Chemical Energetics
1/7
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
8 Terms
Define exothermic reaction
A reaction where heat is released to the surroundings.
Explain in terms of bond making and bond breaking why a reaction is exothermic.
The energy absorbed to break the bonds in the reactants is smaller than the energy released to form the bonds in the products.
Define endothermic reaction
A reaction where heat is absorbed from the surroundings.
Explain in terms of bond making and bond breaking why a reaction is exothermic.
The amount of energy absorbed to break the bonds in the reactants is larger than the energy released to form the bonds in the products.
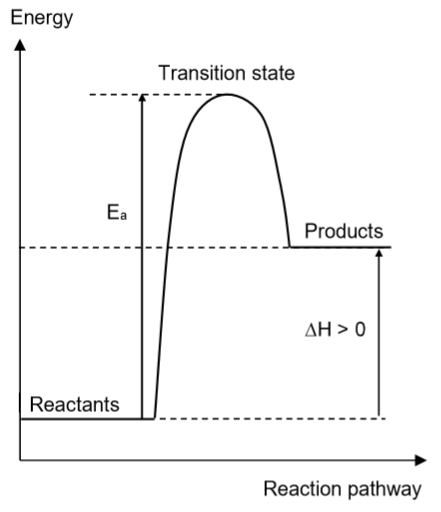
State and explain the type of reaction represented by the diagram
Endothermic reaction. The enthalpy change of the reaction is positive, hence heat was absorbed from the surroundings.
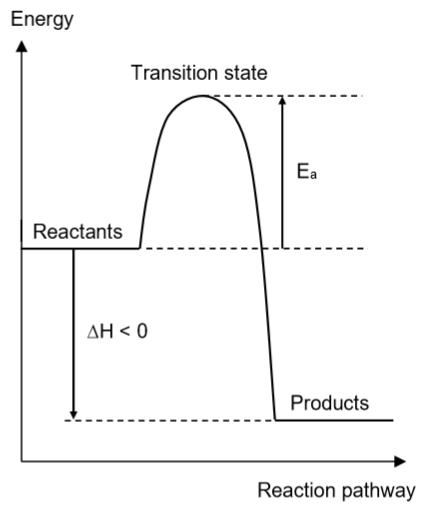
State and explain the type of reaction represented by the diagram
Exothermic reaction. The enthalpy change of the reaction is negative, hence heat was released to the surroundings.
Method to calculate enthalpy change
Q = mcΔT, where Q is energy, m is mass of solution, c is specific heat capacity, ΔT is change in temperature.
ΔH = Q/n, where ΔH is enthalpy change, n is amount of limiting reagent
Reasons why value of enthalpy change of combustion of alcohol calculated in experiment may be less than theoretical value
Heat is lost to the surroundings, so temperature readings are inaccurate.
The combustion of alcohol is incomplete, so the maximum temperature reading is inaccurate.
Some alcohol have evaporated, so the mass of alcohol is inaccurate.