5-4 Multiple Covalent Bonds and Lewis-Dot Structures
1/30
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
31 Terms
Chemical Bond
wants to fill orbitals to get to 8
both sides think they have 8 electrons (delulu atoms)
mutual electrical attraction binding nuclui and valence electrons
Ionic Bond
transfer of electrons
metal atom loses electron(s) to non-metal atom
similar to a extrovert talking to their other friends while their introvert friend is just there
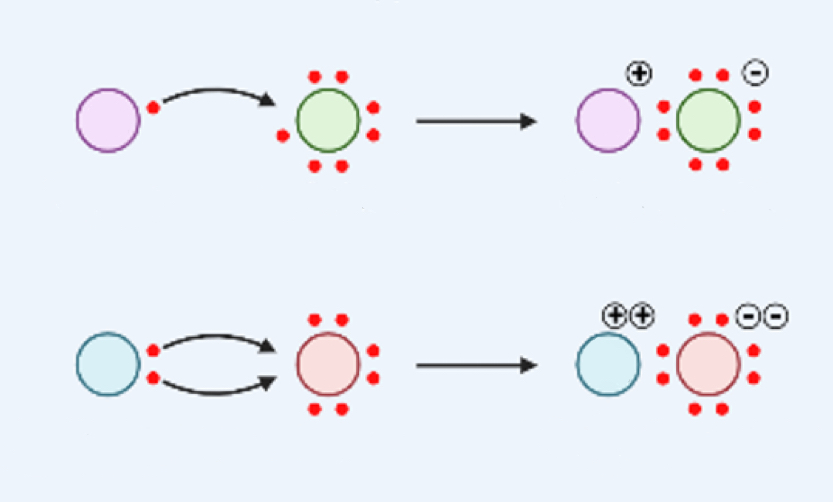
Covalent Bond
sharing of electrons
two non-metal atoms share electrons
everyone has SOME electrons
delulu bond (the two girls who both think the same guy is THEIR man example)
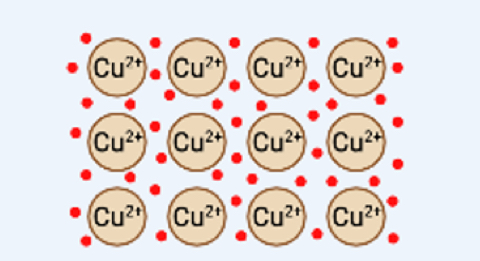
Metallic
one with hella electrons
delocalized
free mobility through latice
positive metal ions attract conducting electrons
Electrons of one atom
are attracted to protons of another
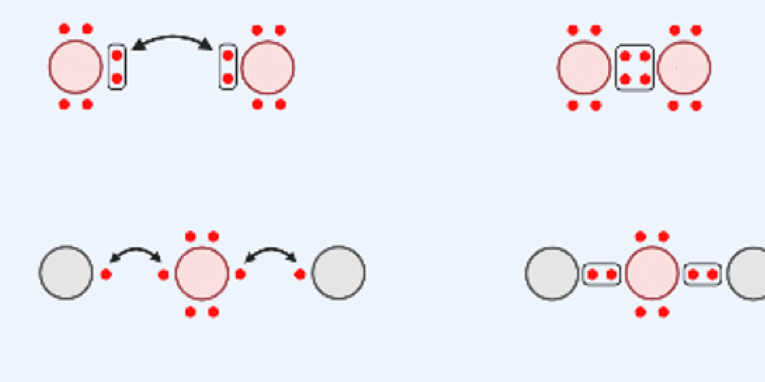
Covalent Bonds
share valence electrons → create molecules
similar to the weird couple at lunch (hard to break apart)
can share more than one electron (single, double, and triple)
Molecule
a group of atoms held together by covalent bonds
Molecular Compound
compounds created by simple units of molecules
Diatomic molecules
two molecules involved
Di=2
Heteronuclear
Homonuclear
Heteronuclear
two different atoms
Homonuclear
two same atoms
Almost always homonuclear atoms are?
H2, N2, F2, O2, I2, C2, B2
Have No Fear Of Ice Cold Beer
Single Bond
two atoms sharing TWO electrons
Double Bond
two atoms sharing FOUR electrons
Triple Bond
two atoms sharing SIX electrons
Lewis-Dot Structures
used to represent molecules
Lone Pair
(Lewis-Dot Structure)
represented by a .
Electrons that are alone (does not have another on the same element to pair with it)
Pair Bonded
Represented by long dashes
shows how valence electrons are arranged in atoms in a molecule
Single bond -
Double bond =
Triple bond -= (imagine 3 lines stacked on top of each other)

How many electrons does row 1 of the periodic table have?
1
How many electrons does row 2 of the periodic table have?
2

How many electrons does row 9 of the periodic table have?
1
How many electrons does row 10 of the periodic table have?
2

How many electrons does row 11 of the periodic table have?
1

How many electrons does row 12 of the periodic table have?
2

How many electrons does row 13 of the periodic table have?
3

How many electrons does row 14 of the periodic table have?
4

How many electrons does row 15 of the periodic table have?
5

How many electrons does row 16 of the periodic table have?
6

How many electrons does row 17 of the periodic table have?
7
How many electrons does Helium have?
1
How many electrons does row 18 of the periodic table have?
8
