Biology 55
1/139
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
140 Terms
microorganisms
organisms that are too small to be seen with the unaided eye
Microbes include ___ .
bacteria, fungi, protozoa, microscopic algae, and viruses
Knowledge of microorganisms allow humans to ____ .
- prevent food spoilage
- prevent disease
- understand causes and transmission of disease to prevent epidemics
The Human Microbiome Project (2007-2016)
- determine microbiota of various areas of the body
- understand relationship between microbiome and human diseases
human microbiota
acquired microorganisms (on/in a healthy human being)
An adult human is composed of ____ and _____ .
30 trillion body cells, 40 trillion bacterial cells
A vet gets ready to clean a sea turtle covered in oil following the Deepwater Horizon oil spill in the Gulf of Mexico in 2010. After the spill, the population of a naturally occurring oil-eating marine bacterium called Alcanviorax borkumensis skyrocketed. Why?
- helped get rid of the oil
- scientists are working on ways to genetically engineer this bacterium to be more efficient in cleaning up future spills
Microbes ____ .
- are pathogenic
- decompose organic waste
- generate oxygen by photosynthesis
- produce chemical products (ethanol, acetone, and vitamins)
- produce fermented foods (vinegar, cheese, and bread)
- produce products (biotechnology) used in manufacturing and disease treatment
Ancient use of microbes:
fermented foods and beverages (beer, wine, bread, yogurt, cheese, and pickled vegetables)
Describe the ancient and early years of microbiology.
- Hippocrates (Father of Western Medicine): disease has a natural cause
- Thucydides (historian): observed immunity
- Marcus Terentius Varro: proposed idea of microbes (certain minute creatures)
What did Antoni van Leeuwenhoek (Dutch merchant 1632-1723) theorize life really look like?
began making and using simple microscopes
What were ‘animalcules’ called by the end of the 19th century?
microoganisms
Aristotle proposed ___ .
Spontaneous Generation
Spontaneous Generation
living things can arise from nonliving matter 384-322 B.C.

Describe an experiment that disproves Spontaneous Generation.
Franscisco Redi’s Experiments (late 1600’s)
- one of the first to refute the idea
- when decaying meat was kept isolated from flies, maggots never developed
- meat exposed to flies was soon infested
- as a result, scientists began to doubt Aristotle’s theory

Who disproved Spontaneous Generation?
Louis Pasteur
What did Robert Koch study (1843-1910)?
causative agents of disease
List Koch’s medical contributions.
- developed Koch’s postulates
- identified the causative agent of anthra and TB (cholera)
List Koch’s beliefs.
- suspected causative agent must be found in every case of the disease and be absent from healthy hosts
- agent must be isolated and grown outside the host
- when agent is introduced into healthy host, host must get the disease
- same agent must be re-isolated from now-diseased experimental host
Who supposed the Germ Theory of Disease?
Louis Pasteur, Robert Koch, and Joseph Lister
Germ Theory of Dieases
diseases may result from microbial infection
How can microbes be classified?
kingdom → phygium → class → order → family → genus → species (most specific and basic taxonomic unit)
superkingdom level
distinguishes between organisms that have membrane-bound nuclei in their cells and those that don’t
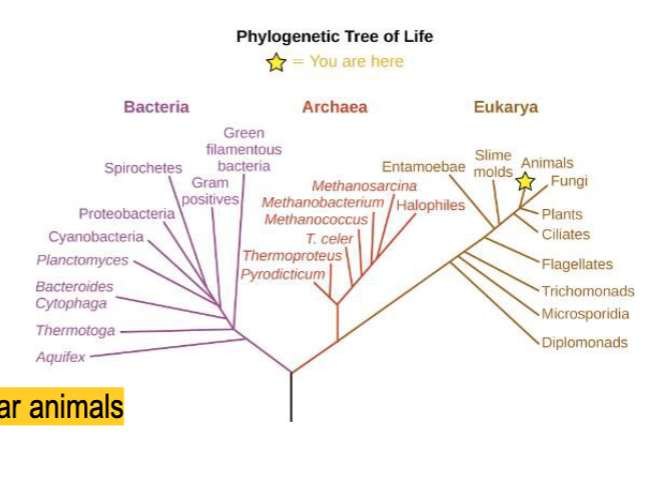
What are the 6 categories that microorganisms are grouped into?
- bacteria or archaea
- fungi, protozoa, algae, or small multicellular animals
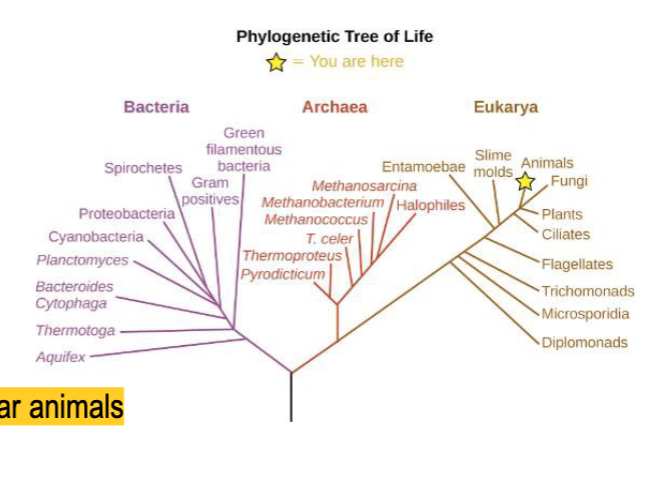
prokaryotic organisms
- unicellular and lack nuclei
- much smaller than eukaryotes
- found everywhere there’s moisture
- some found in extreme environments
- extremophiles are usually archaea
- reproduce asexually
- 2 kinds of prokaryotic organisms: bacteria and archaea
bacteria prokaryotic organisms
cell walls contain peptidoglycan and some lack wall cells
archaea prokaryotic organisms
cell walls contain polymers other than peptidoglycan
eukaryotic microorganisms
- unicellular and multicellular
- have nuclei
- protists, plants, fungi and animals
protist
- unicellular or multicellular eukaryotes
- not plants, animals, or fungi
- similar to animals in nutrient needs and cellular structure
- live freely in water (ex. amoeba)
- some live in animal hosts (parasitic, ex. giardia)
- most are asexual and sexually reproduce
- most are capable of locomotion by: pseudopodia, cilia or flagella
algae (protist)
- unicellular and multicellular eukaryotes
- photosynthetic
- simple reproductive structures
- categorized on basis of pigmentation and storage product composition of cell wall
- many consumer products used contain ingredients from algae
protozoa (protist)
- single-celled eukaryotes
- similar to animals in nutrient needs and cellular structure
- live freely in water
- some live in animal hosts
- most are asexual and reproduce sexually
- most are capable of locomotion
fungi
not plants not photosynthetic
Molds and yeasts are fungi that are categorized as ___ .
eukaryotes with cell walls but not cellulose
molds
- decomposers that obtain food from other organisms
- growth structures (hyphae/mycelium)
-reproduce by sexual and asexual spores
yeasts
- fermentation mostly beneficial but causes spoilage and disease
- reproduce asexually by budding
- some produce sexual spores
Diseases caused by helminths involved ___ .
microscopic eggs and larvae
viruses
acellular microorganisms
matter
anything that takes up space and has mass
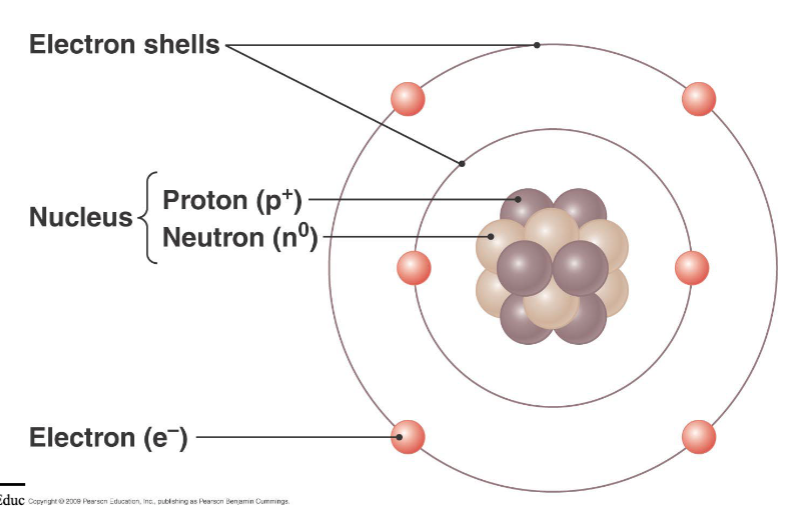
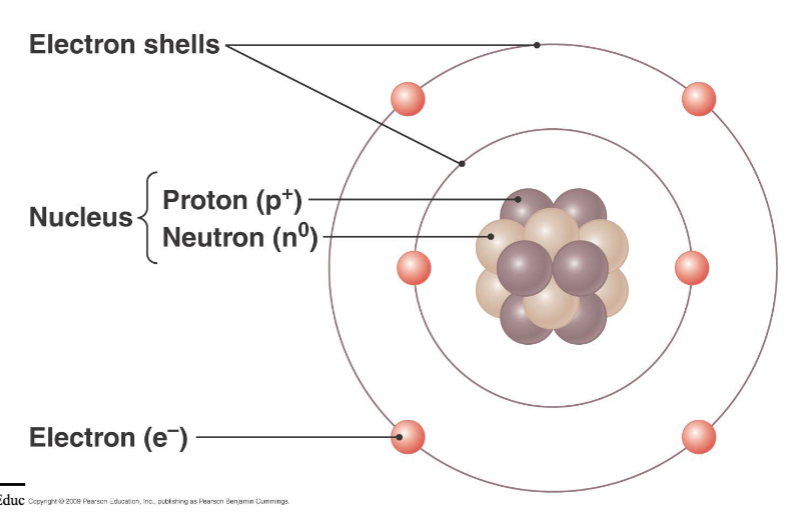
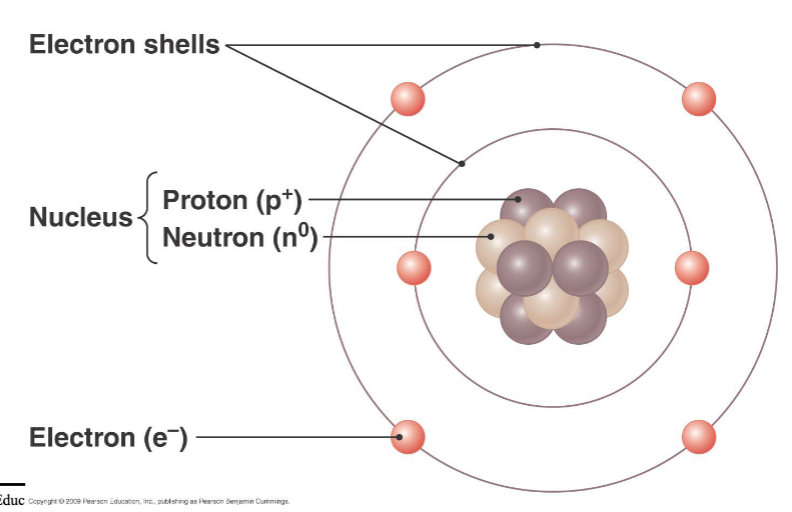
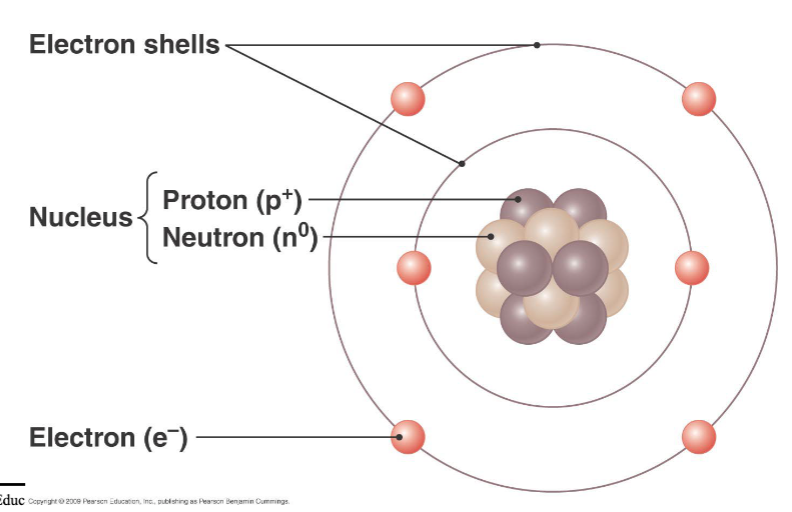
atoms
smallest chemical units of matter
About how many elements are essential to life?
25/92
Which elements make up 99% of living matter (dry weight of cells)?
C, H, O, N, P, and S
Which elements make up most of the rest of the 1% of living matter?
Ca, K, Zn, Fe, Na and trace elements
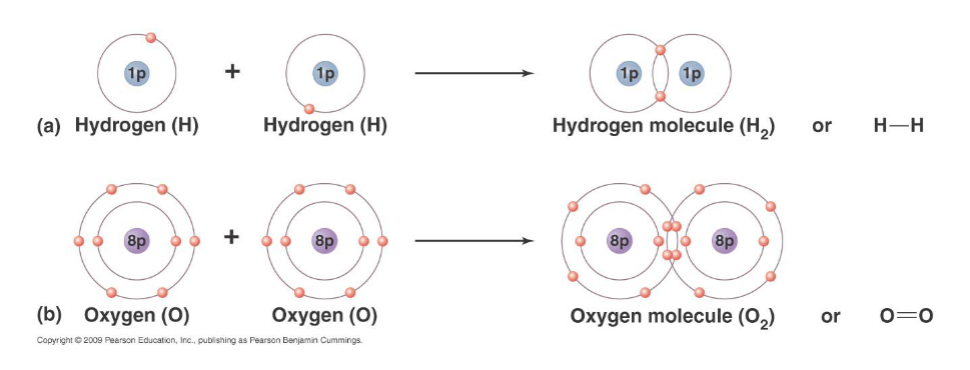
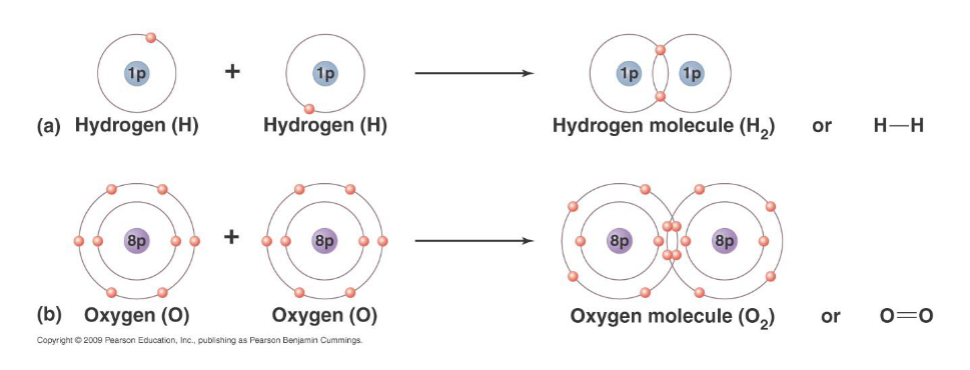
covalent bond
sharing a pair of electrons by two atoms
electronegativity
- attraction of atom for electrons
- the more electronegative an atom, the greater the pull its nucleus exerts on electrons
SPONCH
252341
What is the preferred # of covalent bonds of common biochemicals?
252341 (SPONCH)
How many covalent bonds does sulphur (S) have?
2
How many covalent bonds does phosphorus (P) have?
5
How many covalent bonds does oxygen (O) have?
2
How many covalent bonds does nitrogen (N) have?
3
How many covalent bonds does carbon (C) have?
4
How many covalent bonds does hydrogen (H) have?
1
nonpolar covalent bonds
- shared electrons spend equal amounts of time around each nucleus
- atoms with similar electronegatives
- no poles exist
Why are carbon atoms critical to life?
carbon atoms form 4 nonpolar covalent bonds with other atoms
Organic compounds contain ___ .
carbon and hydrogen atoms
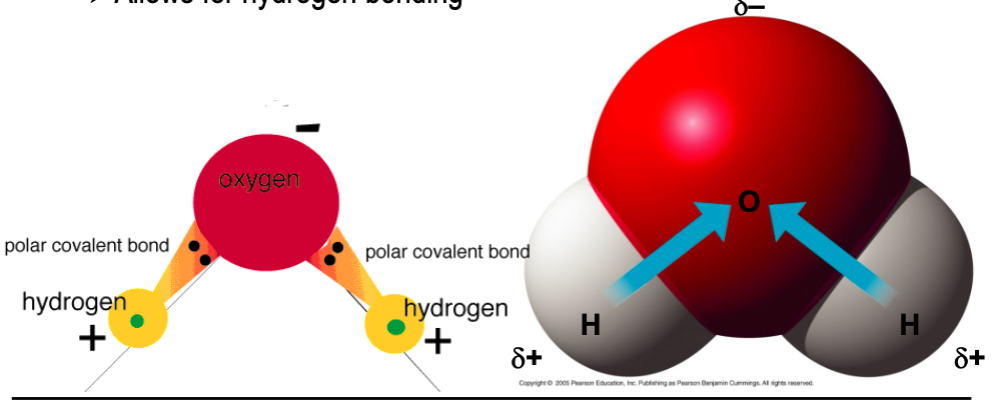
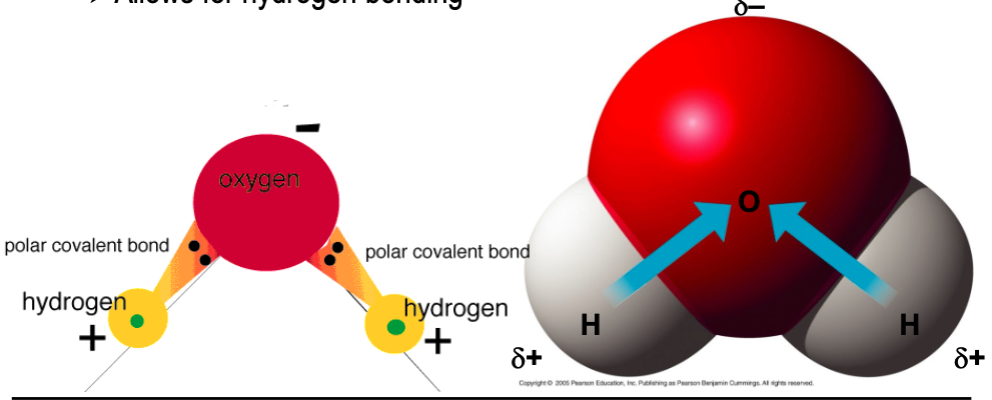
polar covalent bonds
- unequal sharing of electrons due to significantly different electronegativities
- most important polar covalent bonds involved hydrogen
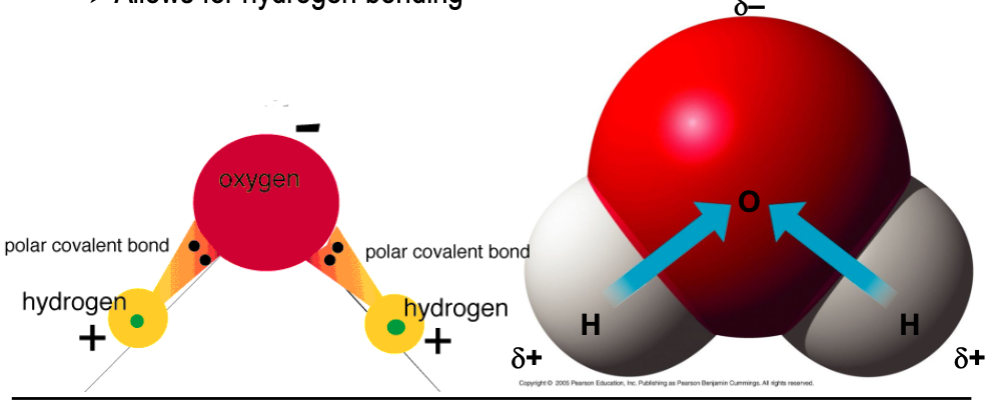
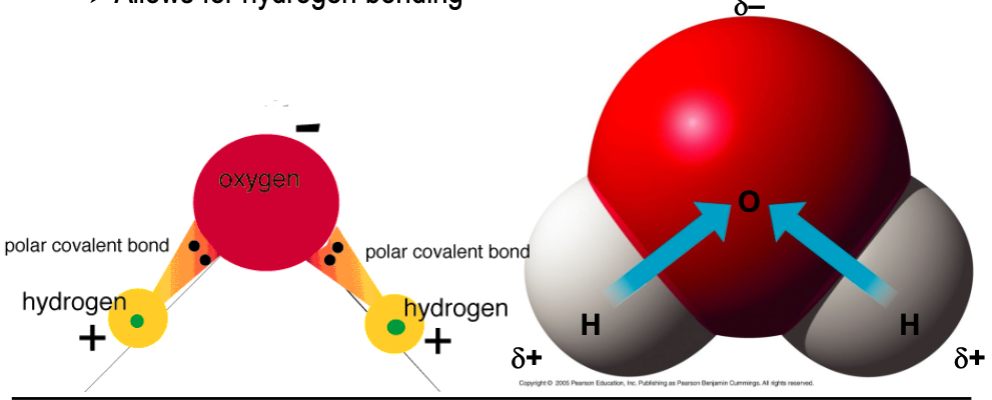
Why do the most important polar covalent bonds involve hydrogen?
they allow for hydrogen bonding
ionic bonds
- occur when 2 atoms with vastly different electronegativities come together
- atoms have either positive (cation) or negative (anion) charges
- cations and anions attract each other and form ionic bonds (no electrons shared)
- typically form crystalline ionic compounds known as salts
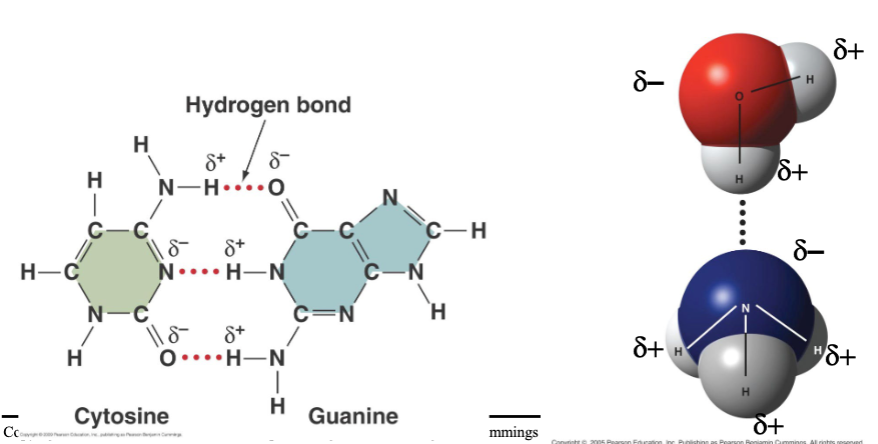
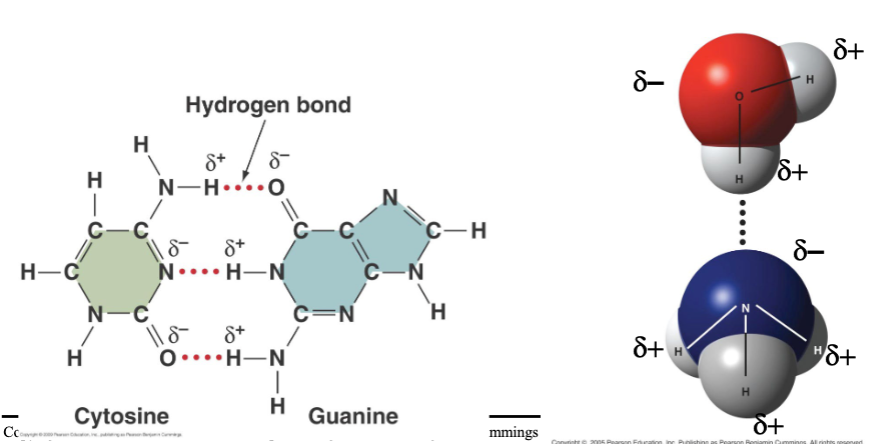
hydrogen bonds
- found when polar covalent bonds present
- electrical attraction between partially charged H+ and full of partial negative charge on different region of same molecule or another molecule
- weaker than covalent bonds but essential for life
- many help stabilize 3D shapes of large molecules
Describe the strength of a non-polar covalent bond.
strong
Describe the strength of a polar covalent bond.
strong
Describe the strength of an ionic bond.
weak (in water)
Describe the strength of a hydrogen bond.
weak
water
- most abundant substance in organisms
- most of its special characteristics due to two polar covalent bonds
Describe the special characteristics when water is formed by two polar covalent bonds.
- cohesive molecules (surface tension)
- excellent solvent
- remains liquid across wide range of temperatures
- can absorb significant amounts of energy without changing temperature
- participates in many chemical reactions
acids and bases
dissociated by water into component cations and anions
acid
dissociates into one or more H+ and one or more anions
base
- binds with H+ when dissolved into water
- some dissociate into cations and OH-
Metabolism requires ___ .
relatively constant balance of acids and bases
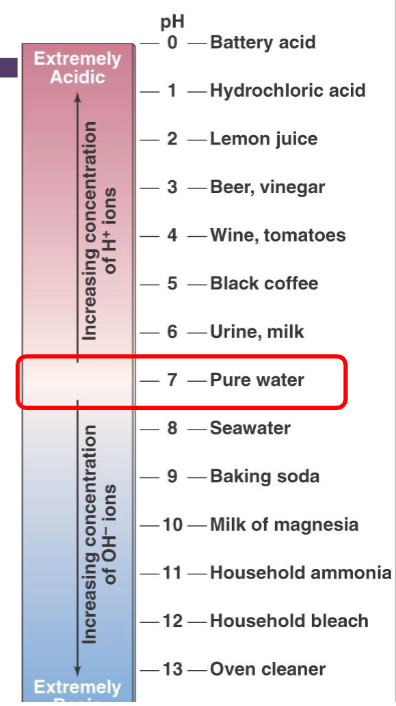
Concentration of H+ in solution is expressed using ___ .
the pH scale
![<p>At pH 7 [OH-] = ___ </p>](https://knowt-user-attachments.s3.amazonaws.com/81788658-69c0-4e7f-b781-139b960a28fa.png)
At pH 7 [OH-] = ___
[H+]
![<p>[H+]</p>](https://knowt-user-attachments.s3.amazonaws.com/29e928d5-2dbc-40ac-8629-b603ef4ee977.png)
![<p>At pH 7 [OH-] = [H+] for every move down in value, the H+ concentration ___ .</p>](https://knowt-user-attachments.s3.amazonaws.com/5f4de137-595d-4b96-b602-2fc12da148d9.png)
At pH 7 [OH-] = [H+] for every move down in value, the H+ concentration ___ .
increases 10 times
Microorganisms differ in __ .
their tolerance of various pH ranges
How do microorganisms differ in their tolerance of various pH ranges?
- most are neutrophiles
- some microbes can change the pH of their environment
Bacillus
endosphere reducing bacteria
reactions
new arrangements ofo atoms (molecules)
Biochemistry involves ___ .
chemical reactions of living things (reactants, substrates, and products)
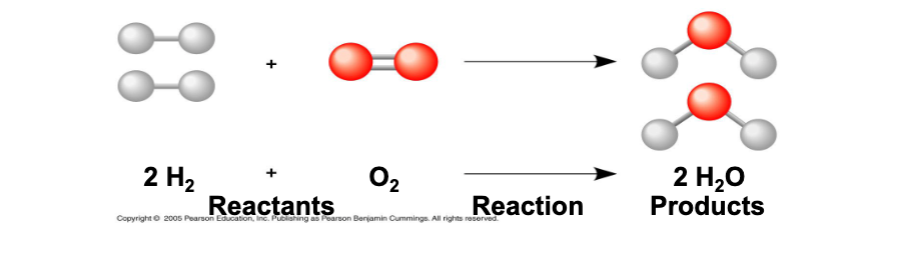
synthesis reactions
- involved the formation of larger more complex molecules
- require energy (endothermic)
- most common type: dehydration synthesis
anabolism
all synthesis reactions in an organism
decomposition reactions
- break bonds within larger molecules to form smaller atoms, ions, and molecules
- release energy (exothermic)
- most common type: hydrolysis
dehydration synthesis
where water molecules are formed
hydrolysis
ionic compounds of water are added to products
catabolism
all decomposition reactions in an organism
exchange reactions
- involve breaking and forming covalent bonds
- involve endothermic and exothermic steps
- involve atoms moving from one molecule to another
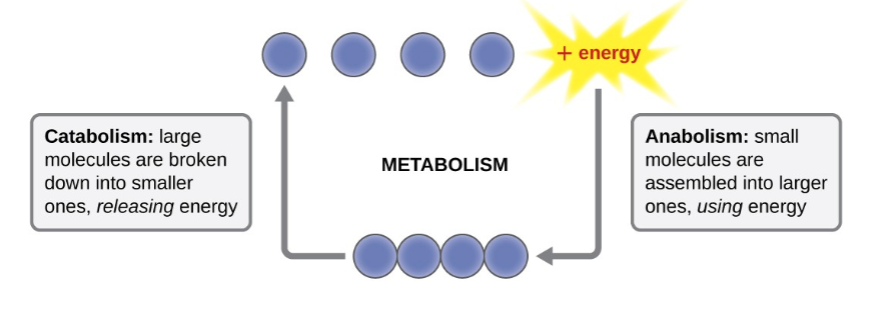
metabolism
sum of all chemical reactions in an organism
Functional Groups and Monomers of Organic Macromolecules
- contain carbon and hydrogen atoms
- atoms often appear in certain common arrangements (functional groups)
- macromolecules
- monomers
macromolecules
large molecules used by all organisms
List the macromolecules found in functional groups.
lipids, carbohydrates, proteins, nucleic acids
monomers
basic building blocks of macromolecules
The hydroxyl structure may be written as __ .
-OH or HO-
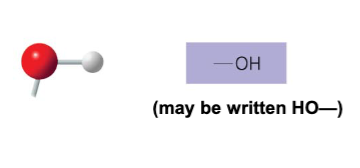
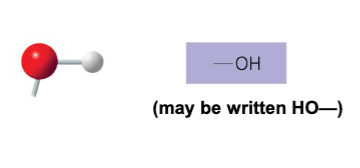
Name of compounds for hydroxyl.
alcohols
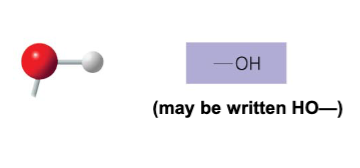
Describe the functional properties of hydroxyl.
- is polar as a result of electronegative oxygen atom drawing electrons toward itself
- attracts water molecules which helps dissolve organic compounds such as sugars
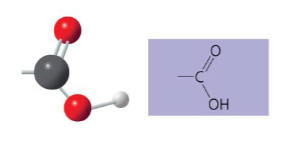
The carboxyl structure may be written as __ .
-COOH
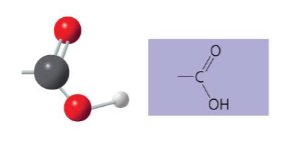
Name of compounds for carboxyl.
carboxylic acids or organic acids
Describe the functional properties of carboxyl.
- has acidic properties due to its source of hydrogen ions
- covalent bond between oxygen and hydrogen is so polar that hydrogen ions (H+) tend too dissociate reversibly
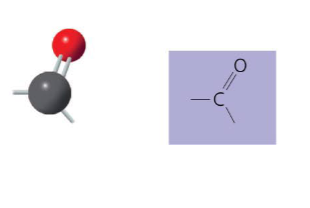
The carbonyl structure may be written as __ .
C=O
Name of compounds for carbonyl.
- ketones: if carbonyl group is within a carbon skeleton
- aldehydes: if carbonyl group is at the end of carbon skeleton
Provide an example of a hydroxyl.
ethanol: alcohol that’s present in alcoholic beverages
Describe an example of carboxyl.
acetic acid: gives vinegar its sour taste
Describe an example of carbonyl.
- acetone: simplest keton
- propanal: an aldehyde