Chapter 21 Atomic spectroscopy
0.0(0)
0.0(0)
Card Sorting
1/54
Earn XP
Description and Tags
Quantitative analytical chemistry chapter 21 atomic spectroscopy
Study Analytics
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
55 Terms
1
New cards
atomic spectroscopy
the study of the electromagnetic radiation absorbed and emitted by atoms.
2
New cards
what happens in atomic spectroscopy
• Samples are vaporized and decompose into atoms and ions at high temperature.
• Some of the metal atoms may be raised to higher energy level and emit characteristic radiation. But large amount of metal atoms will remain in non emitting ground state and can absorb light.
• Concentrations are measured based on their absorbance or emission of light.
• Some of the metal atoms may be raised to higher energy level and emit characteristic radiation. But large amount of metal atoms will remain in non emitting ground state and can absorb light.
• Concentrations are measured based on their absorbance or emission of light.
3
New cards
absorption spectrum of hydrogen in the visible region

4
New cards
emission spectrum of hydrogen in the visible region

5
New cards
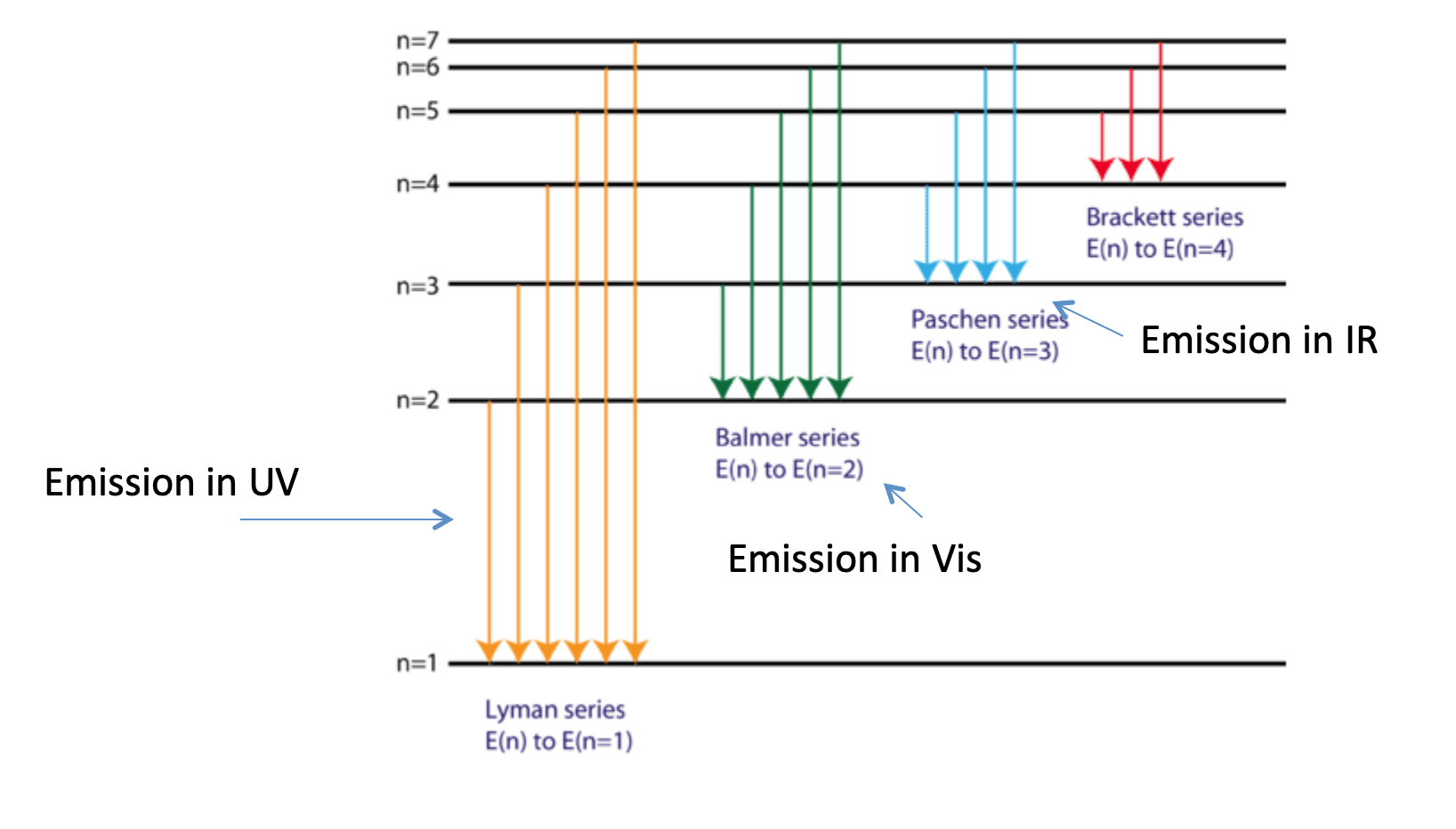
electron transitions for hydrogen atom
6
New cards
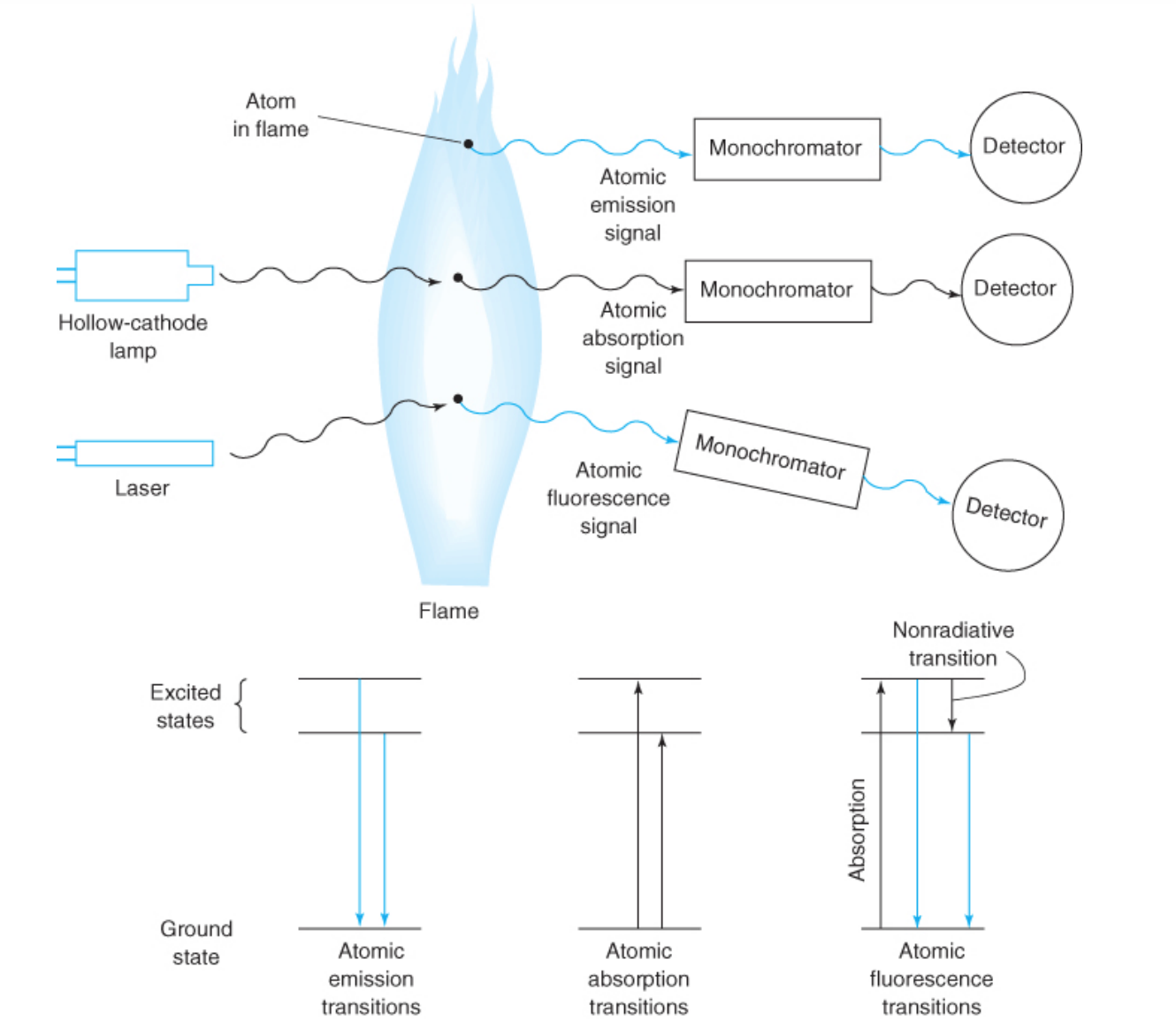
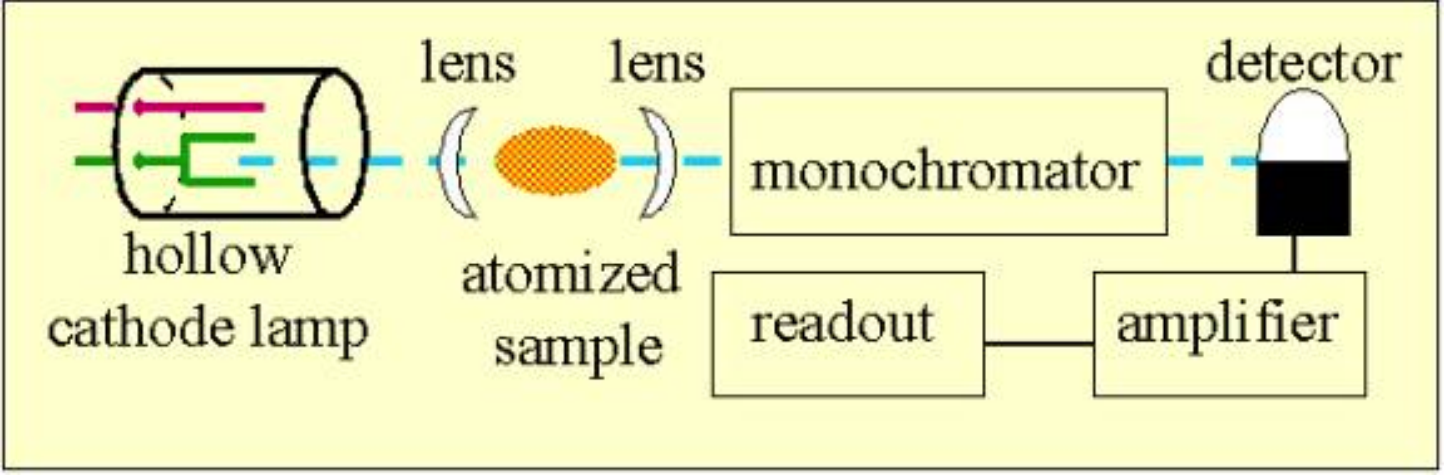
flame atomic absorption spectroscopy
• A liquid sample is aspirated into the flame (2000-3 000 K).
• Liquid evaporates and the remaining solid is atomized in the flame. Atoms absorbs light of specific λs
• Determination of one element at a time. (one lamp one element)
• Liquid evaporates and the remaining solid is atomized in the flame. Atoms absorbs light of specific λs
• Determination of one element at a time. (one lamp one element)
7
New cards
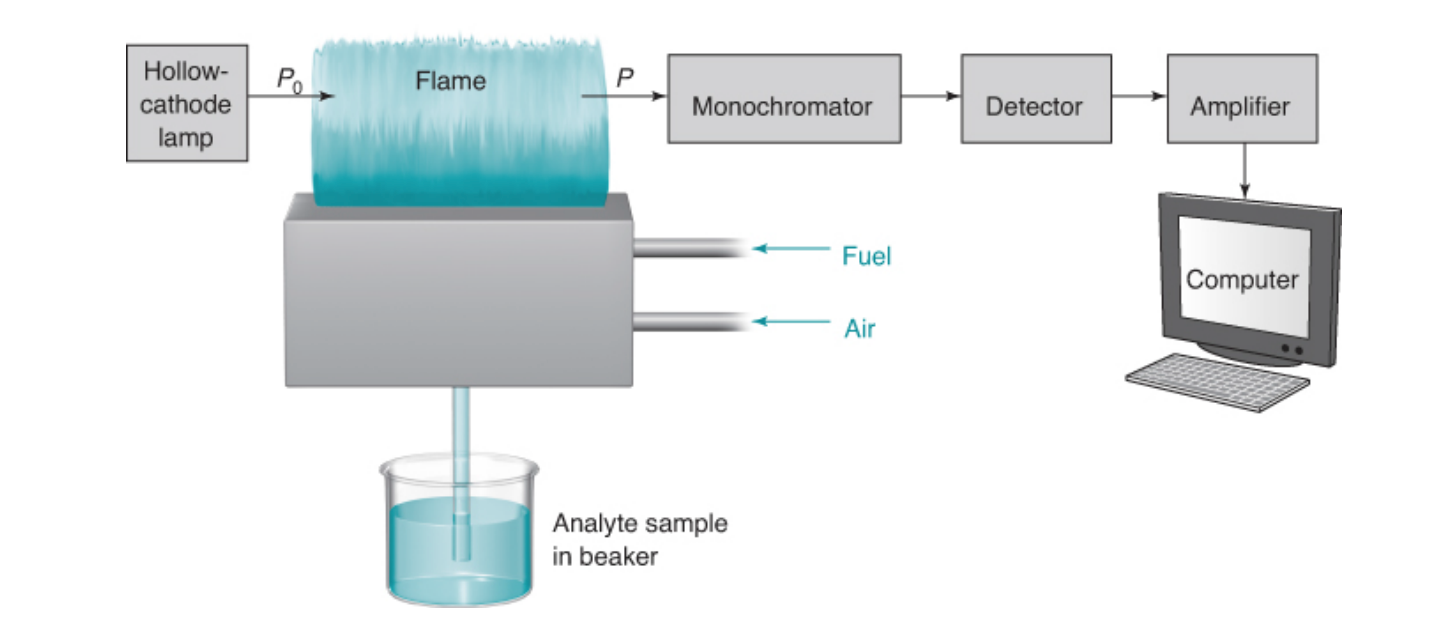
hollow-cathode lamp
passes light through the sample in the flame and the intensity of the transmitted radiation is measured by a photon transducer
8
New cards
0\.001
Spectra of molecules have bandwidths of \~10-100 nm, whereas spectra of atoms consist of sharp lines with widths of \~___ nm.
9
New cards
atomic emission spectroscopy
radiation from hot atoms whose electrons have been promoted to an excited state in the flame or plasma is emitted.
• No lamp is needed, but atomization at higher temperatures (5000 – 8000 K).
• No lamp is needed, but atomization at higher temperatures (5000 – 8000 K).
10
New cards
atomic fluorescence spectroscopy
atoms in the flame are irradiated by a laser to promote them to an excited state from which they can fluoresce to return to the ground state.
11
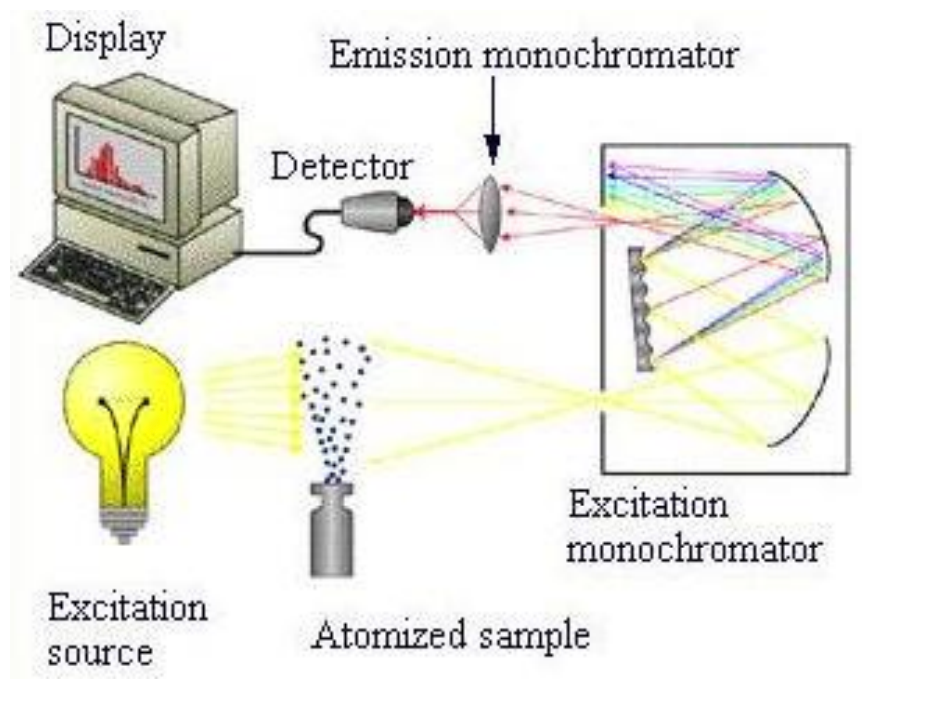
New cards
basic diagram for atomic absorbance spectroscopy
12
New cards
nebulization
formation of small droplets
13
New cards
aerosol
A fine suspension of liquid (or solid) particles in a gas
14
New cards
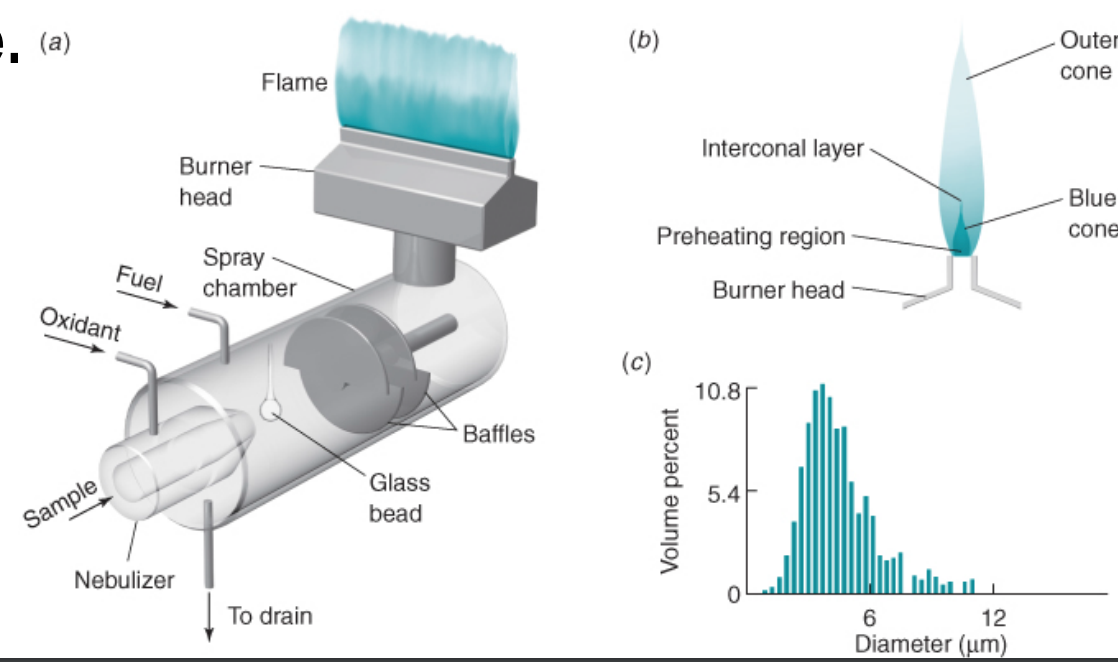
how flame atomic absorption works
• Flames use a premix burner, in which fuel, oxidant, and sample are mixed prior to introduction into the flame.
• Sample solution is drawn into the pneumatic nebulizer by rapid flow of oxidant (usually air) past the tip of the sample capillary.
• Liquid breaks into a fine mist as it leaves the capillary. • The spray is directed against a glass bead, upon which the droplets break into smaller particles.
• The nebulizer creates an aerosol from the liquid sample.
• The mist, oxidant, and fuel flow past baffles that promote further mixing and block large droplets of liquid.
• Excess liquid collects at the bottom of the spray chamber and flows out to a drain.
• Sample solution is drawn into the pneumatic nebulizer by rapid flow of oxidant (usually air) past the tip of the sample capillary.
• Liquid breaks into a fine mist as it leaves the capillary. • The spray is directed against a glass bead, upon which the droplets break into smaller particles.
• The nebulizer creates an aerosol from the liquid sample.
• The mist, oxidant, and fuel flow past baffles that promote further mixing and block large droplets of liquid.
• Excess liquid collects at the bottom of the spray chamber and flows out to a drain.
15
New cards
5%
Aerosol reaching the flame contains only about ____ of the initial sample.
16
New cards
rich flame
excess fuel. increases sensitivity because excess carbon can reduce the metal oxides and hydroxides.
17
New cards
lean flame
excess oxidant. Gives hotter flame
18
New cards
liquid
flame absorption spectroscopy can only use ____ samples.
19
New cards
sensitive
An electrically heated graphite furnace is more ______ than a flame and requires less sample.
20
New cards
how graphite furnace works
• 1-100 µL of sample is injected into the furnace through a hole at the center. The sample should go to the floor.
• Light from a hollow cathode lamp travels through the window at each end of the graphite tube.
• A temperature program is applied
• Light from a hollow cathode lamp travels through the window at each end of the graphite tube.
• A temperature program is applied
21
New cards
Steps of Graphite Furnace experiment
drying, charring, atomization, cleaning
22
New cards
advantages of graphite furnace vs flame absorption.
1. 1-2mL sample volume is the minimum required
for a flame compared to 1µL for a furnace.
2. Flames hold the sample in the optical path for
\~1s as it rises thru the flame; however, a
furnace confines the sample in the light path for
several s.
3. Multiple aliquots of sample can be injected on to the furnace platform and evaporated, pre concentrating the sample in the furnace prior to analysis.
4. Flames require nebulization, which can dilute sample; there is no nebulization step using a furnace.
23
New cards
disadvantages of graphite furnace
• have a limited lifetime.
• Memory effect – interference from previous runs.
• Flame is less expensive
• Memory effect – interference from previous runs.
• Flame is less expensive
24
New cards
direct solid sampling
when a solid is analyzed without sample preparation
25
New cards
matrix
Everything in the sample other than the analyte
26
New cards
matrix modifier
a substance added to the sample to reduce the loss of the analyte during the charring by making the matrix more volatile or the analyte less volatile.
27
New cards
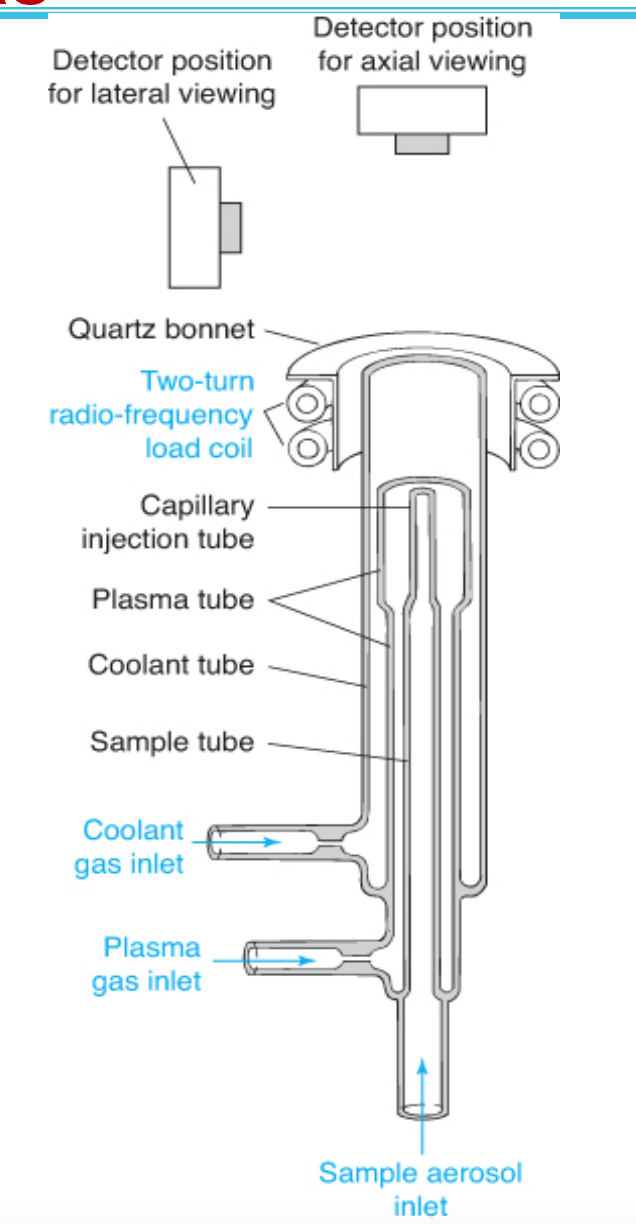
Inductively coupled plasma
• Twice as hot as the flame
• Stable
• Inert Ar environment
• Simultaneous multi-element analysis
• Costs more to purchase and operate than the flame
• Stable
• Inert Ar environment
• Simultaneous multi-element analysis
• Costs more to purchase and operate than the flame
28
New cards
how temperature affects atomic spectroscopy
1. It determines the degree to which a sample breaks
down into atoms.
2. It determines the extent to which an atom is found
in its ground, excited, or ionized state (Boltzmann
distribution)
This affects the strength of the observed signal.
29
New cards
Boltzmann Distribution
describes the relative populations of different states at thermal equilibrium. If equilibrium exists, then N\*/N0 of any two states:
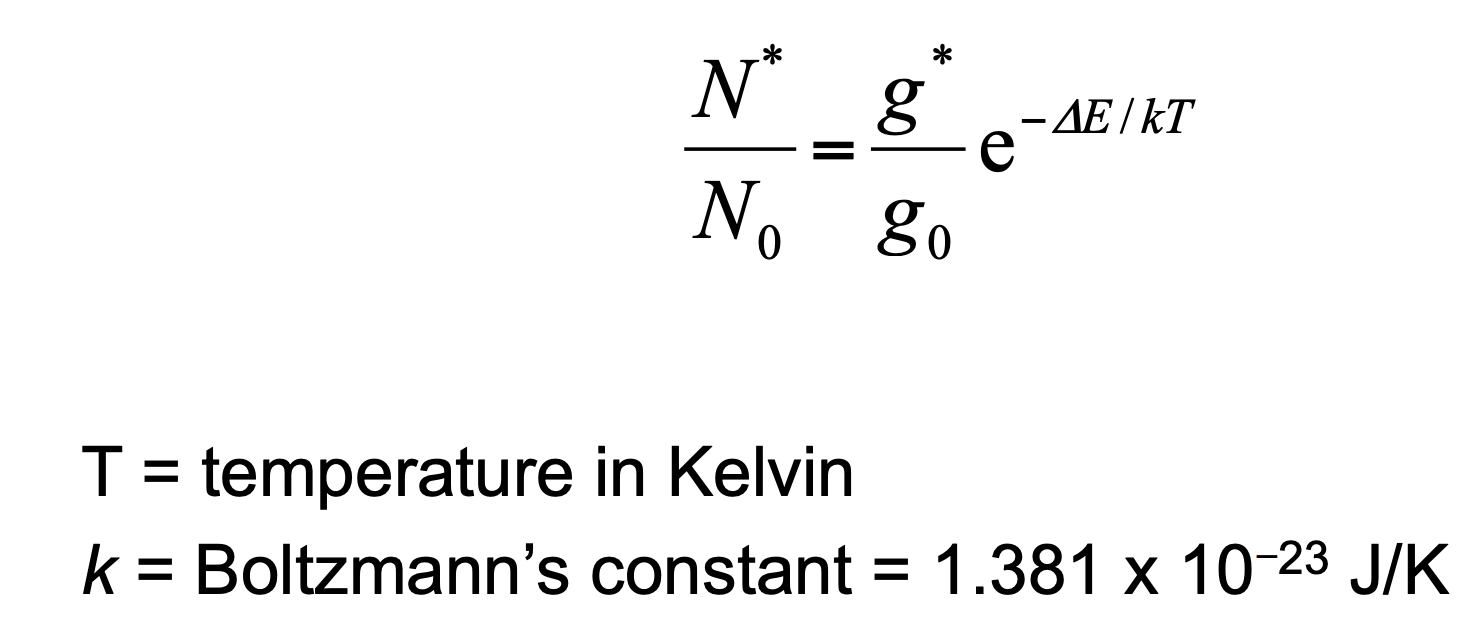
30
New cards
2600
At ____ K, 99.98% of the atoms are in their ground state.
31
New cards
drying step
graphite furnace heats up to remove any solvent from the sample (\~150)
32
New cards
charred step
Graphite furnace removes any interferences by heating up dried solvent
33
New cards
atomization
graphite furnace glows as it is \~3000 degrees. Atoms turn to gas phase so that they can be analyzed.
34
New cards
Cleaning
graphite furnace heats up cell even more to get rid of any sample in the furnace.
35
New cards
10
Varying the temperature by __ K hardly affects the ground-state population and would not noticeably affect the signal in atomic absorption
36
New cards
4
Atomic emission occurs from the excited state, therefore ___ % increase in the excited state would increase the emission intensity by …%

37
New cards
plasmas
what atomic emission typically uses.
38
New cards
10-3 to 10-2 nm
Monochromators generally cannot isolate lines narrower than _____ .
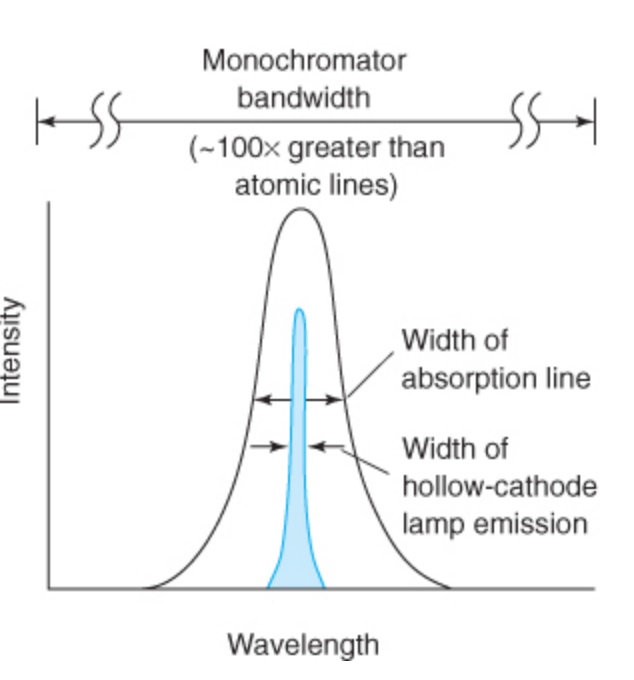
39
New cards
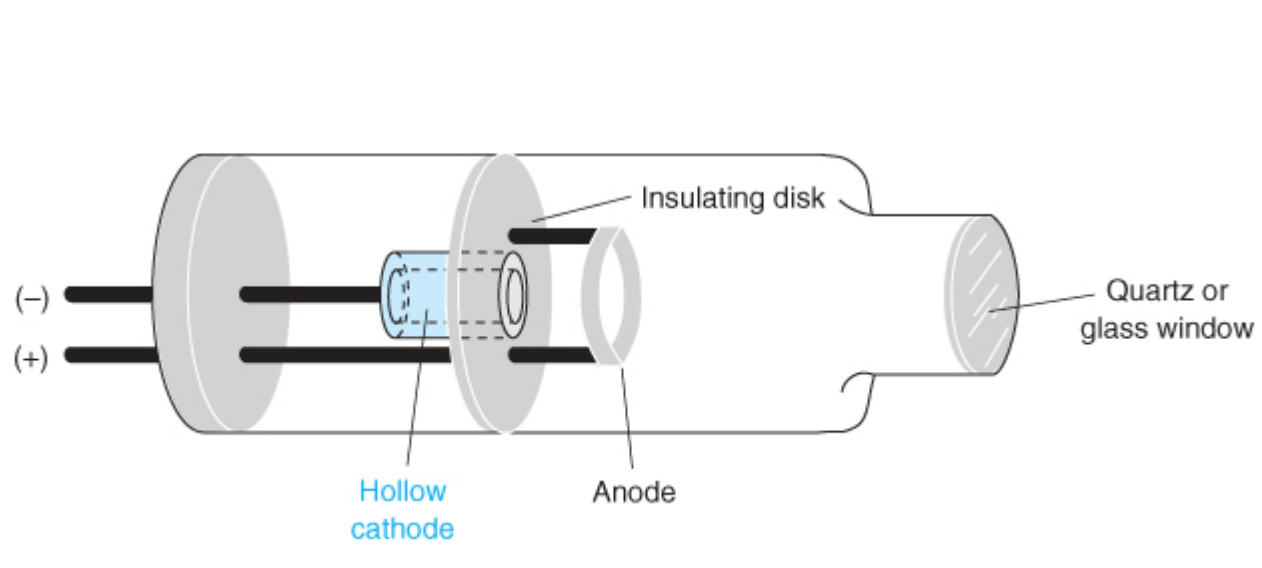
vapor
To produce narrow lines of the correct frequency, we use hollow-cathode lamps containing ____ of the same element being analyzed
40
New cards
substantially narrower
Beer’s law requires that the linewidth of the radiation source should be _______ than the linewidth of the absorbing sample.
41
New cards
Atomic absorption linewidths
have an intrinsic width of \~10-4 nm due to Heisenberg uncertainty, Doppler broadening and Pressure broadening
42
New cards
excited state
The higher the temperature, the higher the percentage of atoms in the ______ .
43
New cards
Heisenberg uncertainty principle
states that the shorter the lifetime of the excited state, the more uncertain its energy
44
New cards
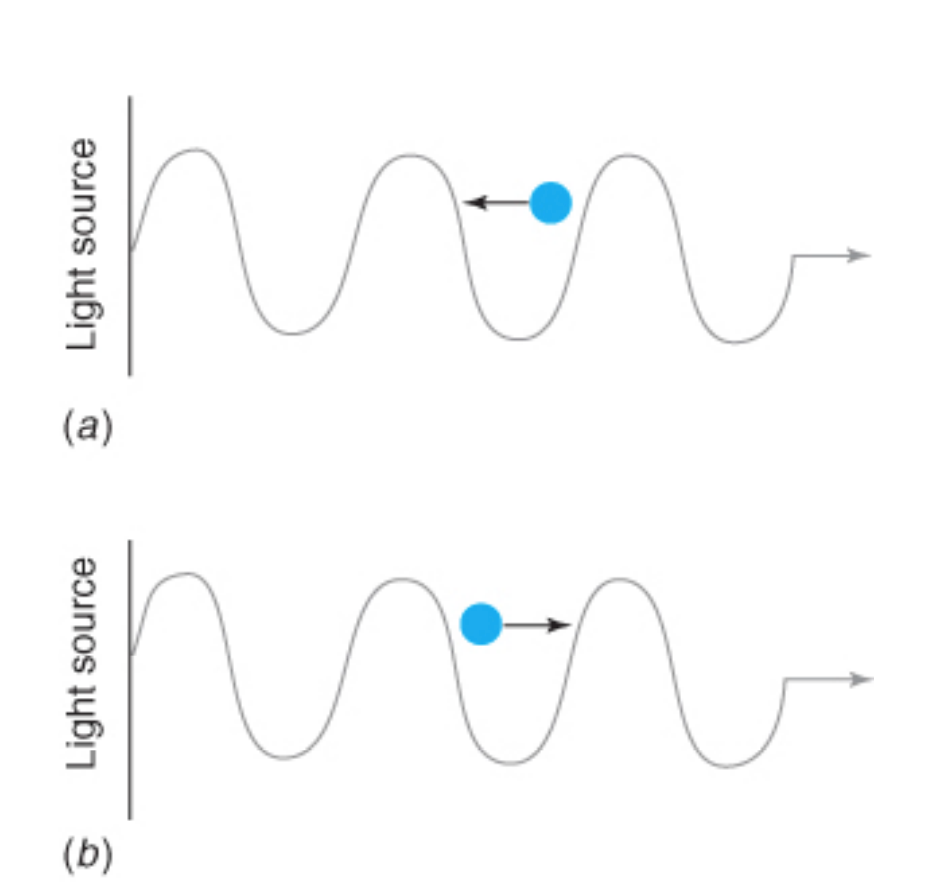
Doppler broadening
• atomic motion occurs in every direction.
• An atom moving toward the radiation source “sees” a higher frequency light than one moving away.
• There is no shift for atoms moving perpendicular to the source.
• The result is a symmetric distribution of λs.
• An atom moving toward the radiation source “sees” a higher frequency light than one moving away.
• There is no shift for atoms moving perpendicular to the source.
• The result is a symmetric distribution of λs.
45
New cards
Pressure broadening
• is caused by collisions of the emitting or absorbing species with other atoms or ions in a hot environment.
• Collisions between atoms shorten the lifetime of the excited state.
• The collision frequency is proportional to the pressure.
• This is similar in magnitude to the effect of Doppler broadening and yields linewidths of 10-3 to 10-2 nm in atomic spectroscopy.
• Collisions between atoms shorten the lifetime of the excited state.
• The collision frequency is proportional to the pressure.
• This is similar in magnitude to the effect of Doppler broadening and yields linewidths of 10-3 to 10-2 nm in atomic spectroscopy.
46
New cards
sputtering
When gaseous cations acquire enough kinetic energy to dislodge some of the metal atoms from the cathode surface producing an electron cloud.
• A portion of the _____ metal atoms are in their excited state and emit characteristic radiation as they return to their ground state.
• The metal atoms eventually diffuse back to the cathode, or deposit on the glass walls of the tube.
• A portion of the _____ metal atoms are in their excited state and emit characteristic radiation as they return to their ground state.
• The metal atoms eventually diffuse back to the cathode, or deposit on the glass walls of the tube.
47
New cards
Background correction
• is necessary in atomic spectroscopy to distinguish the analyte signal from absorption, emission, and optical scattering of the sample matrix, the flame, plasma, or graphite furnace.
• It is critical for graphite furnaces due to residual smoke from charring.
• It is critical for graphite furnaces due to residual smoke from charring.
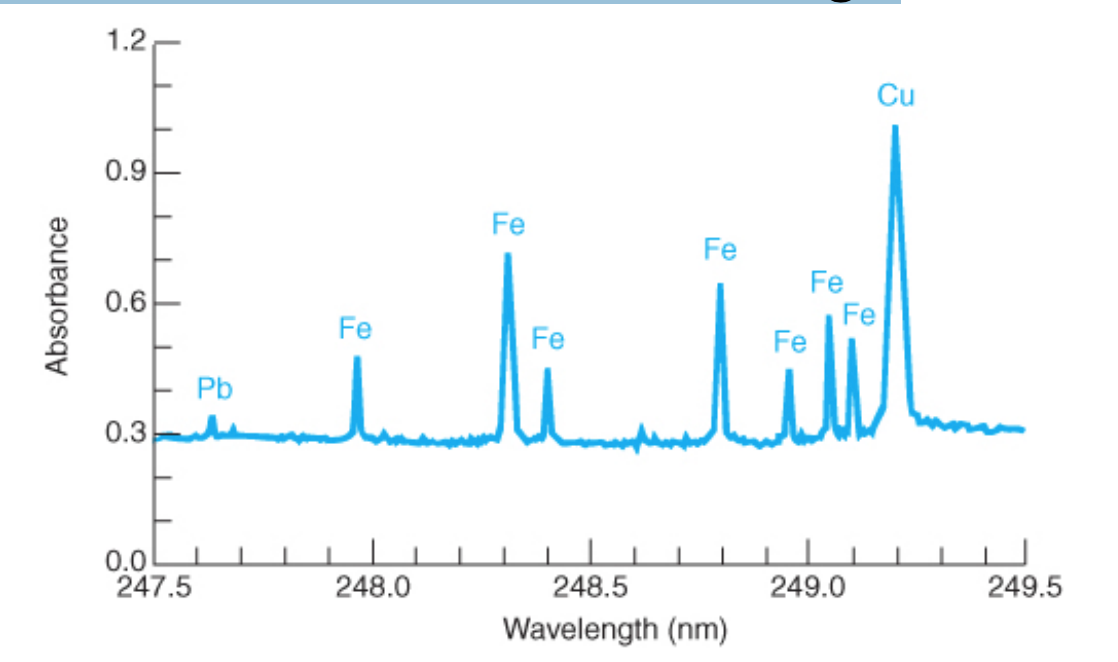
48
New cards
beam chopping
• or electrical modulation of the hollow-cathode lamp (pulsing it on and off), can distinguish the signal of the flame from the atomic line at the same wavelength.
• The figure shows light from the lamp being periodically blocked by a rotating chopper.
• Signal reaching the detector while the beam is blocked must be from the flame emission.
• The figure shows light from the lamp being periodically blocked by a rotating chopper.
• Signal reaching the detector while the beam is blocked must be from the flame emission.
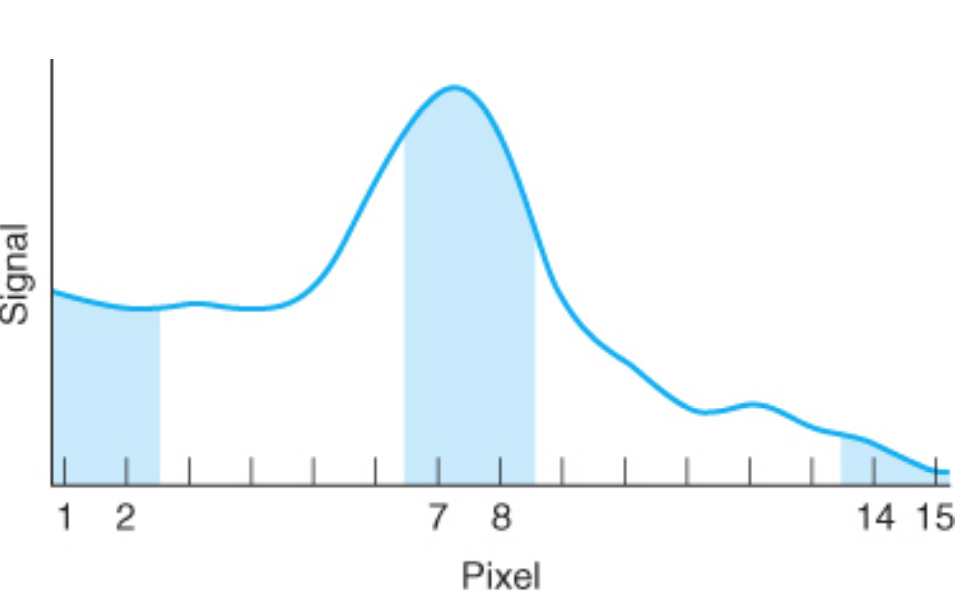
49
New cards
the lamp and the flame
Signal reaching the detector when the beam is not blocked is from _____. It does not correct for scattering
50
New cards
analytical signal
The difference between the lamp and the flame is the ______.
51
New cards
Deuterium lamp background correction
broad emission from a D2 lamp is passed through the flame in alternation with that from the hollow cathode.
Light from the hollow cathode lamp is absorbed by the analyte as well as absorbed and scattered by the background.
Light from the hollow cathode lamp is absorbed by the analyte as well as absorbed and scattered by the background.
52
New cards
Zeeman effect
• The splitting of a spectrum line into three components by the application of a magnetic field.
• Two are shifted and one is not.
• The unshifted one does not absorb the light because it does not have the correct polarization.
• Two are shifted and one is not.
• The unshifted one does not absorb the light because it does not have the correct polarization.
53
New cards
Deuterium lamp correcting
• The strong magnetic field is pulsed on and off.
• Sample and background are observed when the field is off.
• Only background is observed when the field is on.
• The difference between them is the corrected signal.
• Sample and background are observed when the field is off.
• Only background is observed when the field is on.
• The difference between them is the corrected signal.
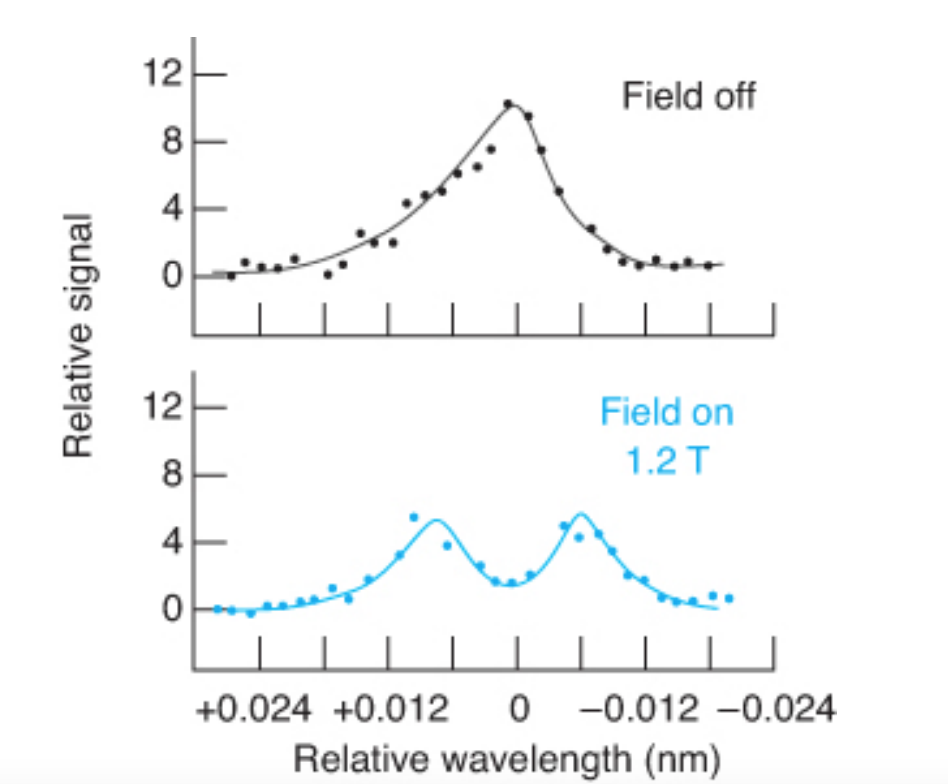
54
New cards
detection limit for furnaces
is typically two orders of magnitude lower than that observed with a flame because the sample is confined in the small volume of the furnace for a relatively long time.
55
New cards
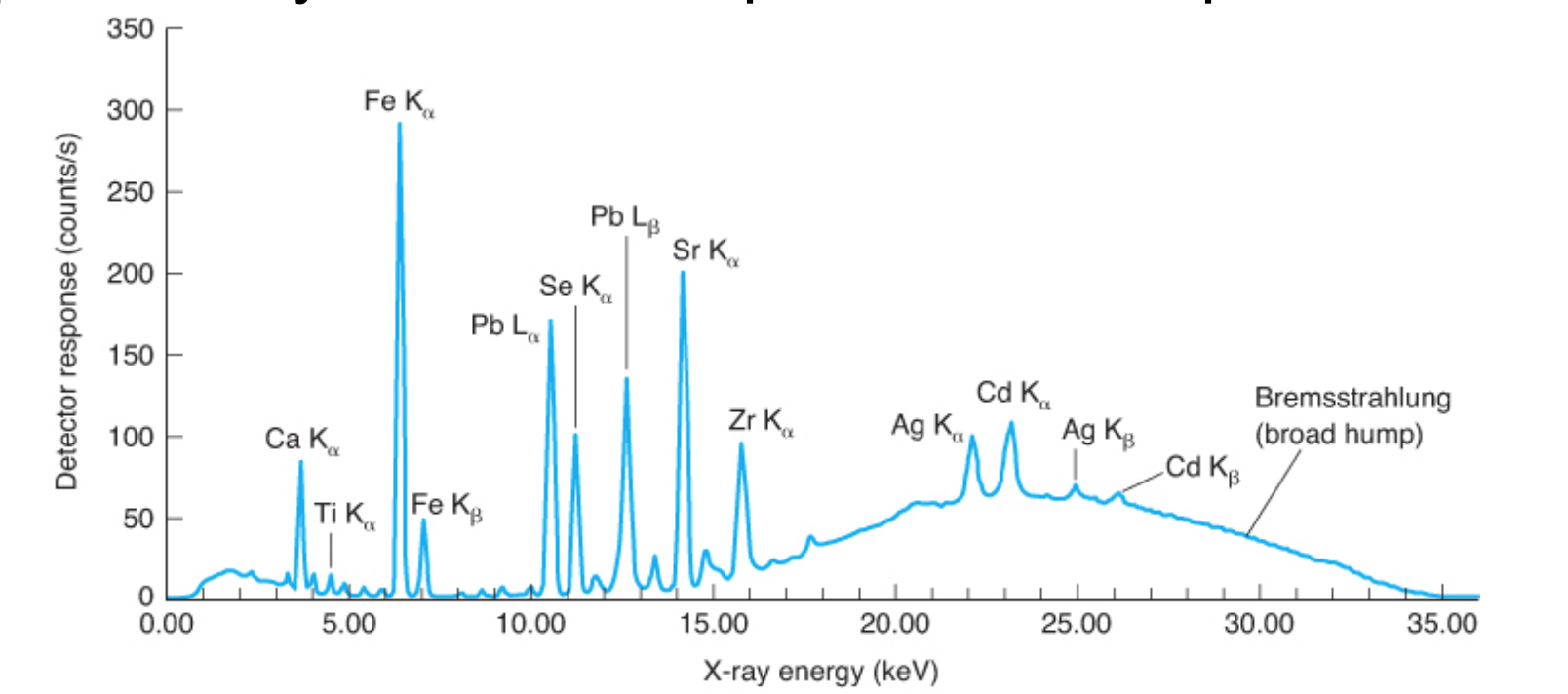
X-Ray Fluorescence
is the emission of X-rays following the absorption of X-rays by a material. Elements are identified by their peak energies and quantified by the number of photons in each peak.