3 - Phases and Gases
1/84
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
85 Terms
covalent > ionic > metallic > *H-bond > *dipole-dipole > *LDF
* Van Der Waals force
put in decreasing order the strengths of IMF bonds: ionic, covalent, dipole-dipole, London dispersion (LDF), metallic, H-bonds
high IMF = increased BP, MP, viscosity; decreased vapor pressure
what does increased IMF strength have to do with boiling point, melting point, viscosity, and vapor pressure (increase/decrease)?
solids > liquids > gases > ideal gases
rank these substances in the order of decreasing IMFs: ideal gases, gases, solids, liquids
critical point (of phase diagrams)
once you get beyond this point in a phase diagram, the phase can no longer be distinguished (liquid vs. gas, etc.)
ex: fog is liquid and gas
why is fog an example of critical point?
because fog is both liquid and gas
triple point (phase diagram)
point where solid, gas, and liquid all exist in equilibrium in any proportion (ex: 50% gas, 20% liq, 30% solid, etc. any combo)
melting
going from solid to liquid
freezing
going from liquid to solid
vaporization
going from liquid to gas
condensation (condense)
going from gas to liquid
sublimation
going from solid to gas
deposition
going from gas to solid
calorimetry
the science of measuring changes to determine heat transfer (heat required to change the phase of substance)
q = n∆H phase change
n = # of moles
∆H = molar enthalpy of phase change (kJ/mol)
equation to calculate heat required to change phase of a substance?
q = mC∆T :^)
m = mass of substance (g)
C = specific heat (J/g[celsius] = J/g(K)
∆T = change in temp (celsius or K)
equation to calculate heat addition to a substance (without changing its phase)?
q = mC∆T = 18g(4.18J/C)(10C) = 720 J = 0.720kJ
q = n∆H phase change = 1mol(40.68kJ/mol) ~ 40.68 kJ
combine: 40.68kJ + 0.72kJ = 41.5kJ
how much energy is needed to convert 18g of 90C water to 100C steam? specific heat of water is 4.18J/Celsius. molar enthalpy of water is 40.68kJ/mol. need to do calculation sorry…
density
measurement of how condense a substance is d
mass / volume
density formula
directly
external pressure is ___________ (directly/inversely) related to density
inversely
external temperature is ___________ (directly/inversely) related to density
directly
IMF is ___________ (directly/inversely) related to density
Vapor pressure (Pvap)
force exerted by GAS particles that vaporize from a solid/liquid
external pressure has NO effect on vapor pressure
external temperature is DIRECTLY related to vapor pressure
no effect
external pressure is _______________ (directly/inversely/no effect) on vapor pressure
directly
external temperature is _______________ (directly/inversely/no effect) on vapor pressure
CH4 (low IMF)
covalent > ionic > metallic > *H-bond > *dipole-dipole > *LDF
which of the following has the HIGHEST vapor pressure? CH4, H2O, CH3OH, (CH3)2O
CH4 > (CH3)2O > CH3OH > H2O
covalent > ionic > metallic > *H-bond > *dipole-dipole > *LDF
rank based on decreasing vapor pressure? CH4, H2O, CH3OH, (CH3)2O
boiling point (BP)
temp at which condensation/vaporization phase transitions over (Pvap = Patm)
directly
external pressure is _______________ (directly/inversely/no effect) on boiling point
directly
IMF is _______________ (directly/inversely/no effect) on boiling point
melting point (mp) / freezing point (fp)
temp at which fusion or crystallization phase transitions occur
directly
IMF is _______________ (directly/inversely/no effect) on melting/freezing point
directly
external pressure is _______________ (directly/inversely/no effect) on boiling point
B.
IMF: covalent > ionic > metallic > *H-bond > *dipole-dipole > *LDF
which has the highest freezing point?
A. CH4
B. H2O
C. CH3OH
D. (CH3)2O
solution
a homogenous mixture of 2+ substances.
solute present in smaller amounts
solvent present in larger amounts
strong electrolyte → complete dissociation
weak electrolyte → partial dissociation
non-electrolyte → no dissociation
what are 3 types of different solute? (think van’t hoff factor)
van’t hoff factor (i)
# of particles made in solution per moles of substance
ex: glucose (C6H12O6) = 1, NaCl = 2 (Na+, Cl-)
3
what is the van’t hoff factor for Ca(NO3)2?
solubility
the amount of substance that can dissolve in a specific solvent at a specific temperature
unsaturated solution solubility → no precipitation (more solute is needed)
what happens to solubility when concentration < solubility?
saturated solution
add more solute = precipitate
what happens to solubility when concentration = solubility?
supersaturated solution
solute wants to precipitate
what happens to solubility when concentration > solubility?
directly
for solids/liquids in water, solubility is ______________________ (directly/inversely/unaffected) related to temperature
unaffected
for solids/liquids in water, solubility is ______________________ (directly/inversely/unaffected) related to pressure
directly
for gas in water, solubility is ______________________ (directly/inversely/unaffected) related to pressure
indirectly
for gas in water, solubility is ______________________ (directly/inversely/unaffected) related to temperature
A.
which electrolytes are ALWAYS soluble in water?
A. group 1 ions, H+, NH4+, NO3-, CH3COO-, ClO4(-)
B. Ag+, Pb2+, Pb4+, (Hg2)2+, Hg2+, CO3 2-, PO4 3-, S2-
B.
which electrolytes are usually soluble in water?
A. group 1 ions, H+, NH4+, NO3-, CH3COO-, ClO4(-)
B. Ag+, Pb2+, Pb4+, (Hg2)2+, Hg2+, CO3 2-, PO4 3-, S2-
0
According to kinetic molecular theory of gases, ideal gas has ________________ IMFs
ideal gases have ZERO IMFs
has particles w/ negligible volume compared to container
average KE directly proportional to temp (K)
has elastic collisions b/w particles and container walls (creates pressure)
ideality is favored with high temp and low pressure since its interactions of particles are minimized
what are the kinetic molecular theory of ideal gases? (5)
directly
According to kinetic molecular theory of gases, avg kinetic nrg of ideal gas is ________________ (directly/inversely/no effect) related to temperature (K)
high, low
ideal gas favors ________ (high/low) temp and __________ (high/low) pressure since its interactions of particles are minimized
avogadro’s law
volume of ideal gas is proportional to # of particles in container at a given pressure and temperature, regardless of the gas identity (colligative properties)
volume: 22.4 L
temp: 0C or 273K
pressure: 1 atm
what are the volume (L), temperature (K or C), and pressure (atm) of an ideal gas at STP?
P1V1 = P2V2
Boyle’s law formula
boyle’s law
pressure is inversely related to volume (n and T constant)
V1/T1 = V2/T2
charles’ law formula
charles law
temperature is directly proportional to volume
Gay-Lussac’s law
pressure is directly proportional to temperature
P1/T1 = P2/T2
gay-lussac’s formula
1 atm = 760 torr = 760 mmHg = 101 kPa
1 atm = _______ torr = _______ mmHg = ______ kPa?
1L = 1000 mL = 1000 cm³ = 0.001 m³
1L = ____ mL = cm³ = m³?
0C = 273 K
0 Celsius = _______ K?
(P1V1)/T1 = (P2V2)/T2
combo of boyle’s and charles’ law. # of moles (n) is still constant
which 2 ideal gas laws can be combined? what is the new equation? and what are the constants?
PV = nRT
R = 0.08 (L * atm)/(K * mol) = 8.2 (L * Pa)/(K * mol)
don’t forget that volume = 22.4 L/mol in STP in these conditions, if volume is not given
ideal gas law equation. what is the constant R in this equation?
dalton’s law
total pressure of mixture of gas equals to the sum of their partial pressures
Ptotal = P1 + P2 + P3 + … Pn
dalton’s law formula
directly
partial pressure of a gas is ___________ (in/directly) proportional to its mole fraction (X)
Pa = Xa(Ptotal), where Xa = na / ntotal
partial pressure and mole fraction formula
STP so P = 1 atm, V = 22.4 L
change all mass to moles → 0.5 mol He, 1 mol O2, 1 mol CO2
Find XHe: 0.5 mol He / 2.5 mol total = 0.2
PHe = XHe(Ptotal) → 0.2(1 atm) = 0.2 atm He
what’s the partial pressure of He in a mixture of 2g He, 32g O2, 44g CO2 at STP? use calculations.
Graham’s law of diffusion/effusion
rate of diffusion/effusion of gases is inversely related to the square root of its molar mass
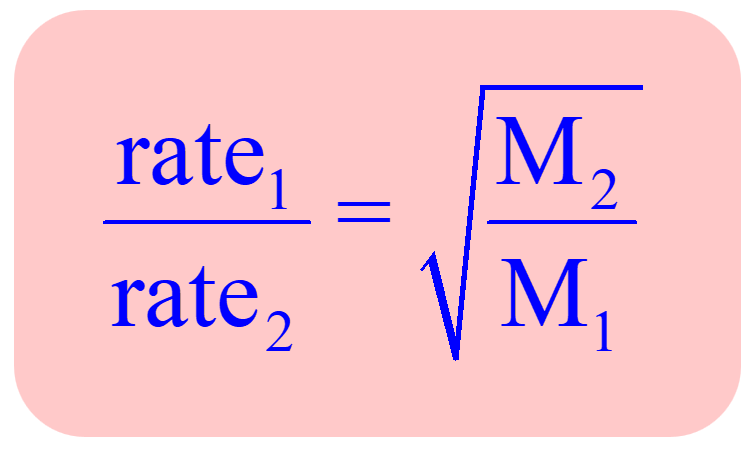
diff/eff rate ∝ sqrt(1/molar mass)
Graham’s law of diffusion/effusion formula
faster
according to Graham’s law, lighter gases (ex: H, He, etc.) diffuse/effuse __________________ (faster/slower) than heavier gases (ex: Ar, Kr, Br, etc.)
decreases (Pideal > Preal)
when particles deviate from ideal gas states (goes into non-ideal), pressure _____________ (increases/decreases) due to an increase of frequency of collisions to container wall
D.
Ideal behavior is seen when the intermolecular forces between particles can be minimized, which occurs at high temperatures and low pressures.
When the pressure of a container of a real gas is less than that predicted by the ideal gas law, this deviation is explained by which of the following statements?
A. The ideal gas law is only valid at very low temperatures and very high pressures.
B. The ideal gas law does not correct for the volume of the particles of gas.
C. The ideal gas law corrects for the shape of the particle of gas, whereas the real gas law does not.
D. The ideal gas law does not correct for the intermolecular forces between the gas particles.
B.
pressure measurements can only be made to assess the force of collisions of gas particles with the container walls, NOT the collisions between particles. for the volume part of the answer choices, remember that ideal gas particles don't occupy space. Therefore, the volume of free space in the container is the same as the entire volume of the container (V – nb = V) for IDEAL!!! However, since real gas particles have significant volume compared to their container size, the volume in the van der Waals equation refers to the free space within the container that is not occupied by all the particles (V – nb < V).
Which of the following statements most clearly defines the P and V – nb terms of the van der Waals equation for real gases?
A. P is the pressure caused by collisions with the walls of the container and between gas molecules, and V – nb is the volume of the container.
B. P is the pressure caused by collisions with the walls of the container, and V – nb is the volume of free space in the container.
C. P is the pressure caused by collisions with the walls of the container and between gas molecules, and V – nb is the volume of free space in the container.
D. P is the pressure caused by collisions with the walls of the container, and V – nb is the volume of the container.
B.
PV = nRT
1 mol O2 makes 2 mols CO2 → 2.5 mol O2 makes 5 mol CO2 (n = 5 mol)
1 atm(V) = 5 mol(0.08)(298K) → 112L
Carbon monoxide gas reacts with oxygen to yield carbon dioxide, according to the balanced equation below. If 2.5 mol of O2 reacts with an excess of CO, what is the maximum volume of CO2 that can be produced at 760 mm Hg and 25°C? (R = 0.0821 L·atm/K·mol)
2 CO + O2 → 2 CO2
A. 61L
B. 122L
C. 112L
D. 5L
A.
constant volume, changing temperature and pressure = gay-lussac law
P1/T1 = P2/T2
600torr/573K = P2/363K → P2 = 380torr
A sample of helium gas at 300°C and 600 torr is in a 200 L container. If the temperature of the gas is raised to 900°C at constant volume, how is the pressure of the gas affected?
A. Pressure will increase by a factor of 2 since the temperature increases by 600°C.
B. Pressure will be unaffected in this instance due to the large volume of the container.
C. Pressure will decrease by a factor of 3 since pressure and temperature are inversely related.
D. Pressure will increase by a factor of 3 since pressure and temperature are directly related.
C.
The rate of effusion is inversely proportional to molecular weight according to Graham’s law. Since N2 gas is lighter than atmospheric air, it would escape more QUICKLY than atm air out of a tire.
Which of the following is NOT a potential advantage to inflating car tires with N2 instead of atmospheric air (79% N2, 20% O2, and 1% other gases including H2O, Ar, and CO2)?
A. Atmospheric air is more likely to cause corrosion of the tire interior compared to N2.
B. Atmospheric air contains water vapor, which is more likely than N2 to condense as the pressure inside the tire increases.
C. If a very small hole were poked in the tire, a tire filled with pure N2 would lose pressure less quickly than a tire filled with atmospheric air.
D. Tires filled with N2 weigh less than those filled with atmospheric air, leading to better fuel efficiency
gas molecules do not liquefy at low temperature
gas molecules don’t experience IMFs (intermolecular forces
gas molecules have negligible VOLUME
what are the most important criteria for an ideal gas? (3)
B.
Two gases, each 1 L at STP, are allowed to spontaneously react in an insulated cylinder with a floating piston to form two new gaseous products such that total moles are constant before and after the reaction. Immediately after the completion of the reaction the volume of the cylinder was measured to be 1.82 L. What can be said about the reaction
A. The reaction was isothermal.
B. The reaction was endothermic.
C. ΔS for the reaction was negative.
D. The reaction was exothermic.
D.
One mole of an ideal gas occupies 22.4 L at STP.
What is the volume of two moles of O2 at STP?
A. 22.4 L
B. 89.6 L
C. 11.2 L
D. 44.8 L
A.
The pressure due to the nitrogen and carbon dioxide gases is 330 + 250 = 580 torr. Since the total pressure is 725 torr, the partial pressure of oxygen is the difference: 725 — 580 = 145 torr. Since 145 is 1/5 of 725, the percent pressure of O2 is 1/5 = 20%.
A flask contains a mixture of three gases: oxygen, nitrogen, and carbon dioxide. The pressure exerted by N2 is 330 torr, and that exerted by CO2 is 250 torr. If the total pressure of the gases is 725 torr, what is the percent pressure of O2?
A. 20%
B. 32%
C. 40%
D. 15%
D.
According to the kinetic theory, which of the following is NOT true of ideal gases
A. All collisions among gas molecules are perfectly elastic.
B. There are no attractive or repulsive forces between gas molecules.
C. For a sample of gas molecules, average kinetic energy is directly proportional to temperature.
D. There is no transfer of kinetic energy during collisions between gas molecules.
D.
Which of the following equations could be used as the basis for determining the relative rates of diffusion of two gases in an enclosed flask once the valve is opened?
A. F = kQq/r2
B. PE = mgh
C. PV = nRT
D. KE = (1/2)mv2
C.
A property of an "ideal" gas is that it is composed of particles that have negligible volume and do not exert intermolecular forces.
at 25C, the volume of an ideal gas follows PV = nRT → 24.4L NOT 22.4L
The term “ideal gas” refers to a gas for which certain assumptions have been made. Which of the following is such an assumption?
A. the law PV = nRT² is strictly obeyed
B. intermolecular molecular forces are infinitely large
C. individual molecular volume and intermolecular forces are negligible
D. 1 gram-mole occupies a volume of 22.4L at 25C and 1 atmospheric pressure