CHEM 2370, Chapter 8, Exam 4
1/59
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
60 Terms
Hydrogenation: regiochemistry/rxn type
Syn addition, stereospecific
Hydrogenation: conditions
H2, metal catalyst, pressure
Hydrogenation: products
alkane with an H on either side, syn
Acid Catalyzed Hydration: mechanism
alkene takes an H from catalyst → carbocation rearranges → water attaches to carbocation → catalyst takes an H
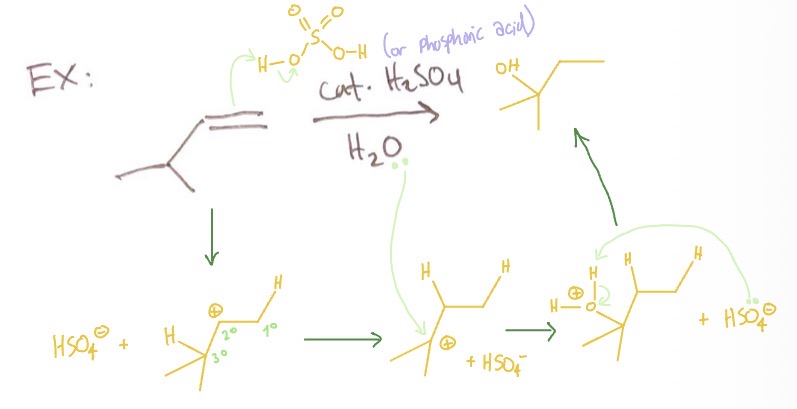
Acid Catalyzed Hydration: conditions
catalyst H2SO4 or H3PO4, H2O
Acid Catalyzed Hydration: products
alkene with OH on highest degree outside of where alkene was
Acid Catalyzed Hydration: regiochemistry/rxn type
mark, addition of water, rearrangement possible
Acid Catalyzed Hydration: intermediate
carbocation
Oxymercuration-Demercuration: conditions
Hg2+, THF/H2O, NaBH4, OH-
Oxymercuration-Demercuration: products
alkane with OH on higher degree, H on lower
Oxymercuration-Demercuration: OR- variant
alkane with OR instead of OH
Oxymercuration-Demercuration: regiochemistry/rxn type
mark, addition of water, no rearrangement
Hydroboration-Oxidation: regiochemistry/rxn type
anti-mark, no rearrangement, syn addition, stereospecific
Hydroboration-Oxidation: conditions
BH3, THF, H2O2, OH-
Hydroboration-Oxidation: products
alkane with H on higher degree and OH on lower degree
Ionic Addition: mechanism
alkene takes an H from HBr/ HI on lower degree → carbocation rearranges → Br-/I- attaches to carbocation
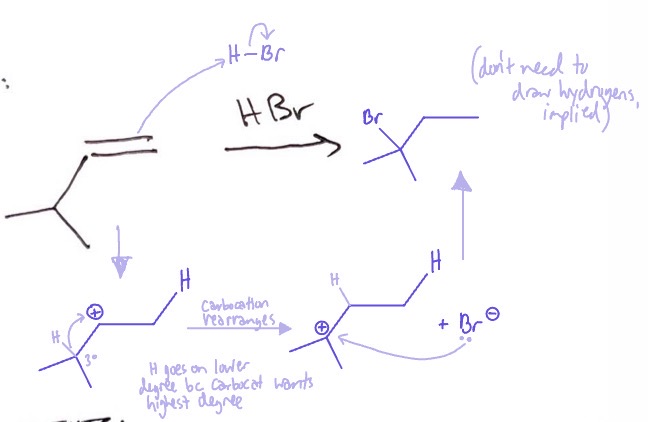
Ionic Addition: conditions
HBr or HI
Ionic Addition: products
alkane with Br/I on highest degree carbon outside of where alkene was
Ionic Addition: regiochemistry/rxn type
mark, rearrangement possible
Radical Additions: regiochemistry/rxn type
anti mark
Radical Additions: conditions
HBr/HI and peroxide
Radical Additions: products
Br/I on lower degree, H on higher degree
Halonium Ion: mechanism
alkene takes partially positive Br/Cl to form pseudocarbocation and EN → Br-/Cl- attaches to higher degree partially positive carbon and EN → SN2 like inversion and EN
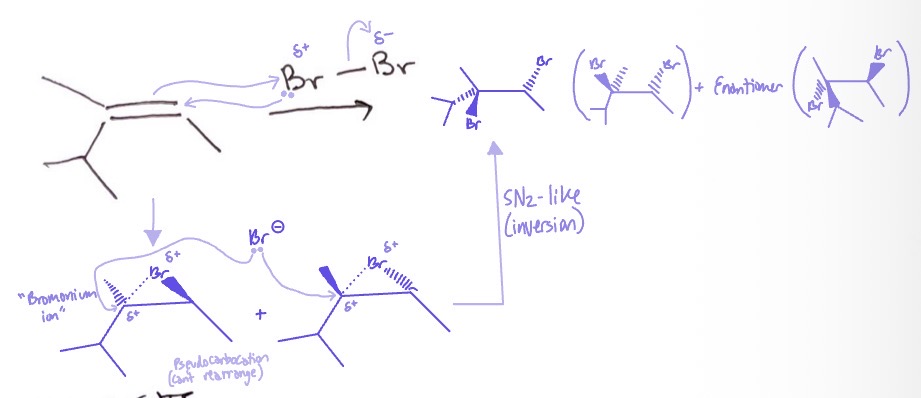
Halonium Ion: conditions
Br2 or Cl2
Halonium Ion: products
Br/Cl on either side and enantiomer
Halonium Ion: regiochemistry/rxn type
anti-addition, no rearrangement, SN2-like
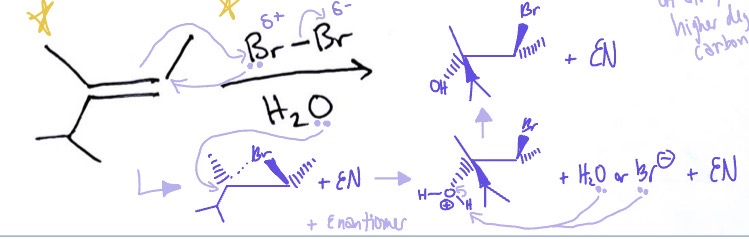
Halonium Ion: Halohydrin variant
Z (cis) alkene, Br2/Cl2, H2O → Br/Cl on lower degree side and OH on higher degree side and EN
Simmons-Smith Cyclopropane Synthesis: mechanism
alkene and carbene attack each other → cyclopropane

Simmons-Smith Cyclopropane Synthesis: conditions
carbene (CH2 / CR2)
Simmons-Smith Cyclopropane Synthesis: products
syn cyclopropane
Simmons-Smith Cyclopropane Synthesis: regiochemistry/rxn type
syn addition
Simmons-Smith Cyclopropane Synthesis: in-situ carbenes
diiodomethane (CH2I2), Zn(Cn) → cyclopropane
Syn-Dihydroxylation: conditions
Z alkene, OsO4, pyridine, NaHSO3/H2O
or
E alkene, KMnO4, OH-, cold, H2O
Syn-Dihydroxylation: products
OH on either side and enantiomer
Syn-Dihydroxylation: regiochemistry/rxn type
syn additions, no rearrangement
Oxidative Cleavage: conditions
KMnO4, OH-, heat, H3O+
or
O3, CH2Cl2, -78°C, Me2S
Oxidative Cleavage: products
KMnO4 form gives ketones and/or carboxylic acids
Ozone form gives ketones and/or aldehydes
Alkynes: H2, metal catalyst, pressure
hydrogenated all the way to an alkane

Alkynes: H2, Ni2B or P-2
syn addition of H, product is alkene
Alkynes: Lindlar’s Catalyst
syn addition, alkene product
Alkynes: Li or Na, NH3Cl or CH3CH2NH2
anti addition of H, product is alkene
Alkynes: halogenation
X2 + alkyne → anti addition of X on alkene + X2 → second anti addition of X on alkane
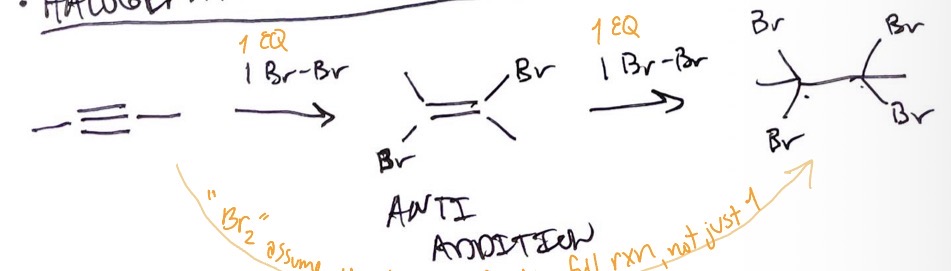
Alkynes: hydrohalogenation
alkyne + excess HX → alkene with X and H on opposite sides + EN → alkane with 2 X and 2 H on opposite sides and EN
Alkynes: addition of water, mercury form
alkyne, HG2+, H3O+, H2O → mark enol → ketone
Alkynes: addition of water, boron form
alkyne, BH3, THF, H2O2, OH- → anti-mark enol → aldehyde
alkyne: oxidative cleavage, potassium permanganate form
alkyne, KMnO4, OH-, heat, H3O+ → carboxylic acid + CO2
alkyne: oxidative cleavage, ozone
alkyne, O3, CH2Cl2, -78°C, Me2S → 2 carboxylic acids
Terminal Alkyne Synthesis
viscinal/geminal halide has 2X + excess NaNh2 → alkene with 1X → alkyne → deprotonated alkyne + H3O+ → alkyne
alkyne ion synthesis
NaNH2 takes H from alkyne → alkynide ion
reactions of alkynide ion
good leaving group leaves alkane and deprotonated alkyne attaches in its place
hydrohalogentation intermediate
carbocation
hydroboration with peroxide intermediate
radical
hydration intermediate
carbocation
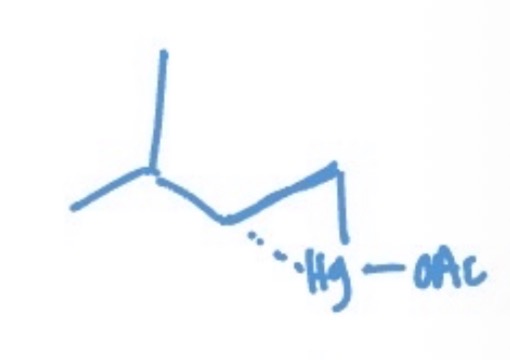
oxymerc-demerc intermediate
mercurinium ion bridge
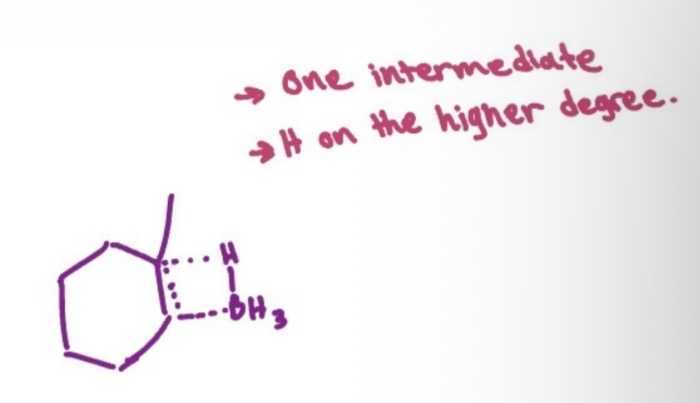
hydroboration-oxidation intermediate
hydroxy-boranes
alcohol addition intermediate
carbocation
halogenation intermediate
bromonium ion
bromination in water intermediate
bromonium ion
bromination in alcohol intermediate
bromonium ion
in-situ simmons-smith cyclopropane synthesis intermediate
carbene