IB BIO HL 1- Unit 4: Proteins and Enzymes.
1/75
Earn XP
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
76 Terms
Denaturation
-changing of a protein's 3D shape
-irreversible
-pH changes denatures proteins
-heat denatures proteins
-Animal proteins are stable at body temp
-Plant proteins are stable at air temp
Tertiary structure
The way polypeptides fold and coil to form a complex molecular shape
-caused by interactions between R-groups
-tertiary structures may be important for the function of the enzyme
quaternary structure
The interaction between multiple polypeptides or R-groups ressult in a single/larger, biologically active protein
-quaternary structure may be held by a variety of bonds
-glycoproteins have carbohydrates attached
Secondary structure
-the way the chains of amino acids fold or turn upon themselves
-held together by hydrogen bonds between non-adjacent amine (N-H) and carboxyl (C-O) groups.
-may form an alpha helix or beta pleated sheet or a random coil
-provides a little bit of structural stability (due to H-bonds)
Amino acid structure
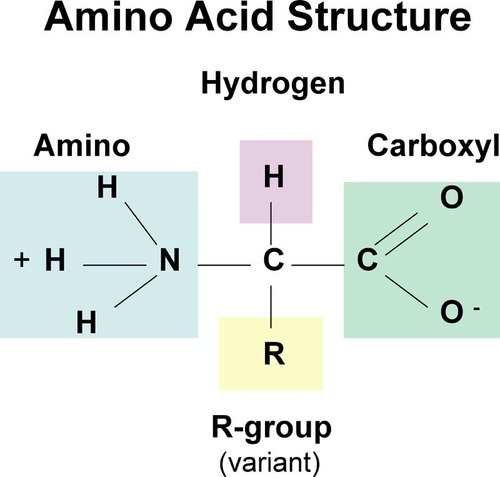
conjugated proteins
-a protein to which another chemical group is attach by either covalent bonding or other interactions
Collagen + Actin and myosin
Collagen: A long fibrous protein that forms fibers and run through tissue, it makes your skin tight and bones strong.
Actin + Myosin: long fibrous protein that form cellular cytoskeleton and muscle fibers. Actin is pulled by myosin to contract a muscle.
Enzymes + Insulin
Enzymes: globular protein with a specific 3D shape that can bind to other molecules at a specific active site. They can change shape to catalyse reactions.
Insulin: globular protein that fits into a receptor that is coubled with a glucose channel in the cell membrane
Two types of amino acids
essential: must be consumed by diet (9)
Non-essential: can be synthesized in your body (11)
globular
-round, spherical
-functional (transport, enzymes)
-soluble
-irregular sequences
-very heat sensitive
-hydrophobic r-groups are folded on the inside
-can be conjugated
-Quaternary structure more likely
primary structure
the order/sequence of the amino acids of which the protein is composed
-formed by covalent peptide bonds between adjacent amino acids
-controls all subsequent levels of structure because it determines the nature of the interactions between r-groups of different amino acids
fibrous
-long
-narrow
-strength + supports your cell
-insoluble
-repetitive amino acid sequences
-less heat sensitivity
-hydrophobic r-groups that are exposed
-may have a Quaternary structure
-can be conjugated
Integral membrane proteins
-non-polar amino acids are closest to the lipids (embedded within the lipid bilayer
Amino acid characteristics
-20 amino acids
-acidic or basic
-ringed structure usually means non-polar
non-conjugated proteins
are purely protein in their Quaternary structure
protein chains
-Dna code is variable (can be infinite # of combinations)
-shortest polypeptide is 11 amino acids long
-longest (tintin) is over 35,000 amino acids longs
Enzyme
-globular protein
-increases reaction rate
-lowers the activation energy
-biological catalyst
active site
-is the location on an enzyme where the substrate fits perfectly (lock and key)
"how do enzymes lower the activation energy of reaction?"
-Enzymes bring substrate(s)into the active site. This positions substrates in a way that they can chemically change.
-Due to the binding, the bonds in the substrate molecule are less stable. Unstable bonds are easier to change.
-The binding lowers the overall energy level needed for the substrate to change into the product.
-This is known as activation energy which is therefore reduced.
Examples of anabolism
-anabolism includes the formation of large carbohydrates like the glycogen and large proteins.
-the reactions of photosynthesis are anabolic in total as they build up a carbohydrate
examples of catabolism
-All digestion reactions of macromolecules are catabolic as they break down a carbohydrate.
-the cellular respiration of macromolecules are catabolic, are they break down larger molecules.
changes in shape
both substrate and enzymes change when binding occurs
Enzyme catalysis and molecular motion
Most enzyme reactions occur when the substrates are dissolved in water.
All dissolved molecules are in random motion each moving separately.
The coming together of a substrate molecule and an active site is known as a collision.
Collisions are the result of random movements of both substrate and enzyme.
If not immobilized, the enzyme can move too.
Enzymes tend be larger than the substrate(s) and therefore move more slowly.
The substrate may be at any angle to the active site when the collision occurs.
Successful collisions are when the substrate and active site happen to align and bind.
Enzyme Kinetics and Molecular Collisions
Immobilization of Enzymes
enzymes physically confined or localized in a certain defined region of space with retention of their catalytic activities, and which can be used repeatedly and continuously
Ex: Production of Lactose Free Milk by Immobilized Lactase Enzyme:
Enzyme can be reused
Product is enzyme free
Enzyme is more stable and long-lasting when adhered to an inert matrix like alginate.
Denaturing enzymes
For enzymes a change in structure means a change in the active site. If the active site changes shape the substrate is no longer able to bind to it.
Factors affecting the rate of enzyme activity
Temperature, pH and substrate concentration can all affect the rate of activity of enzymes. Above are sketch graphs showing how each factor affects enzyme activity.
The effect of temperature on the rate of enzyme activity
Low temperatures result in insufficient thermal energy for the activation of a given reaction.
Increasing the temperature increases the speed and motion of enzyme and substrate, resulting in higher enzyme activity
A higher kinetic energy will result in more frequent collisions between enzyme and substrate
At an optimal temperature (varies per enzymes), the rate of enzyme activity will be at its peak
Higher temperatures destabilize enzyme as thermal energy breaks hydrogen bonds within the enzyme.
The enzyme (particularly the active site) loses its shape, resulting in a loss of enzyme activity (denaturation)
The effect of pH on the rate of enzyme activity
Changing the pH will alter the charge of the enzyme, possibly changing protein solubility shape of the molecule
Changing shape or charge of the active site will diminish its ability to bind to the substrate, halting enzyme function
Enzymes have an optimum pH and moving outside of this range will always result in a diminished rate of reaction
Different enzymes may have a different optimum pH ranges
The effect of substrate concentration
on the rate of enzyme activity (graph)
Measurements in enzyme-catalyzed reactions.
Glycolysis:
Intracellular path
Linear pathway
Each step is enzyme catalyzed
Krebs Cycle
Intracellular path
Cyclic pathway
Each step is enzyme catalyzed
Calvin Cycle
Intracellular path
Cyclic Path
Each step is enzyme catalyzed
Digestion
Extracellular path
Each process in the digestive system is enzyme catalyzed
Heat Generation
Metabolic reactions are not 100% efficient in energy transfer; some is lost as heat.
Mammals, birds, and some other animals depend on the heat to maintain constant body temperature
Allosteric Sites & Non-Competitive Inhibition
Site other than active site.
Site is specific
Inhibitors bind to allosteric site
Binding causes conformational changes, altering active site
Non-competitive because inhibitor is not specific for active site
Catalysis is prevented
Binding is reversible
Allosteric Sites & Non-Competitive Inhibition: Antabuse (disulfiram)
Used to treat alcoholism
Drug allosterically binds to aldehyde dehydrogenase
Acetaldehyde is not broken down into acetate so acetaldehyde builds us and causes extreme symptoms
Competitive Inhibition
Inhibitor competes with substrate to bind to active site
Competitive Inhibition: Statins
Drugs that inhibit cholesterol synthesis
Binds to active site of enzyme preventing conversion to cholesterol.
Inhibition Graphs
Competitive:
Requires more substrate to reach maximum enzyme activity
Non-competitive:
Increasing substrate concentration has no effect
Maximum enzyme activity reduced
Feedback Inhibition:
Feedback Inhibition: Threonine conversion to Isoleucine
End product builds up and eventually inhibits enzyme #1.
Self-regulatory
Mechanism Based Inhibition:
Inhibitor (orange) binds to the active site as a substrate. (yellow is actual substrate)
Forms stable intermediate–enzyme complexes or reactive electrophilic species
Can lock up or destroy the enzyme
New enzyme synthesis is required to restore the enzymatic activity.
Mechanism Based Inhibition: Penicillin
Penicillin inhibits transpeptidase weakening bacterial cell walls
A mutational change in transpeptidase would result in penicillin resistance
Dipeptide
A molecule made up of two amino acids joined together by a peptide bond
Oligopeptide
A molecule consisting of a few amino acids, typically between 2 and 20, linked by peptide bonds.
Polypeptide
A long chain of many amino acids (more than 20) connected by peptide bonds. Polypeptides are the building blocks of proteins.
State where in the cell polypeptide formation occurs
Polypeptide formation occurs in the ribosomes,
Compare the source of amino acids by plant and animal cells.
Plants synthesize amino acids via photosynthesis. In contrast, animals obtain amino acids by digesting dietary proteins into their building blocks. Unlike plants, animals rely on essential amino acids from food since they cannot produce them all.
Outline why vegan diets require attention to food combinations to ensure essential amino acids are consumed.
Vegan diets require careful attention because plant-based protein sources often lack one or more essential amino acids, making them incomplete proteins (e.g., beans lack methionine, while rice lacks lysine). By combining complementary proteins, such as beans and rice, vegans can ensure they consume all the essential amino acids necessary for protein synthesis and overall health
Outline the effect of R-group structure on the properties of an amino acid.
The R-group, or side chain, is the part of an amino acid that varies between different amino acids. It determines the amino acid's properties, such as whether it is polar, nonpolar, or charged. This affects how the amino acid interacts with water and other molecules, influencing protein structure and function. For example, hydrophobic R-groups tend to cluster inside proteins, while hydrophilic ones are on the outside.
Identify the “backbone” of a polypeptide.
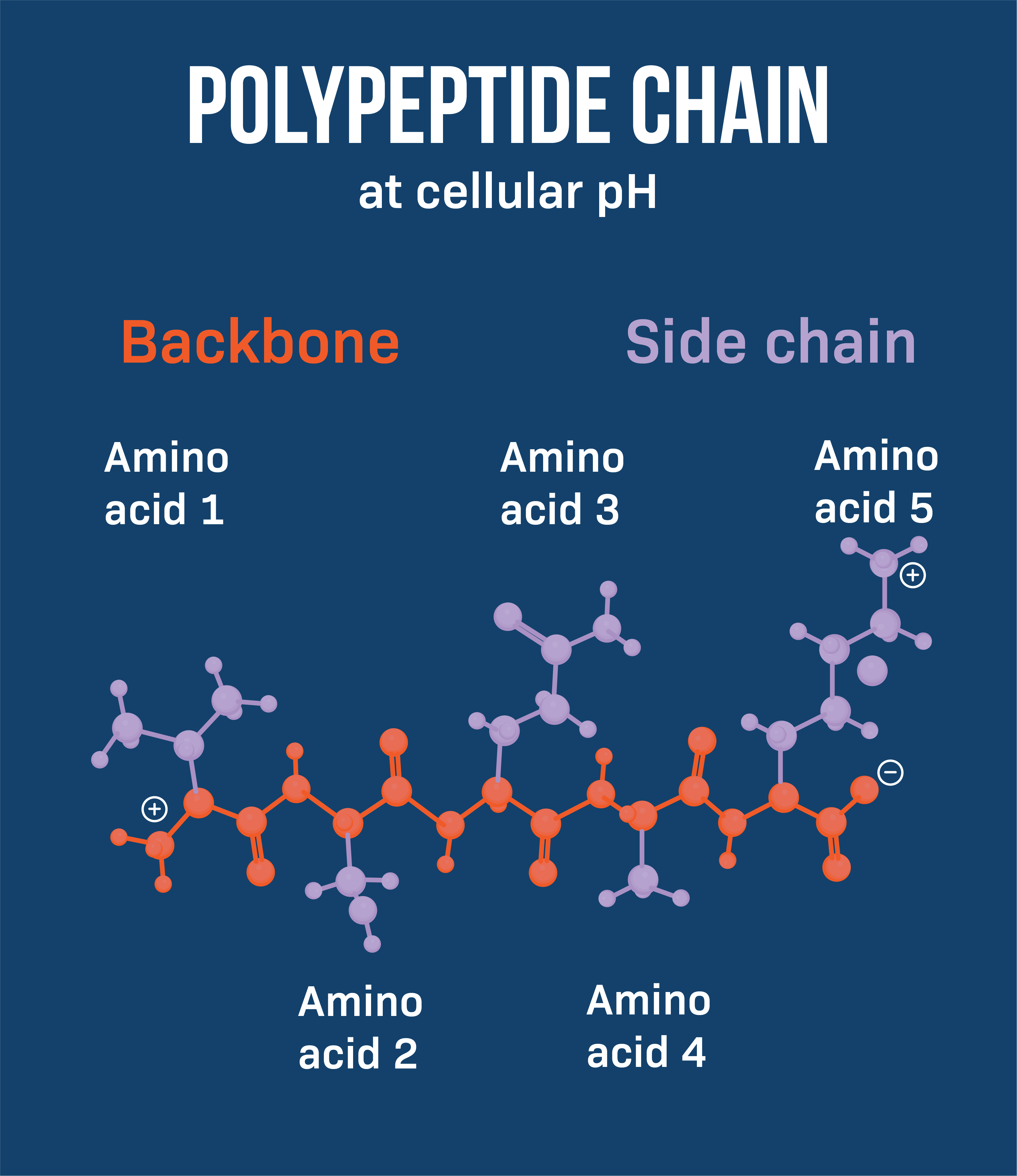
Define “conformation” as related to protein structure
Conformation refers to the specific three-dimensional shape or structure of a protein, determined by the folding and arrangement of its polypeptide chains. This shape is essential for the protein's function and is influenced by interactions such as hydrogen bonds, ionic bonds, hydrophobic interactions, and disulfide bridges.
Outline how a DNA sequence codes for a polypeptide.
A DNA sequence codes for a polypeptide by first being transcribed into mRNA in the nucleus. The mRNA then travels to the ribosome, where its codons are read to match specific tRNAs carrying amino acids. These amino acids are joined together in the correct order to form the polypeptide.
Explain the effects of polar and non-polar R-groups.
Polar R-groups are hydrophilic and attract water, so they are usually found on the outside of proteins where they can interact with water. Non-polar R-groups are hydrophobic and avoid water, clustering on the inside of proteins to help stabilize their structure. These interactions influence how proteins fold and function.
amino acid arrangements in soluble globular proteins
These proteins fold into a compact shape with hydrophilic amino acids on the outside, interacting with water, and hydrophobic amino acids tucked inside, away from water.
amino acid arrangements in integral membrane proteins
These proteins span the membrane, with hydrophobic amino acids located in the regions embedded in the lipid bilayer and hydrophilic amino acids exposed on the surfaces facing the aqueous environments.
amino acid arrangements in channel proteins
These form pores through membranes, with hydrophobic amino acids on the exterior to interact with the lipid bilayer, while hydrophilic amino acids line the interior to allow the passage of water or polar molecules.
Define catalyst
A catalyst is a substance that speeds up a chemical reaction by lowering the activation energy required for the reaction to occur, without being consumed or permanently altered in the process.
Define metabolism.
Metabolism is the sum of all chemical reactions that occur within an organism to maintain life, including catabolic reactions that break down molecules to release energy and anabolic reactions that build molecules using energy.
Define specificity in relation to enzyme structure and function.
Specificity refers to an enzyme's ability to catalyze only one specific reaction or act on a specific substrate. This is due to the unique shape of the enzyme's active site, which fits only particular substrate molecules, like a "lock and key."
Control of metabolism by enzymes.
Enzymes control metabolism by speeding up chemical reactions. They ensure reactions happen only when needed by being activated or inhibited. Feedback inhibition prevents overproduction by turning off enzymes when enough product is made. Enzymes are also found in specific cell locations to make processes more efficient.
Variation in specificity.
Variation in enzyme specificity refers to the differences in how enzymes interact with substrates:
Absolute Specificity: Some enzymes, like sucrase, act on only one specific substrate (e.g., sucrose).
Group Specificity: Enzymes like lipase act on a group of related molecules, such as all triglycerides.
Linkage Specificity: Enzymes like amylase target specific bonds (e.g., glycosidic bonds) within a molecule.
Broad Specificity: Some enzymes, like proteases, act on a wide variety of substrates with common features, such as peptide bonds.
Manipulated, responding, and controlled variables.
Manipulated Variable: The variable that is intentionally changed or controlled by the experimenter to observe its effect (also called the independent variable).
Example: The temperature in an enzyme activity experiment.Responding Variable: The variable that is observed and measured as it changes in response to the manipulated variable (also called the dependent variable).
Example: The rate of enzyme activity.Controlled Variables: The variables that are kept constant throughout the experiment to ensure that any changes in the responding variable are only due to the manipulated variable.
Example: The pH and concentration of the enzyme solution.
Units and methods for reaction rates.
Reaction rates are measured as the change in concentration of reactants or products over time, with units like mol/L/s or mg/min. Methods include measuring concentration changes using spectrophotometry or titration. Gas production can be tracked with a gas syringe, while color changes are monitored using colorimetry. Other methods include measuring pH changes with a pH meter or mass loss during the reaction.
Investigative techniques to analyze biological processes
Biologists use a variety of investigative techniques to study living organisms. Microscopy helps observe cells and tissues, while spectrophotometry measures light absorption to analyze concentrations. Techniques like chromatography and gel electrophoresis separate mixtures or biomolecules for detailed analysis. Field studies allow scientists to observe organisms in their natural habitats, complementing laboratory techniques.
Intracellular enzymes
-Function inside cells to regulate metabolic processes (e.g., DNA polymerase for DNA replication).
-Involved in cell maintenance, energy production, and synthesis of macromolecules.
extracellular enzymes
Function outside cells, often secreted to break down large molecules (e.g., amylase in saliva for starch digestion).
Break down substrates for nutrient absorption or other external functions.
Examples of intracellular: Catalase
breaks down hydrogen peroxide within cells
example for extracellular: trypsin
digests proteins in the small intestine
Reason for metabolic pathways.
Metabolic pathways exist to efficiently control and coordinate chemical reactions in cells. They allow for the gradual release or use of energy in small, manageable steps, preventing harmful energy spikes or losses. Each step is catalyzed by a specific enzyme, ensuring precision and regulation through feedback mechanisms. Pathways also enable the cell to produce a variety of molecules from a common set of precursors, ensuring adaptability and efficiency.