ELECTRICITY STATE OF MATTER TEST
1/226
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
227 Terms
Electrostatics
The branch of physics that deals with stationary or resting electric charges.
Electrodynamics
the branch of physics that deals with moving charges. (to be discussed after electrostatics)
Why only electrons for electricity?
Protons are tightly bound to the nucleus
Remember electron binding energy?
Difficult to remove
Electrons can move much more easily
Electrification
Describes the process of electron charges being added to or subtracted from an object
Difference between ionization?
Ionization - atom
Electrification - object
Laws of electrostatics!!!! Quiz question !!!


Repulsion-Attraction
Inverse square law
Distribution
Concentration
Movement
Remember
Ideas
During
Concentrated
Moments
Laws of Electrostatics
1st law –
repulsion-attraction
opposites attract
Laws of Electrostatics 2nd law
Inverse square law
Force between two charges directly proportional to product of their magnitudes and inversely proportional to square of distance between them

If an object has a charge of 4 coulombs at 4mm, what will the charge be at 8mm?
4 coulombs = (8mm)2
x (4mm)2
4 coulombs = 64
x 16
64x = 64
x = 1 coulomb
Laws of Electrostatics
3rd law
Distribution
Charges reside on external surface of conductors
Equally throughout insulators
Attempt to repel from each other
Laws of Electrostatics
4th law
Concentration
Greatest concentration of charges will gather at sharpest area of curvature
Why is the law of concentration of electricity important in the manufacturing process of an x-ray tube?
Sharp or rough edges could concentrate the electrons and promote discharges
Inside of tube is highly polished and rounded

Laws of Electrostatics
5th law
Movement
Only negative charges move along solid conductors
The electrons move along a solid conductor
Methods of Electrification
—quizzzzz
Friction
Contact
Induction

Method of Electrification- FRICTION
Occurs when one object is rubbed on another
E.g., rubbing balloon on wool sweater, combing hair during winter
Electrons travel from one object to another
Ideal during cold weather/low humidity
Method of Electrification - Contact
Occurs when two objects touch
Permits electrons to move from one object to another
E.g., walking across floor (electrification by friction) and touching doorknob (electrification by contact)
Method of Electrification – Contact (Static Discharge)
Static discharge – a form of contact electrification
Releases excess energy as light photons
CONTACT DOES NOT ALWAYS OCCUR BEFORE ELECTRONS NEUTRALIZE (discharge)
Occurs when:
Difference in charges = great
Distance between = small
*Tree branch artifact
*Humidifier in dark room
Method of Electrification - Induction
Used in operation of electronic devices
Process of electric fields acting on one another without contact
Considered most important because it is used in the operation of electronic devices
Electroscope
device used to demonstrate the presence of electrons
What are the five laws of electrostatics?

Repulsion-Attraction
Inverse square law
Distribution
Concentration
Movement
What are the three methods of electrification?

Friction
Contact
Induction
What is the difference between ‘static’ and ‘dynamic?
Static – such as electrostatic, relative non-motion
Dynamic – such as electrodynamic, motion
Basic Factors of Electrodynamics
Electric current
Properties of conducting materials
Electrical circuit
Electron sources
Describing Current Flow
Electric current – electrons that are moving predominantly in the same direction
Electrons move from areas of high to low concentration
Conventional current flow
Common household wiring = 2mm/sec of actual physical movement
Electricity moves at 3x108 (300,000,000) meters/sec
Almost the speed of light
Current flow – encouraging conditions
Vacuum – space from which air has been removed (few atoms to oppose electron flow)
Some gases (Ne) – promote drifting of electrons
Conductors and superconductors
Materials that easily permit electrons to flow. It has the ability to conduct electrons
Conductors
Copper
Aluminum
Superconductor
Titanium
Material’s Conductivity
Dependent on configuration of atoms:
Valence band
Conduction band
Material’s Conductivity - Length
Directly related to resistance
The longer the length, the more the resistance
Material’s Conductivity - Diameter
Inversely related to resistance
The smaller the diameter, the more the resistance
Material’s Conductivity - Temperature
Directly related to resistance
The higher the temperature, the greater the resistance
Increased atomic motion due to increased temperature prevents electrons from flowing freely
Good conductor – Sweet green tea
Short length (short distance away from me)
Large diameter (spout is large enough to fill my glass in 2 seconds flat)
Cool temperature (how else in the summer??)
Made of the right stuff – valence of +1, +2, or +3 (has to have enough sugar or it isn’t worth it)
In addition, overlapping valence band
Insulator
Material that resist the flow of electrons.
Plastic
Rubber
glass
Good insulator – doesn’t describe SGT
Long length
Small diameter
Hot temperature
Valence of -1, -2, or -3
Separation of conduction and valence bands
Conduction band is the area beyond the valence band.
Semiconductor- defintion
Conducts or resists (can depend on temperature)
+/- 4 valence
Semiconductors
Ability to conduct under certain circumstances and insulate under others
Silicon
Germanium
Carbon – compressed??? diamond
Valence electrons +/- 4
Compounds including these can also be used as semiconductors
Boron
Aluminum
Gallium
(All have a +3 valency)
Current
Flow of electrons in a circuit.
What kills
Voltage
Force that drives current through a circuit. quantity
not deadly
Deadly Current Threshold
4mA can induce ventricular fibrillation. ( irregular heart beat)
Lethal Current Level
80-100 mA can cause death.
What is a fuse?
A fuse is a thin wire enclosed in a casing that plugs into a circuit.
What happens to a fuse when a circuit is closed?
The fuse experiences the same current as any other point along the circuit.
What is the purpose of a fuse?
To disintegrate when it heats up above a certain level to protect the circuit.
What happens if the current climbs too high in a fuse?
The fuse burns up the wire, opening the circuit.
How does a fuse protect building wiring?
By opening the circuit before excess current can damage it.
How does an electromagnet in a circuit breaker prevent short circuits?
As current increases, the magnetism around the circuit increases. If the current becomes too high, the magnet pulls itself out of the circuit, opening it before damage occurs.
What is one method used by some circuit breakers to prevent short circuits?
Some circuit breakers use a small explosive charge that ignites at a lower level than a fire would start.
What happens when the explosive charge in a circuit breaker ignites?
It pushes the breaker away from the other end, opening the circuit.
What is an electrical circuit?
A pathway that permits electrons to move in a complete circle from their source.
What material is commonly used for electrical circuits?
Copper wire
What do electrical circuits allow electrons to do?
Move in a complete circle from their source, through resisting electrical devices and back to the source.
What is a series circuit?
A circuit in which all parts are connected end to end to provide a single path of current.
How does the total potential difference of a series circuit compare to parallel circuits when all other factors are the same?
Series circuits supply greater total potential difference than parallel circuits.
What happens to the total voltage in a series circuit as more resistance is added?
The total voltage increases.
What happens to total resistance in a parallel circuit as more resistors are added?
Total resistance drops.
What happens to total amperage in a parallel circuit as more resistors are added?
Total amperage increases.
What happens to total voltage in a parallel circuit when more resistors are added?
Total voltage remains the same.
What is a characteristic of parallel circuits regarding total resistance?
Parallel circuits offer less total resistance to electrical current when all other factors are the same.
What is a disadvantage of parallel circuits?
Increased amperage may cause a short circuit, which can cause a fire.
Short Circuit
Excessive current flow causing potential hazards.
Siemens (S)
A reciprocal ohm, as in 1/R. ac and dc
Total Voltage in Series
Sum of voltages across each component.
Vt = V1 + V2 + V3
Total Voltage in Parallel
Voltage remains constant across all components.
Vt = V1 = V2 = V3
Power in Series Circuit
Calculated as P=IV, where I is current.
Total Resistance in Parallel
1/Rt = 1/R1 + 1/R2 + 1/R3
Total Current in Series
It = I1 = I2 = I3
Total Current in Parallel
It = I1 + I2 + I3
Total Resistance in Series
Rt = R1 + R2 + R3
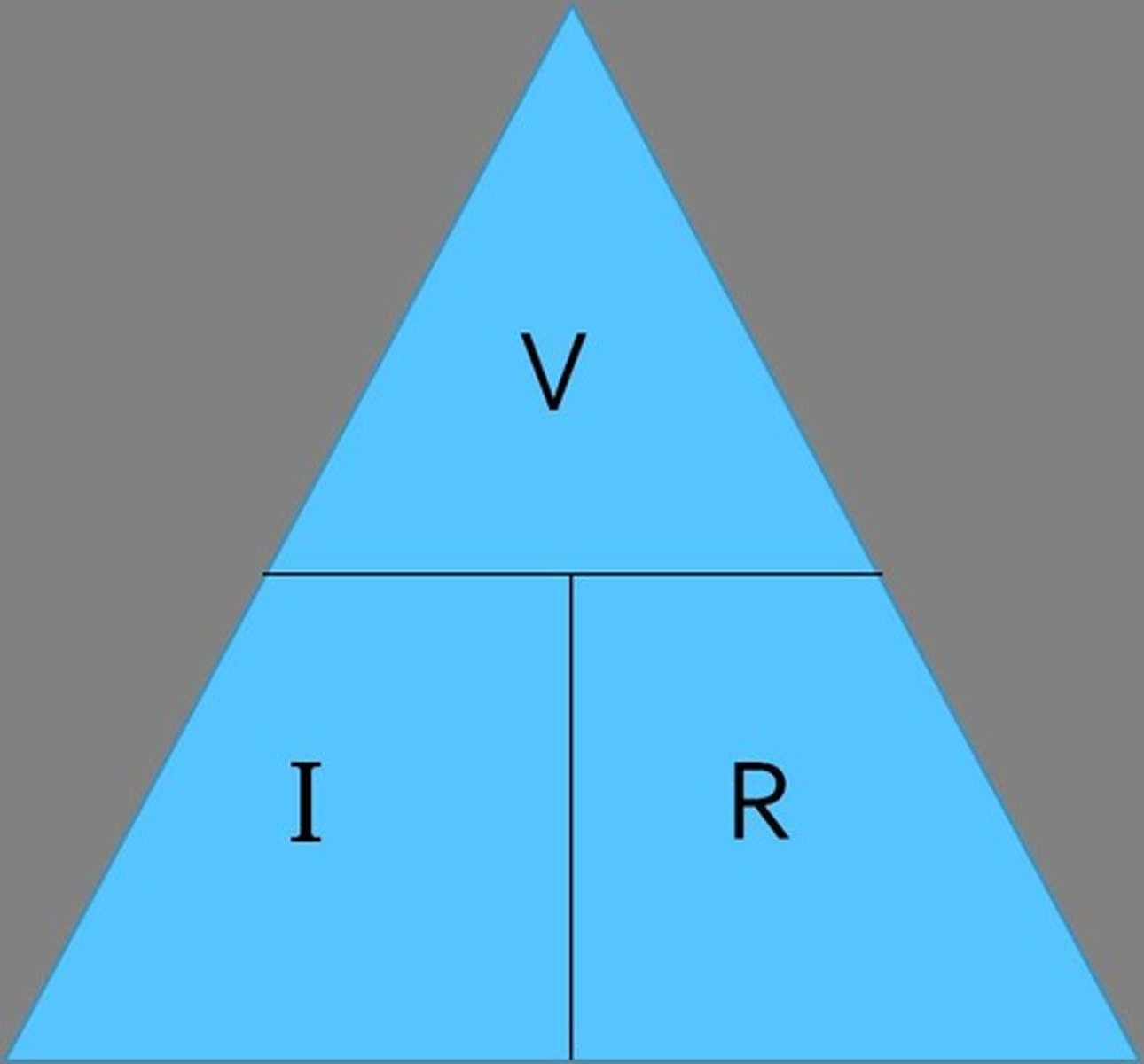
Ohm's Law
V=IR; relationship between voltage, current, resistance.
Amperage Increase in Parallel
Adding resistors increases total current.
Power Calculation
Power is the product of voltage and current.
Conductance
Reciprocal of resistance, measured in siemens.
P- V
E- I
P- R
SI
VOLTS- PLUS
(I)CURRENTAMPS-EQUALS
RESISTANCE OHMS- PLUS
SI- SERIES
E- V
P - I
I- R
PEN
VOLTS- EQUALS
I CURRENTS AMPS- PLUS
RESISTANCE- INVERSE
PARALLEL
Radiography
Art and science of imaging internal structures.
Natural Science
Study of natural phenomena and laws.
Physical Science
Study of non-living matter and energy.
Biological Science
Study of living organisms and life processes.
States of Matter
Solid, liquid, gas based on molecular attraction.
Atomic Theory
Concept explaining matter's structure and behavior.
Dalton's Theory
Elements separated by mass; foundation of periodic table.
Plum Pudding Model
JJ Thompson's model of atom with electrons in a cloud.
Rutherford's Model
Dense nucleus surrounded by negative electron cloud.
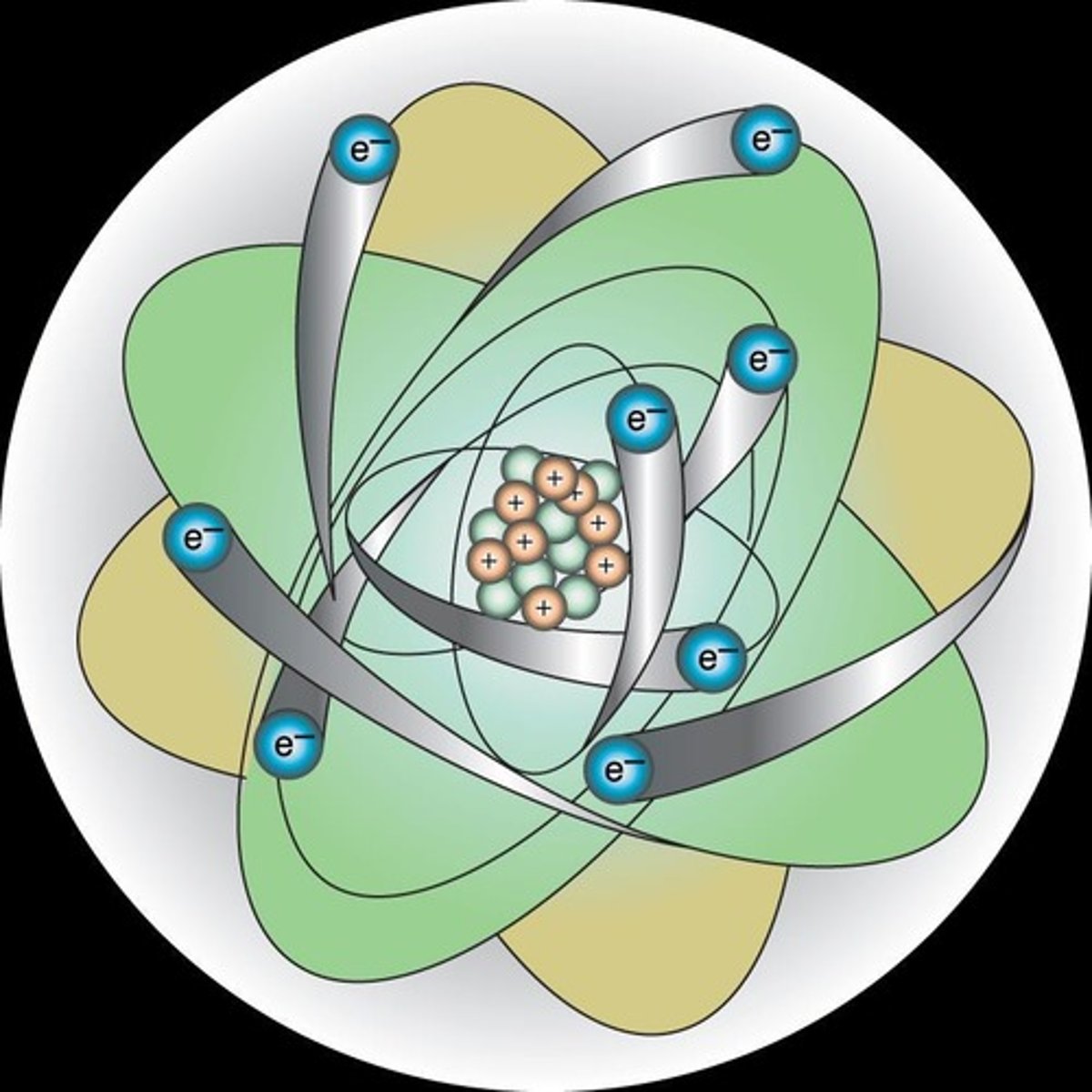
Bohr Model
Atom resembles miniature solar system with orbits.
Subatomic Particles
Protons, neutrons, and electrons make up atoms.
Proton
Positive charge particle located in the nucleus.
Neutron
Neutral particle located in the nucleus.
Electron
Negative charge particle orbiting the nucleus.
Atomic Number (Z#)
Number of protons in an atom's nucleus.
Atomic Mass Number (A)
Sum of protons and neutrons in nucleus.
Atomic Weight
Average mass of an element's isotopes.
Periodic Table
Arrangement of elements based on atomic structure.
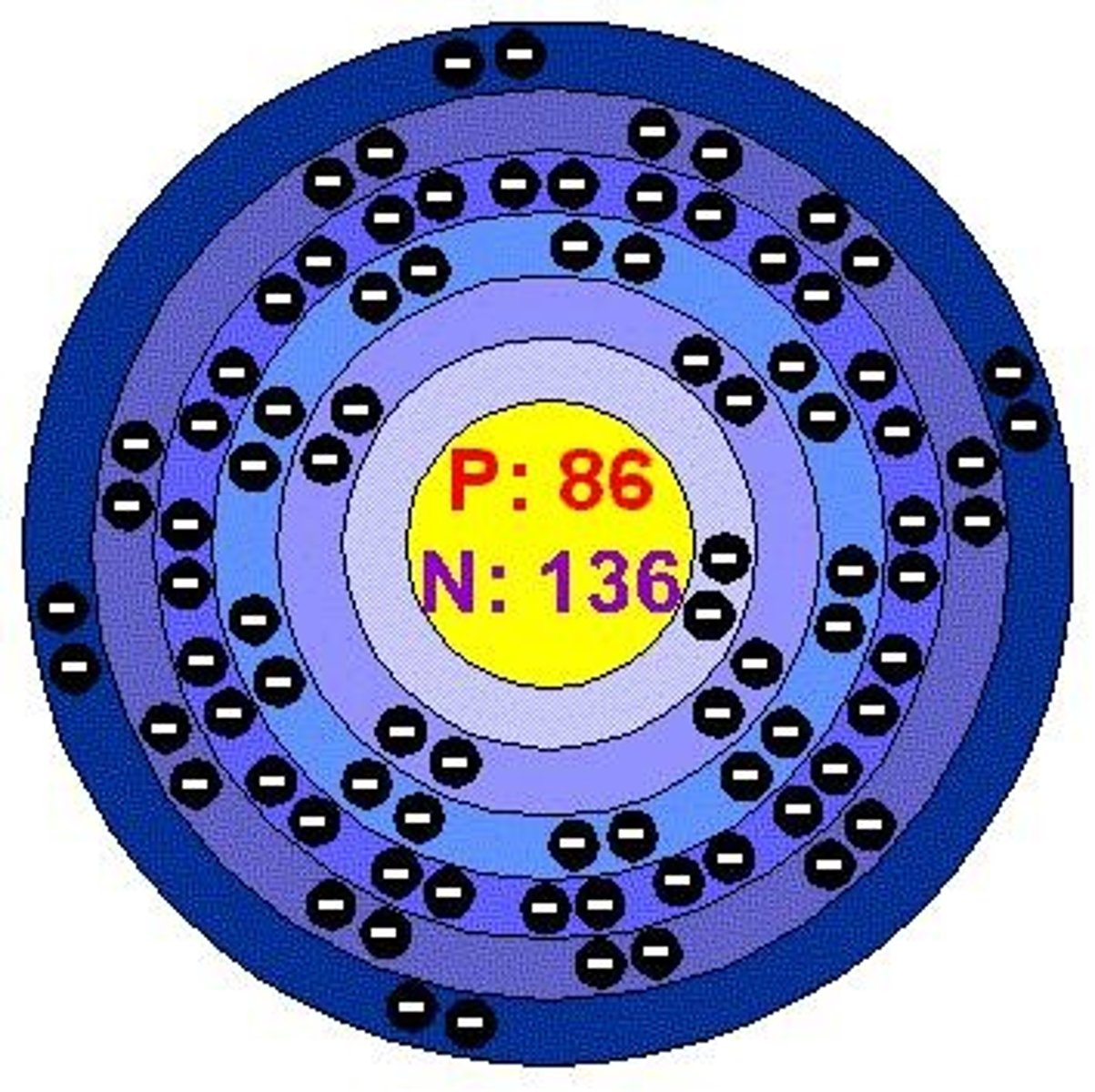
Electron Shells
Regions where electrons are likely to be found.
Orbital Electrons
Electrons defined by their probable location.
Electron Capacity Formula
Formula 2n² determines maximum electrons per shell.
K Shell DEFINITION
Innermost shell with highest binding energy.
Atomic Shells
Electron configuration based on 2n² formula.
K Shell
First shell with a maximum of 2 electrons.
L Shell
Second shell with a maximum of 8 electrons.
M Shell
Third shell with a maximum of 18 electrons.
N Shell
Fourth shell with a maximum of 32 electrons.