Chemistry Exam 2
1/29
Earn XP
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
The first law of thermodynamics
a. energy is neither created nor destroyed during a change in a system.
b. cannot be used to predict the direction of a reaction.
c. applies to all defined systems in the universe.
d. a, b, and c are correct.
d. a, b, and c are correct.
Entropy is conveniently defined as
a. the amount of kinetic energy that can be utilized by a system.
b. the amount of work done on a system by the surroundings.
c. the amount of randomness or freedom each component has in a system.
d. the way a system responds when heat flows out of it to the surroundings.
c. the amount of randomness or freedom each component has in a system.
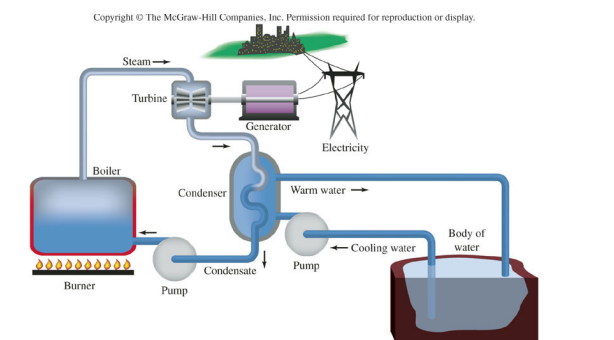
In the following diagram of a power plant, the kinetic energy in the steam is directly converted to
a. electrical energy in the generator.
b. mechanical energy in the turbine.
c. potential energy in the turbine.
d. kinetic energy in the water.
b. mechanical energy in the turbine.
A power plant cannot convert 100% of the energy stored in the fuel to electricity because
a. energy conversions include some loss of heat to the surroundings because compartments cannot be completely insulated.
b. conversion of potential energy to mechanical energy results in loss of entropy.
c. a and b are both incorrect.
d. a and b are both correct.
d. a and b are both correct.
The combustion of methane gas is given by
CH4 (g) + 2O2 (g) ® CO2 (g) + 2H2O (g)
Much heat is given off during this reaction. The reaction is
a. exothermic.
b. endothermic .
c. neither.
d. it cannot be determined.
a. exothermic.
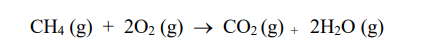
The reaction in question 5 requires breaking the bonds that hold one mole of methane. The amount of energy required to do so is
a. 416 kJ.
b. 832 kJ.
c. 1,248 kJ.
d. 1,664 kJ.
d. 1,664 kJ.
The reaction in question 5 yields one mole of carbon dioxide. The amount of energy that is given off by the formation of the bonds in one mole of CO2 is
a. 803 kJ.
b. 1,606 kJ.
c. 2,409 kJ.
d. 3,212 kJ.
b. 1,606 kJ.
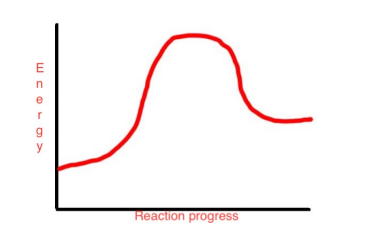
This reaction progress diagram describes a change that is
a. exothermic
b. endothermic
c. isothermic.
d. a, b, and c are correct.
b. endothermic
In the reaction A + B ® X + Y, reactants A+B contain 560 kJ, and products X+Y contain 210 kJ. The reaction is
a. exothermic
b. endothermic
c. isothermic
d. a, b, and c are correct.
a. exothermic
As molecules undergo changes during a reaction, bonds between atoms in reactant molecules must be broken in order to allow the atoms to rearrange into product molecules. The process of breaking bonds in the reactants is
a. exothermic
b. endothermic
c. isothermic.
d. a, b, and c are correct.
b. endothermic
During distillation, heat is applied to a sample and the vapors ascend up a column. Short hydrocarbon will condense (go from the gas to the liquid phase)
a. at the top of the column because the short molecules remain in the gas phase at the hot temperature near the bottom of the column, but condense at the cooler temperatures near the top.
b. at the middle of the column because the steam interferes with the attractive forces between molecules.
c. at the bottom of the column because that is where the attractive forces between molecules are greatest.
d. at the bottom of the column because the attractive forces between molecules increase as the molecules become shorter.
a. at the top of the column because the short molecules remain in the gas phase at the hot temperature near the bottom of the column, but condense at the cooler temperatures near the top.
Fuel cracking is
a. a process used to create diesel from tar.
b. a process used to increase the price of gasoline.
c. a process used to generate diesel from food industry waste.
d. a process used to generate gasoline from diesel.
d. a process used to generate gasoline from diesel.
Consider hydrocarbons that contain only single bonds. The energy content per molecule
a. increases as the length of the hydrocarbon increases.
b. decreases as the length of the hydrocarbon increases.
c. stays the same irrespective of the length of the hydrocarbon.
d. cannot be predicted from the length of the hydrocarbon.
a. increases as the length of the hydrocarbon increases.
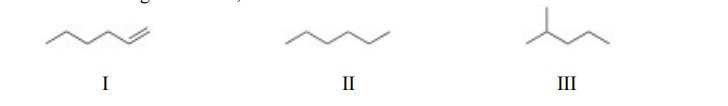
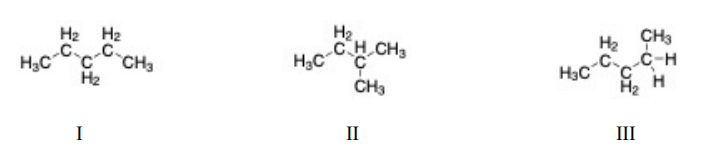
Of the following molecules, the structural isomers are I II III
a. I and II.
b. I and III.
c. II and III.
d. I, II, and III.
c. II and III.
Of the following molecules, the structural isomers are I II III
a. I and II.
b. I and III.
c. I, II and III.
d. none
a. I and II.
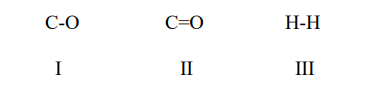
A polar bond is shown in
C-O C=O H-H
I II III
a. I and II
b. I and III '
c. I, II, and III
d. II and III
a. I and II
The bond polarity of N-H, in comparison to that of C-H, is
a. greater
b. identical
c. lower
d. always changing
a. greater
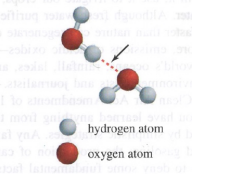
The arrow in the drawing points to a(n)
a. ionic bond
b. covalent bond
c. hydrogen bond
d. hydrophobic interaction
c. hydrogen bond
Water has a
a. lower boiling point than predicted by its molar mass due to the covalent O-H bonds.
b. higher boiling point than predicted by its molar mass due to the covalent O-H bonds.
c. lower boiling point than predicted by its molar mass due to hydrogen bonds.
d. higher boiling point than predicted by its molar mass due to hydrogen bonds.
d. higher boiling point than predicted by its molar mass due to hydrogen bonds.
A solution of potassium chloride (KCI) in water is predicted to
a. conduct electricity.
b. have only one type of ion.
c. be a poor electrical conductor.
d. contain solute molecules.
a. conduct electricity.
Water that has been cooled to 0-4°C
a. has more covalent bonds than 5-20°C water, making it less dense.
b. is denser than 5-20°C water, therefore it floats to the top of the warmer water below it.
c. has fewer hydrogen bonds than 5-20°C water, and this affects the density.
d. is less dense than 5-20°C water, therefore it floats on top of the warmer water below it.
d. is less dense than 5-20°C water, therefore it floats on top of the warmer water below it.
A solution of KOH is predicted to
a. exhibit high electrical resistance.
b. have a basic pH.
c. neutralize basic solutions.
d. a, b, and c are correct.
b. have a basic pH.
Oil and vinegar salad dressing, which has a pH of 5.8, is classified as
a. a slightly basic solution
b. an acidic solution
c. a poor electrolyte
d. a molecular solution
b. an acidic solution
Mixing hydrochloric acid (HCI) with water, would result in
a. emitted chlorine gas
b. an insoluble mixture
c. a solution of pH above 7.0
d. an increase in the concentration of protons
d. an increase in the concentration of protons
The amount of protons in a 2 Liter soda bottle is 0.00542 moles. The concentration of protons in this bottle is
a. 0.00108 M
b. 0.0302 M
c. 0.00271 M
d. 0.00542 M
c. 0.00271 M
The amount of protons in a 1L bottle of soda is 2.683×10^-3 moles. The pH of the soda is
a. 1.7
b. 1.9
c. 2.2
d. 2.6
d. 2.6
A solution of the strong base KOH has a concentration of 0.0083 M. The pOH of this solution is
a. 1.8
b. 1.9
c. 2.1
d. 6.5
c. 2.1
The pOH of a NaOH solution is 4.7. The pH of this solution is
a. 9.3
b. 8.6
c. 5.5
d. 4.7
a. 9.3
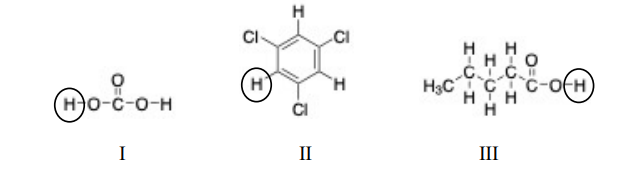
In the molecules below, an acidic proton is indicated in
a. I and II
b. I and III
c. I, II, and III
d. II and III
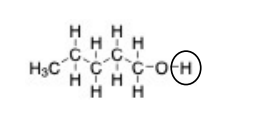
In the molecule shown, the indicated proton is
a. non-acidic because the O-H bond is non-polar.
b. non-acidic because the O-H is connected to a carbon that is only connected to C and H atoms.
c. acidic because the O-H bond is non-polar.
d. acidic because the O-H bond is ionic.
b. non-acidic because the O-H is connected to a carbon that is only connected to C and H atoms.