Glucose Homeostasis
1/47
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
48 Terms
-Where the signalling molecule is released from
-Where the signalling molecule binds to
How do we describe the relationship between the source cell and the target receptor?
-Autocrine (local mediators)
-Paracrine
-Endocrine
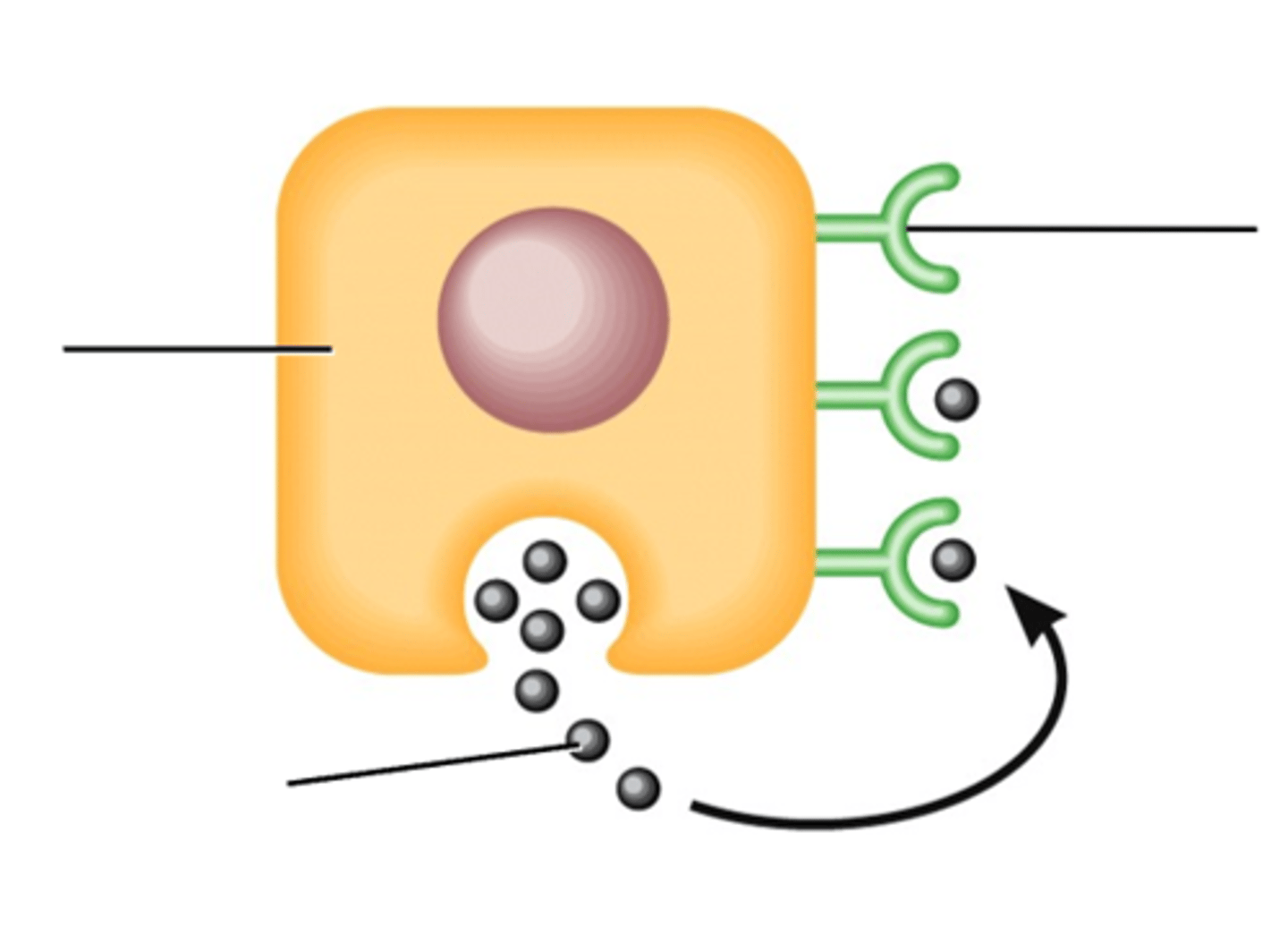
What are the main cell signalling mechanisms?
The source cell produces a signalling molecule that binds to a specific receptor on its own cell surface or receptors inside the source cell.
Describe the autocrine cell signalling mechanism
Macrophages secrete interleukin 1 which binds to a receptor on the macrophage itself.
Give an example of when autocrine cell signalling is used
The source cell releases the signalling molecule which binds to a different cell which is close to the source cell.
Describe the paracrine cell signalling mechanism
-Renal autoregulation
-Neuromuscular junctions
Give an example of when paracrine cell signalling is used
The source cell releases the signalling molecule into the circulatory system where then it can reach any cell of the body which receives blood supply.
Describe the endocrine cell signalling mechanism
Hormonal circulation.
Give an example of when endocrine cell signalling is used
Endocrine.
What cell signalling mechanism is this?
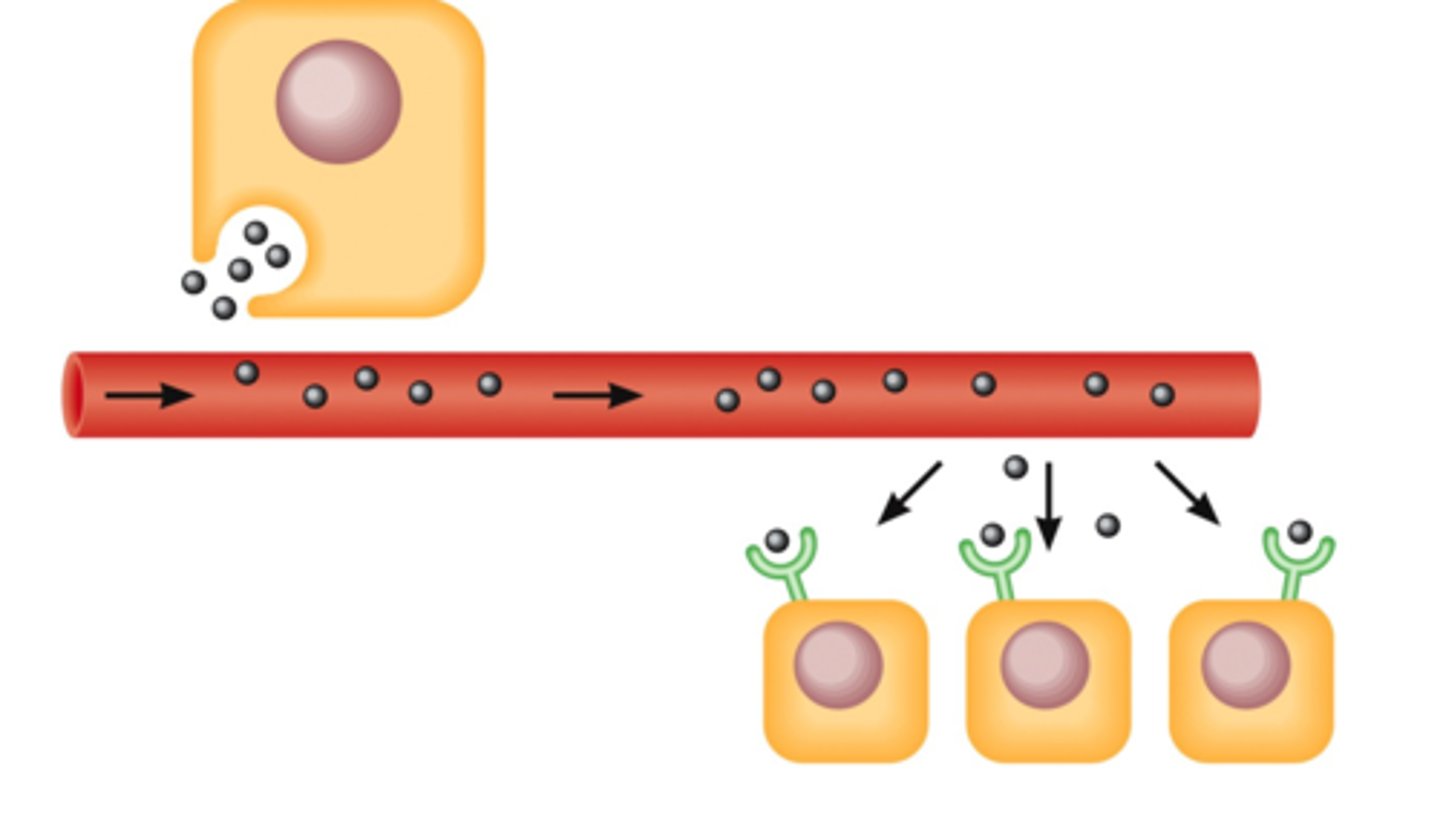
Autocrine.
What cell signalling mechanism is this?
Paracrine.
What cell signalling mechanism is this?

Glands that secrete hormones directly into the circulatory system to be carried toward a distant target.
What are endocrine glands?
-Two lobes
-Right lobe is within the meso-duodenum
-Left lobe is within the deep leaf of the greater omentum
Describe the structure and relational topography of the canine pancreas
Both.
Is the canine pancreas and endocrine or exocrine gland?
-Most of the exocrine cells are arranged into clusters called acini
-Distributed amongst the acini are clusters of endocrine cells
--These clusters are called pancreatic islets (Islets of Langerhans)
Describe the arrangement of exocrine and endocrine cells within the pancreas
They produce digestive enzymes which flow through ducts into the GI tract.
What is the function of the exocrine cells of the pancreas?
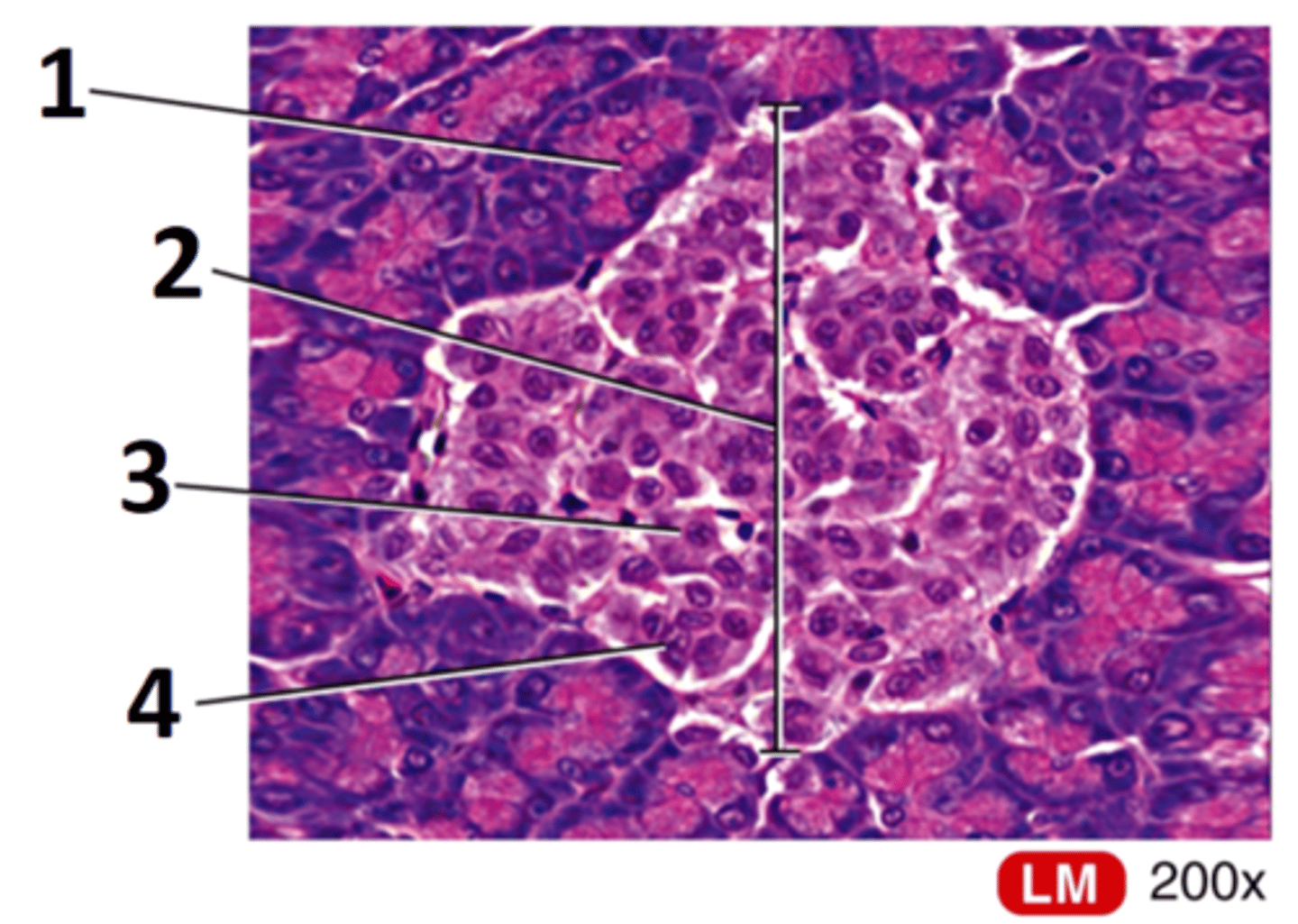
1 = Acini
2 = Pancreatic islet (Islet of Langerhan)
3 = Beta cell
4 = Alpha cell
Label this histological slide of the canine pancreas
-Alpha cell (secretes glucagon)
-Beta cell (synthesise proinsulin, secretes insulin)
-Delta cell (secretes somatostatin)
-F cell (secretes pancreatic polypeptide)
What are the 4 types of cell within the pancreatic islet? What is their function?
5-8 minutes.
What is half-life of insulin?
Increase in blood glucose concentration.
What is the primary control of insulin secretion?
1) Glucose enters the beta cell via GLUT 2.
2) Raised intracellular glucose levels fuels the formation of more ATP.
3) Raised levels of ATP block the potassium channels of the beta cell and hence potassium accumulates within the cytoplasm. This causes depolarisation of the cell membrane.
4) Depolarisation of the cell membrane causes voltage-gated calcium channels to open causing calcium to flood into the cell from ECF.
5) This influx of calcium ions triggers exocytosis of vesicles loaded with insulin (and hence insulin is released)
6) Blood glucose concentration decreases and so ATP production decreases. This means that potassium channels no longer become blocked and the cell membrane repolarises (so insulin secretion stops)
Describe what happens to a beta cell when blood glucose concentration is increased
-Stimulates the uptake of glucose by muscle and fat cells
-Causes drops in blood glucose concentration
What is the action of insulin? What effect does this have on blood glucose concentration?
Negative feedback.
What type of homeostatic feedback is used by beta cells to regulate blood glucose concentration?
GI hormones, GIP and GLP-1:
-Anticipatory release of insulin is thought to prevent a sudden surge in glucose absorption following a meal
Parasympathetic activity:
-Increases insulin secretion during and after a meal
Sympathetic activity:
-Decreases insulin secretion
Increase in plasma amino acid concentration (typically after a meal):
-Increases insulin secretion
Other than blood glucose concentration, what else controls the levels of insulin that are secreted by beta cells and how?
-Moves through specific protein channels (GLUT transporters)
-This is because it is not lipid soluble
How does glucose move into and out of cells? Why?
Increases the expression of GLUT 4 transporters.
How does insulin cause increased glucose reabsorption in cells?
No.
-Brain (uses GLUT 3)
-Mammary gland, GI tract and kidney tubules (use secondary active transport coupled to sodium transport)
Do all cells require insulin in order to take glucose into the cell? If not, which cells do not require insulin?
1) Insulin binds to a tyrosine kinase receptor.
2) This receptor phosphorylates insulin-receptor substrates.
3) Secondary messenger pathways alter protein synthesis and existing proteins
4) Cell metabolism and membrane transport is changed (as more GLUT 4 transport proteins are manufactured).
Briefly describe how insulin effects its target cell
They would starve as they would be completely unable to absorb glucose (so no glycolysis can occur).
What would happen to insulin-dependant cells if they become unresponsive to insulin?
-The liver is responsible for taking excess glucose out of the blood returning from the gut
-Insulin stimulates the liver enzymes to convert the glucose to glycogen to ensure an inward concentration gradient after a meal
Why is the metabolism of insulin different in the liver? How?
GLUT 2.
What glucose transporter is found on the cell surface of hepatocytes?
-Means beta cells in the pancreas do not produce insulin
-Therefore hepatocytes make glucose (glycolysis) and transport it out into the blood using GLUT 2 transporters
What occurs to hepatocytes when there is a drop in plasma glucose concentration (fasted state)?
-Means beta cells in the pancreas are producing insulin
-Glucose enters the hepatocyte using GLUT 2 transporters
-Insulin stimulates hexokinase (which causes glucose-6-P to be produced from glucose)
-Glucose-6-P is then converted to glycogen by glycogen synthase to maintain a low intracellular concentration of glucose
What occurs to a hepatocytes when there is an increase in plasma glucose concentration (fed state)?
-Cells of the CNS are not sensitive to insulin
--This means that the CNS cells themselves can not increase or decrease the rate of glucose transport
-Levels of glucose in the CSF is directly proportional to blood sugar
--SO excess glucose increases the osmolarity of the CSF and draws water out of the neurones
--OR too little glucose causes starvation of the neurones
Why is it essential that blood glucose concentration is maintained to preserve the function and health of the CNS?
-Increases glucose oxidation
-Increases glycogenesis
-Increases Lipogenesis
-Increases protein synthesis
Overall effect = increased stores of glycogen, fat and protein
Summarise the functions of insulin
4-6 minutes.
What is the half-life of glucagon?
Decrease in plasma glucose concentration.
What is the primary control of glucagon secretion?
Increase in plasma amino acid concentration (typically after a meal)
-Stimulates secretion of glucagon
Parasympathetic activity
-Stimulates secretion of glucagon
Sympathetic activity
-Stimulates secretion of glucagon
Other than a decrease in plasma glucose concentration, what else controls glucagon secretion? How?
To prevent postprandial hypoglycaemia in carnivores; promotes the production of glucose if the diet is mainly protein-based.
Why does an increase in plasma amino acids after a meal stimulate the release of both insulin and glucagon?
-Increases gluconeogenesis
-Increases glycogenolysis
-Increases ketogenesis
Summarise the functions of glucagon
-Decreases secretion of growth hormone
-Paracrine inhibition of insulin and glucagon release
What is the function of somatostatin released by delta cells?
Currently unknown.
What is the function of pancreatic polypeptide from F cells?
Type 1:
-Due to inadequate insulin secretion
Type 2:
-Due to abnormal target cell responsiveness
What are the main types of diabetes mellitus? Describe them
Type 1.
What type of diabetes mellitus are dogs more likely to have?
Type 2.
What type of diabetes mellitus are cats more likely to have?
-Hyperglycaemia
-Polydipsia and polyuria (PUPD)
-Ketoacidosis
What are the clinical signs of diabetes mellitus?
-DM causes hyperglycaemia
-This means that the renal threshold for glucose reabsorption is exceeded
-This causes glycosuria (glucose in the urine) which causes increased osmotic diuresis (water movement into the urine)
-This causes an increase in urination (Polyuria), ECF volume decrease and plasma osmolarity increase
-This then stimulates the thirst centre in the hypothalamus (and hence Polydipsia ensues)
Primary polyuria causes secondary polydipsia
How does diabetes mellitus (DM) cause PUPD?
-As the body is in a fasted state (despite having eaten), adipose tissue is broken down causing fatty acids to be released into the bloodstream
-The liver uses Beta-oxidation to break down fatty acids into Acetyl-CoA which can then enter the TCA cycle
-As oxaloacetate (a TCA intermediate) is diverted to produce glucose, the TCA turns slower causing Acetyl-CoA to build-up
-This build-up means that Acetyl-CoA is converted to ketone bodies such as acetone and Beta-hydroxybutyrate over time
-Large amounts of ketone bodies leads to ketoacidosis which will make the animal ill
How does diabetes mellitus cause ketoacidosis?