.
Periodontally healthy subjects received a dental prophylaxis using rubber cup and paste, followed by dental flossing.
After the initial prophylaxis or SRP, subjects refrained from oral hygiene procedures for 7 days
what are future plans researchers have for P. gingivalis
Identify using techniques presented how the organism interacts with other member of the oral cavity
Can we understand precisely what genes are changed in health to disease
Can we use it to look for biomarkers at the chair side before we have disease-aim is for personalized treatments
How can the inform we gained on P. gingivalis be used in treatment plans?
Generally microorganisms associated with pockets deeper than 4 mm are reduced to very low or even undetectable levels after periodontal treatment.
Scaling and root planning (“deep cleaning”) or periodontal surgery, disrupt the microbiota of the periodontal pocket by mechanically scraping the biofilm off the root surface to a point that the microbial richness and biodiversity are significantly decreased.
Antibiotic treatments alone or in combination with scaling and root planing disrupt the relative proportions of the taxa within the community.
However, when the selective pressure is lifted, the community tends to return to equilibrium without persistence of antibiotic resistance in the various taxa