2.4: Properties of Period 3 elements and their oxides
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
What is the equation for the reaction of Na with water?
2Na + 2H2O → 2NaOH + H2
What is the equation for the reaction of Mg with water?
Mg + 2H2O → Mg(OH)2 + H2
Explain which is more reactive with water, Na or Mg?
Na is more reactive
Only has to lose 1 electon, whereas Mg needs to lose 2 electrons
So less energy is required
How can the rate of reaction for Mg and water be increased, and why?
The reaction is originally very slow
Using steam instead of water
As steam provides reaction with greater energy
What is the equation for the reaction of Mg with steam?
Mg + H2O(g) → MgO(s) + H2
What is the observation of the reaction of Mg and steam?
Bright white flame
What are the formulas names and oxidation states for the oxides that form from the period 3 elements?

What is the reaction of Na and O2, what are the reaction conditions, observations and product?
4Na(s) + O2(g) → 2Na2O(s)
Heated
Bright yellow flame
White solid
What is the reaction of Mg and O2, what are the reaction conditions, observations and product?
2Mg(s) + O2(g) → 2MgO(s)
Heated
Bright white flame
White solid
What is the reaction of Al and O2, what are the reaction conditions, observations and product?
4Al(s) + 3O2(g) → 2Al2O3(s)
Heated
Bright white flame
White powder
What is the reaction of Si and O2, what are the reaction conditions, observations and product?
Si(s) + O2(g) → 2SiO2(s)
Powdered Si, heat strongly
Bright white sparkles
White powder
What is the reaction of P and O2, what are the reaction conditions, observations and product?
4Ps) + 5O2(g) → 2P4O10(s)
Heated
White/yellow flame
White clouds
What is the reaction of S and O2?
S(s) + O2(g) → 2SO2(s)
2S(s) + 3O2(g) → 2SO3(g) (only with a catalyst and very high temperature)
Powdered S, heated
Blue flame
Toxic fumes
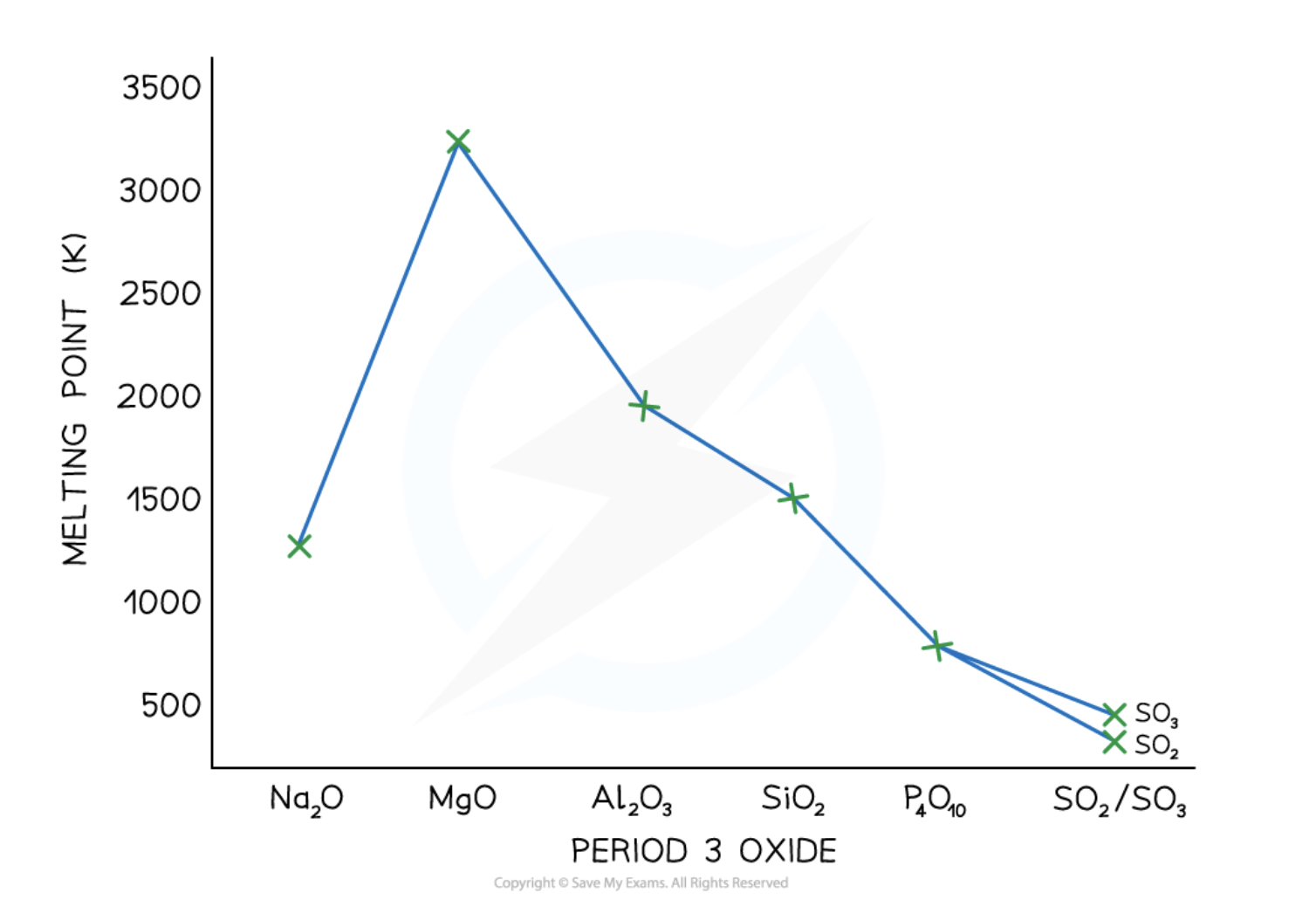
What is the trend of the melting point of period 3 oxides?
Increases and then rapidly decreases
Na2O: ionic
MgO: ionic
Al2O3: ionic but shows covalent character, so much lower than MgO
SiO2: macromolecular
P4O10: simple covalent
SO2: simple covalent
The reactions of the oxides of the elements Na–S with water, limited to Na2 O, MgO, Al2 O3 , SiO2 , P4 O10 , SO2 and SO3 , and the pH of the solutions formed.
What is the equation of Na2O with water, and what is the approximate pH of the solution formed?
Na2O(s) + H2O(l) → 2NaOH(aq)
14
What is the equation of MgO with water, and what is the approximate pH of the solution formed?
MgO (s) + H2O (l) → Mg(OH)2 (aq)
10
What is the equation of Al2O3 with water, and what is the approximate pH of the solution formed?
No reaction, as Al2O3 is insoluble in water
What is the equation of SiO2 with water, and what is the approximate pH of the solution formed?
No reaction, as SiO2 is insoluble in water
What is the equation of P410 with water, and what is the approximate pH of the solution formed?
P4O10 (s) + 6H2O (l) → 4H3PO4 (aq)
2
What is the equation of SO2 with water, and what is the approximate pH of the solution formed?
SO2 (g) + H2O (l) → H2SO3 (aq)
2-3
What is the equation of SO3 with water, and what is the approximate pH of the solution formed?
SO3 (g) + H2O (l) → H2SO4 (aq)
0-1
What is the definition of amphoteric?
Compounds that act as both an acid and a base
What is the equation for the reaction of Na2O and HCl?
Na2O (s) + 2HCl (aq) → 2NaCl (aq) + H2O (l)
What is the equation for the reaction of MgO and HCl?
MgO (s) + 2HCl (aq) → MgCl2 (aq) + H2O (l)
What are the equations for the reaction of Al2O3 reacting with H2SO4/NaOH?
Al2O3 (s) + 3H2SO4 (aq) → Al2(SO4)3 (aq) + 3H2O (l)
Al2O3 (s) + 2NaOH (aq) + 3H2O (l) → 2NaAl(OH)4 (aq)
What is the equation for the reaction of SiO2 and NaOH?
SiO2 (s) + 2NaOH (aq) → Na2SiO3 (aq) + H2O (l)
What is the equation for the reaction of P4O10 and NaOH?
P4O10 (s) + 12NaOH → 4Na3PO4 + 6H2O (l)
What are the equations for the reactions of SO2/SO3 with NaOH
SO2 (g) + 2NaOH (aq) → Na2SO3 (aq) + H2O (l)
SO3 (g) + 2NaOH (aq) → Na2SO4 (aq) + H2O (l)