Rate of reactions
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
Collision theory: for a chemical reaction to happen:
reactant particles must colide with eachother.
the particles must have enough energy for them to react
How can the mean rate of reaction be calculated:
Mean rate of reaction = quantity of reactant used DIVIDED by time taken
Measuring Rate of reaction using a graph gradient:
the steeper the line, the greater the rate of reaction.
The greater the frequency of successful collisions, the greater the rate of reaction. If the concentration of a reacting solution or the pressure of a reacting gas is increased:
the reactantparticles become more crowded
the frequency of collisions between reactant particles increases
the rate of reaction increases
Calculating the rate of reaction from the gradient of a tangent:
rate of reaction = change in mass or volume of product DIVIDED by change in time
If a large lump is divided or ground into a powder:
its total volume stays the same
the area of exposed surface increases
the surface area to volume ratio increases
If the temperature of the reaction mixture is increased:
reactantparticles move more quickly
the energy of the particles increases
the frequency of successful collisions between reactant particles increases
the proportion of collisions which are successful increases
the rate of reaction increases
A catalyst is a substance that:
increases the rate of a reaction
does not alter the products of the reaction
is not chemically changed or used up at the end of the reaction
How do catalysts work:
A catalyst provides an alternative reaction pathway that has a lower activaion energy
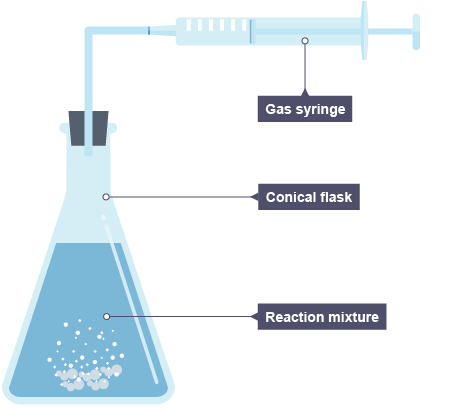
Required practicle - Measure the production of a gas ( method )
Support a gas syringe with a stand, boss and clamp.
Using a measuring cylinder, add 50 cm3 of dilute hydrochloric acid to a conical flask.
Add 0.4 g of calcium carbonate to the flask. Immediately connect the gas syringe and start a stop clock.
Every 10 seconds, record the volume of gas produced.
When the reaction is complete, clean the apparatus as directed by a teacher.
Repeat steps 1 to 5 with different concentrations of hydrochloric acid.
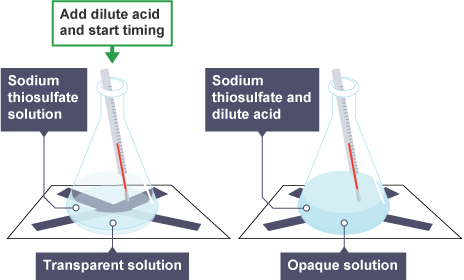
Required practicle -investigate the rate of reaction by colour change
Using a measuring cylinder, add 50 cm3 of dilute sodium thiosulfate solution to a conical flask.
Place the conical flask on a piece of paper with a black cross drawn on it.
Using a different measuring cylinder, add 10 cm3 of dilute hydrochloric acid to the conical flask. Immediately swirl the flask to mix its contents, and start a stop clock.
Look down through the reaction mixture. When the cross can no longer be seen, record the time on the stop clock.
Measure and record the temperature of the reaction mixture, and clean the apparatus as directed by a teacher.
Repeat steps 1 to 5 with different starting temperatures of sodium thiosulfate solution.
What is the rate of reaction
The rate of a reaction is a measure of how quickly a reactant is used up, or a product is formed.