chemical bonding and molecular structure
1/58
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
59 Terms
molecules
group of atoms
since atoms cant exist independently except noble gases
chemical bond
the attractive force that holds various constituents together like molecules, atoms, and ions in different chemical species is called…..
bonding is natures way of attaining stability
Kossel-Lewis approach
1916
first to provide an explanation of valence which was based on inertness of noble gases.
Lewis and his theory
he pictured atom in terms of positively charged ‘kernel”{nucleus and electrons} and the outermost shell that could accommodate 8 electrons that would occupy each corner of a cube
Lewis postulated
atoms achieve the stable octet when they are linked by chemical bonds
kossel brough attention to with respect to chemical bonding
1] the highly negative halogens and highly positive alkali metals are separated by noble gases
2] by formation of negative ion from a halogen and positive ion from alkali metal is associated with gain and loss of an electron
3] the cation and anion thus attain noble gas configuration
4] the anion and cation are stabilized through electrostatic attraction
electrovalent bond and electrovalence
the bond formed as a result of the electrostatic attraction between cations and anions was termed as …..
the electrovalence is equal to the number of unit charge of the ion
octet rule : 1916
electronic theory of chemical bonding
atoms can combine either by transfer of valence electrons from one atom to another
OR
by sharing of valence electrons in order to attain stable octet
covalent bond by Langmuir 1919
he abandoned the idea of a kernel and stationary cubical arrangement of the octet
each bond is formed as result of sharing electron pair between atoms
each combining atom at least contributes one electron to the octet
the combining atoms attain noble gas configuration
formal charge :
in case of polyatomic ions the net charge is possessed by the ion as a whole and not by a particular atom
the lowest energy structure is the one with smallest formal charge on atoms
factor based on a pure covalent view of bonding in which electron pairs are shared equally by neighbouring atoms

limitations of octet rule :
1} incomplete octet [LiCl', BeH2}
2}odd electron molecules
3}expanded octet
4}though the rule is based on the inertness of noble gases it fails to explain why Xenon makes compounds
5} does not account to the shape of the molecule
5} does not explain the relative stability of the molecules
formation of ionic compound depends upon
the ease of formation of the positive and negative ions from their respective parent neutral atoms
the arrangement of the positive and negative ions in the lattice of the crystalline compound
ionic bonds will be formed easily between elements with comparatively low ionization enthalpies and elements with highly negative electron gain enthalpy
electron affinity
the negative of the energy change accompanying electron gain
ionic compounds in their crystalline state
consist of orderly 3-D arrangements of cations and anions held together by coulombic force energies
lattice enthalpy
defined as the energy required to completely separate one mole of solid ionic compound into gaseous constituent ions
bond length
defined as the equilibrium distance between the nuclei of two bonded atoms in a molecule
measured by spectroscopic methods , X-ray diffraction and electron diffraction
covalent radius
measured approx as the radius of an atom’s core which is in contact with the core of another atom
Van Der Waal radius
The Van Der Waals radius is a measure of the size of an atom that is not bonded to other atoms. It is defined as the distance at which the electron clouds of two non-bonded atoms begin to overlap, indicating the effective size of the atom in terms of steric hindrance and molecular interaction. This radius is typically larger than the covalent radius, as it accounts for the presence of van der Waals forces, which include attractions and repulsions between atoms, crucial for understanding molecular behavior, packing in solids, and the interactions between biomolecules.
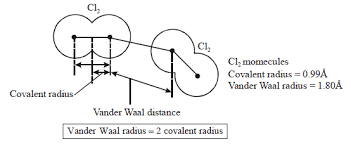
bond angle
defined as the angle between the orbitals containing bonding electron pairs around the central atom in molecules/complex ion
bond enthalpy / dissociation enthalpy
it is defined as the amount of energy required to break one mole of bonds of a particular type between two atoms
KJ mol^-1
bond enthalpy is directly proportional with bond strength
in polyatomic molecules
second time it undergoes a change because of a change in chemical environment
hence average bond enthalpy is used
bond order
number of bonds formed between two atoms in a particular molecule
isoelectronic species and ions have same bond orders
bond order is directly proportional to bond enthalpy
inversely proportional to bond length
resonance
a single Lewis structure is inefficient to represent a molecule, hence a number of structures with similar energies position of nuclei and bonding and non bonding pairs of e-.
this is called resonance hybrid
represented by double headed arrow
features of resonance
it stabilizes the molecule as the energy of the hybrid is less than all the constitute canonical structures
misconceptions of resonance
canonical forms are of no real existence
the molecule does not exist as a single canonical form for a fraction of time.
there is no such equilibrium between the canonical forms as we have between tautomeric forms {keto and enol}
the molecule has a single structure which is the resonance hybrid
polarity of bonds
heteronuclear molecules :
shared pair of electron pair between gets displaced more towards more electronegative element
this also known as dipole moment
dipole moment= charge(Q) * distance of separation ( r)
it is the vector sum of all the dipole moments of various
Fajan’s rule
ionic bonds have partial covalent character
1] the smaller the size of cation and larger the size of anion the greater the covalent character
2] greater the charge of the cation = greater the covalent character
3] for cations with the same size, transition metals are more polarizing than the on with noble gas configuration{alkali/alkaline }
cations polarizes the anion pulling electronic charge towards itself increasing the electronic charge between the two.
VSEPR theory
valence shell electron pair repulsion theory
theory is based on the repulsive interactions of the electron pairs in valence shell
Sidgwick and Powell in 1940
developed by = Nyholm and Gillespie {1957}
postulates of VSEPR
1] shape of a molecule depends upon the number of valence shell electron pairs around the central atom
2] pairs of electrons in valence shell repel one another since the electron clouds are negatively charged
3] these electron pairs tend to occupy such positions in space that minimize repulsion and maximize the repulsion
4] the valence shell is taken as a sphere with electron pairs localizing on the spherical surface at maximum distance from one another
5] multiple bond is treated as if it is a single electron pair and the two or three electron pairs of a multiple bond are treated as single super pair
6] theory is applicable to resonance structure
repulsive interaction between types of electron pairs
lone pair-lone pair > lone pair- bond pair > bond pair -bond pair
difference between the lone pair and bond pair
lone pair are localized on the central atom. occupies more space
bonded pair is shared between two atoms. occupies less space
division of molecules in 2 categories
1] molecules in which central atom has no lone pair
2] molecules in which central atom has one or more than one lone pair
limitations of VSEPR
DOES NOT EXPLAIN THE SHAPES OF POLYATOMIC MOLECULES
Valence bond theory
Heitler and London 1927
discussed in terms of quality not quantity
attractive forces:
1] nucleus of one atom and its own electrons
2] nucleus of one atom and other atoms
repulsive forces:
1]electrons of two atoms
2] nuclei of two atoms
magnitude of new attractiveness is more than new repulsive forces
two atoms approach each other and potential decreases
stage: net force of attraction balances the net repulsion and is said to be bonded .
bond enthalpy
energy gets released after when the bond is formed and hence the bond is more stable than that of isolated atoms.
the energy released is called ………..
orbital overlap concept
in the formation of a molecule there is a minimum energy state where the atoms are so close that their atomic orbitals merge.
this partial merging of atomic orbitals is called overlapping which results in pairing of electrons.
extent of overlapping = the strength of the covalent bond {directly proportional }
they can be positive, negative, neutral/zero depending on the phase and direction of the orientation of amplitude of orbital wave function
how is covalent bond formed
covalent bond is formed as result of pairing two electrons with opposite electron spins
positive overlap
orbitals forming bonds should have same sign/phase and orientation is space
sigma bond
axial/head on overlap
this type of covalent bond is formed by the end to end {head on} overlap of bonding orbitals along the internuclear axis
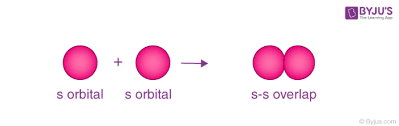
s-s overlap
there is overlap of two half filled s-orbitals along the internuclear axis
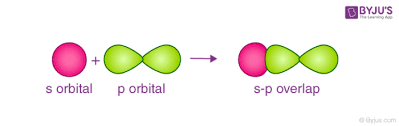
s-p overlap
occurs when half filled s-orbitals of one atom and half filled p-orbitals of another atom overlap
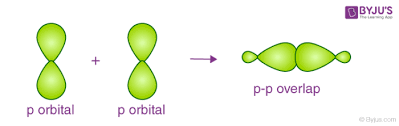
p-p overlapping
between half filled p orbitals
pi bond
sidewise overlapping
atomic orbitals overlap in such a way that their axes remain parallel to each other and perpendicular to the internuclear axes.
consists of two saucer type charged clouds above and below the plane of the participating atoms
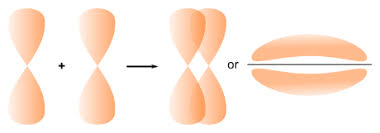
strength of sigma and pi bonds
strength depends upon the extent of overlapping.
sigma : overlapping takes place till large extent. it stronger than pi bond
formation of multiple bonds
pi bond is formed in addition to sigma bond
hybridization
introduced by Pauling
according to Pauling the atomic orbitals combine to form new set of equivalent bonds known as hybrid bonds.
definition: process of intermixing of the orbitals of slightly different energies so as to redistribute their energies. resulting in formation of new set of orbitals of equivalent energy and shape
features of hybridization
1] the number of hybrid orbitals is equal to the number of atomic orbitals that get hybridized.
2] the hybridized orbitals are always equivalent in energy and shape
3] hybrid orbitals are more effective in forming stable bonds then pure atomic orbitals
4]these orbitals are directed in space is a specific preferred direction to minimize repulsion between electron pairs
5] the type of hybridization indicates the geometry og the molecule formed
important conditions for hybridization to happen
1] the orbitals present in the valence shell of the atom must be hybridized
2] the orbitals undergoing hybridization should have almost equal energy
3] promotion of electrons is not necessary condition prior the hybridization
4] it is not necessary that only half filled orbitals participate in hybridization
hybridisation of elements involving d orbitals
the energy of 3d is comparable to 3s and 3p and also to those of 4s and 4p
molecular orbital theory {features}
f Hund and RS Mullikan 1932
1] the electrons i a molecule are present in various molecular orbitals
2] atomic orbitals of comparable energy and similar energy combine to form molecular orbitals
3] in molecular orbital an atom is influenced by more than one nuclei depending on the number of the atoms present in the molecule { atomic orbital- monocentric: molecular orbital - polycentric
4] the number of molecular orbitals formed is equal to the number of atomic orbitals formed
5] when two atomic orbitals combine one bonding and one anti bonding molecular orbital is formed
6] B.O has lower energy and is more stable than ABO
7] the electron distribution probability is give by molecular orbitals
8] filled according to aafbau’s rule, pauli,s exclusion and hunds rule
Bonding molecular orbitals
bonds formed by addition of atomic orbitals.
the two orbitals reinforce each other due to constructive interference
electron density is located between the nuclei of the bonded atoms because of which the repulsion is least
electron tend to hold nuclei together and stabilize the molecule and hence has low energy
energy is lower than the parent atomic orbital
Antibonding molecular orbitals
the electron waves cancel each other due to destructive interference
most of the electron density is located away from the nuclei hence maximum repulsion
electron destabilizes the molecule because of mutual repulsion is more than the attraction
the energy is more than the parent atomic orbital
conditions for combination of atomic orbitals
1] the combining atomic orbitals must have same or similar energy level
2] the combining atomic orbitals must have the same symmetry about the molecular axis [ by convention z axis is taken as molecular axis]
3] the combining atomic orbitals must overlap to maximum extent[ greater the overlap greater the electron density
stability of molecules
it is stable if no of bonding atomic orbitals > no of antibonding atomic orbitals
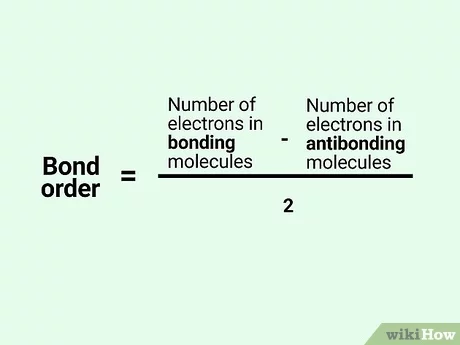
bond order
a positive bond order means stability
negative bond order means instability
bond length
the bond length decreases as the bond order increases
magnetic nature
if all molecular orbitals are doubly occupied, the substance is diamagnetic ( repelled by magnetic field}
if one or more molecular orbitals and singly occupied then it is paramagnetic { attracted by magnetic field}
hydrogen bonding
def—- attractive force which binds hydrogen atom of one molecule with the electronegative atom of another molecule
it is weaker than covalent bond
intermolecular hydrogen bond
it is formed between two different molecules of the same or different compounds
intramolecular hydrogen bond
it is formed when the hydrogen atom id between the two highly electronegative atoms present in the same molecule 2