3) Polar reactions, Nucleophiles, and Electrophiles
1/14
Earn XP
Description and Tags
page 4
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
15 Terms
What is electrostatic/ionic attraction?
The force that pulls oppositely charged particles towards each other
What is a polar reaction?
a chemical reaction in which electrons move from an electron-rich species (nucleophile) to an electron-deficient species (electrophile), driven by differences in charge or electronegativity.
What is a nucleophile?
Atoms in organic compounds with partial negative charges
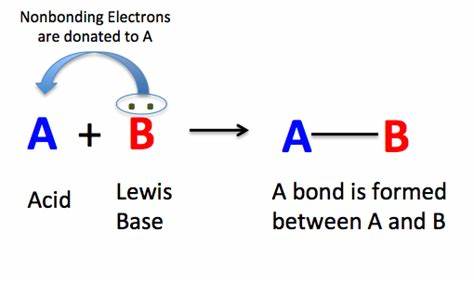
What is a Lewis base?
An electron donor (electron-RICH)
What is considered electron-rich?
A nucleophile
What is a electrophile?
Atoms in organic compounds with partial positive charges
What is a lewis acid?
An electron acceptor (e- POOR)
What is considered electron-poor?
An electrophile
What is a heteroatom?
An atom that’s NOT carbon or hydrogen
What does a lone pair do to a heteroatom?
It becomes nucleophilic
Pi bonds are..
nucleophilic!
Why are pi bonds nucleophilic and easier to break?
It has high e- density (- charge) and electrons are more exposed unlike sigma bonds due to being above and below the plane
What is a mechanism?
a step-by-step description of how a reaction occurs at the molecular level. It shows the movement of electrons, the breaking and forming of bonds, and the intermediates formed during the reaction.
Mechanisms for polar reactions use
double headed arrows (hetereolysis) where the nucleophile (LB) attacks an electrophile (LA)
What happens if there’s no formal charges on the starting atoms?
Additional bonds must be broken to create stable products