21 Buffers and Neutralisation
1/20
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
21 Terms
buffer def
a solution that resists change to pH upon addition of small amounts of H+ and OH-
(the pH does change but only slightly)
how are buffers made
from a weak acid and the salt of a weak acid
(or weak base/salt of a weak base)
Ka formula
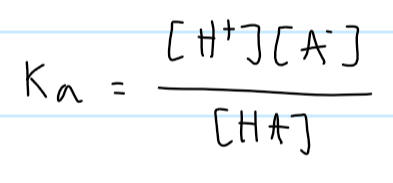
what affects Ka
only temperature
henderson hasselbalch equation
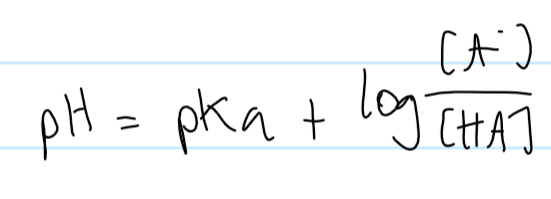
when HA : A- is 1:1, what’s the relationship between pH and pKa?
pH=pKa
indicator def
a weak acid where HA and A- have different colours
equivalence point def
the point at which the moles of acid = moles of base
end point def
the point at which the indicator will change colour
how do we choose an indicator
choose one that changes colour at the equivalence point
what is the pH of the equivalence point also = to
the pH of the salt formed