Chemistry I Module of the MCAT Self Prep eCourse: Lesson 5: Balancing Equations (Pro)
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
16 Terms
Lesson 5: Balancing Equations
Lesson 5: Balancing Equations
CRB The law of conservation of matter says that the amount of matter does not change in a chemical reaction. How do you keep this in mind as you balance a chemical reaction?
If a certain number of atoms of an element are reactants in a chemical reaction, that same number of atoms must be present in the products, and vice versa.
CRB The law of conservation of matter could also be applied to electrons, so that electrons are not created nor destroyed in a reaction, only transferred from one element to another. If this is true, which of the following can be changed in a chemical reaction?
(A) Number of nucleons
(B) Oxidation state
(C) Electron affinity
(D) None of the above can change
(B) Oxidation state
The oxidation state of an atom is similar to an atom's "ownership" of valence electrons, and could change when interacting with other compounds, like in a chemical reaction.
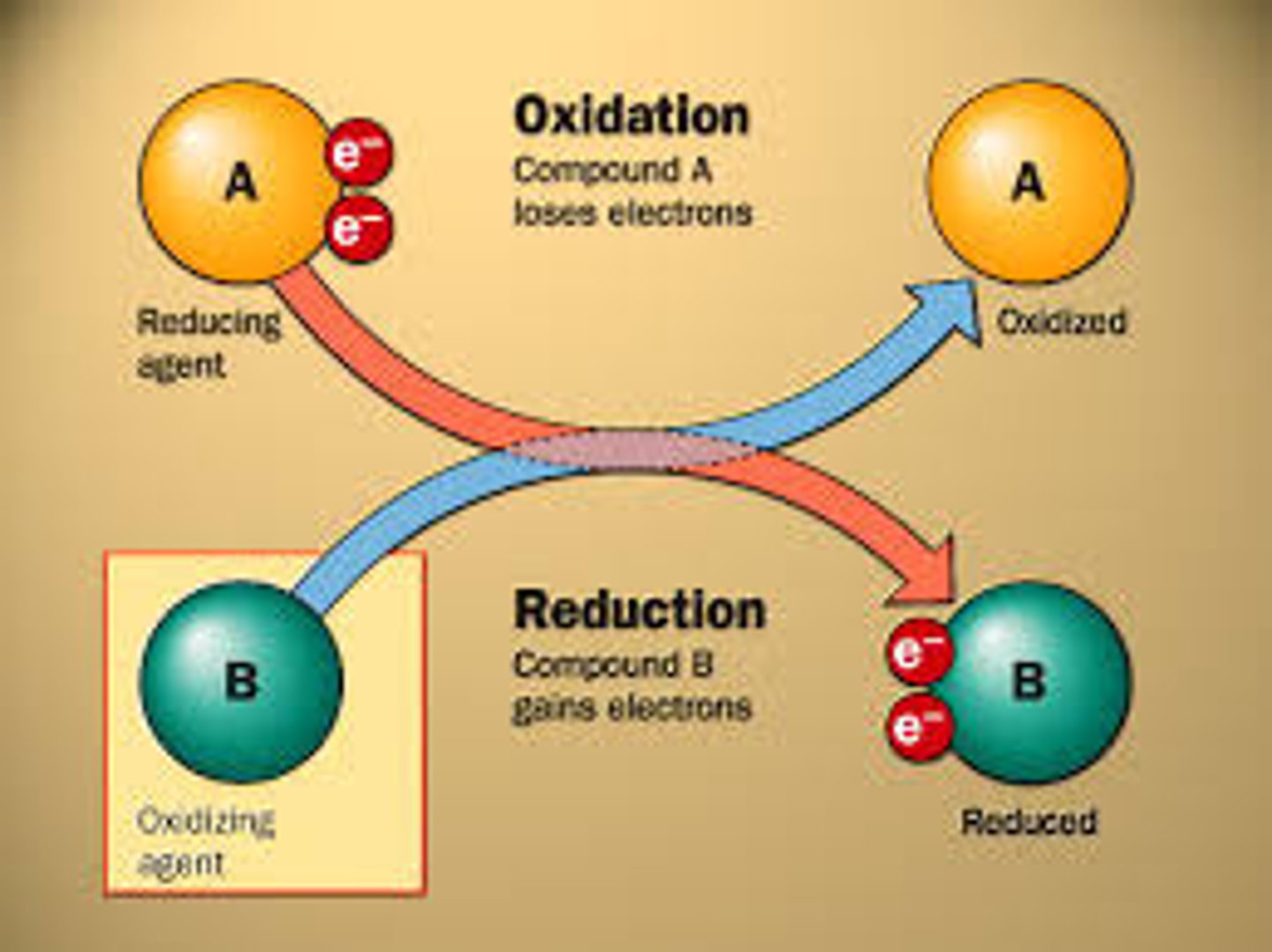
CRB Which of the following is NOT a commonly used rule when determining oxidation states of elements in a compound?
(A) Group 1 metals have a +1 oxidation state and group 2 metals have a +2 oxidation state
(B) Hydrogen will have a +1 oxidation state when bound to an atom more electronegative than Carbon, and a 0 oxidation state when bound to an atom less electronegative than Carbon
(C) Halogens will have a -1 oxidation state
(D) Oxygen and elements in its group (16 or 6A) has a -2 oxidation state
(B) Hydrogen will have a +1 oxidation state when bound to an atom more electronegative than Carbon, and a 0 oxidation state when bound to an atom less electronegative than Carbon
Actually, a Hydrogen atom generally will have a +1 oxidation state in most compounds, but will have -1 oxidation state when bound to less electronegative atoms (typically metal hydrides).
Balance the following chemical equation: Al + O2 -> Al2O3
4Al + 3O2 -> 2Al2O3
Balance the following chemical equation: CH4 + O2 -> CO2 + H2O
CH4 + 2O2 -> CO2 + 2H2O
Balance the following chemical equation: C6H12O6 + O2 -> CO2 + H2O
C6H12O6 + 6O2 -> 6CO2 + 6H2O
How is the formal charge of Chlorine changing in this hypothetical reaction: Li(s) + Cu(s) + OCl- -> CuO + LiCl?
(A) Chlorine goes from a -1 to a +1 formal charge
(B) Chlorine goes from a +1 to a -1 formal charge
(C) Chlorine goes from a 0 to a -1 formal charge
(D) Chlorine does not change, staying at a -1 formal charge
(C) Chlorine goes from a 0 to a -1 formal charge
Li(s) + Cu(s) + OCl⁻ -> CuO + LiCl
Chlorine's formal charge goes from 0 to -1.
We know that Chlorine starts out at a 0 formal charge based on the Lewis dot structure of Hypochlorite and how we calculate formal charges.
We also know that Chlorine has a -1 formal charge in LiCl because of the nature of that ionic bond.

What is the key characteristic of a disproportionation reaction compared to other reduction-oxidation reactions?
In a disproportionation reaction, a single element is both oxidized and reduced.
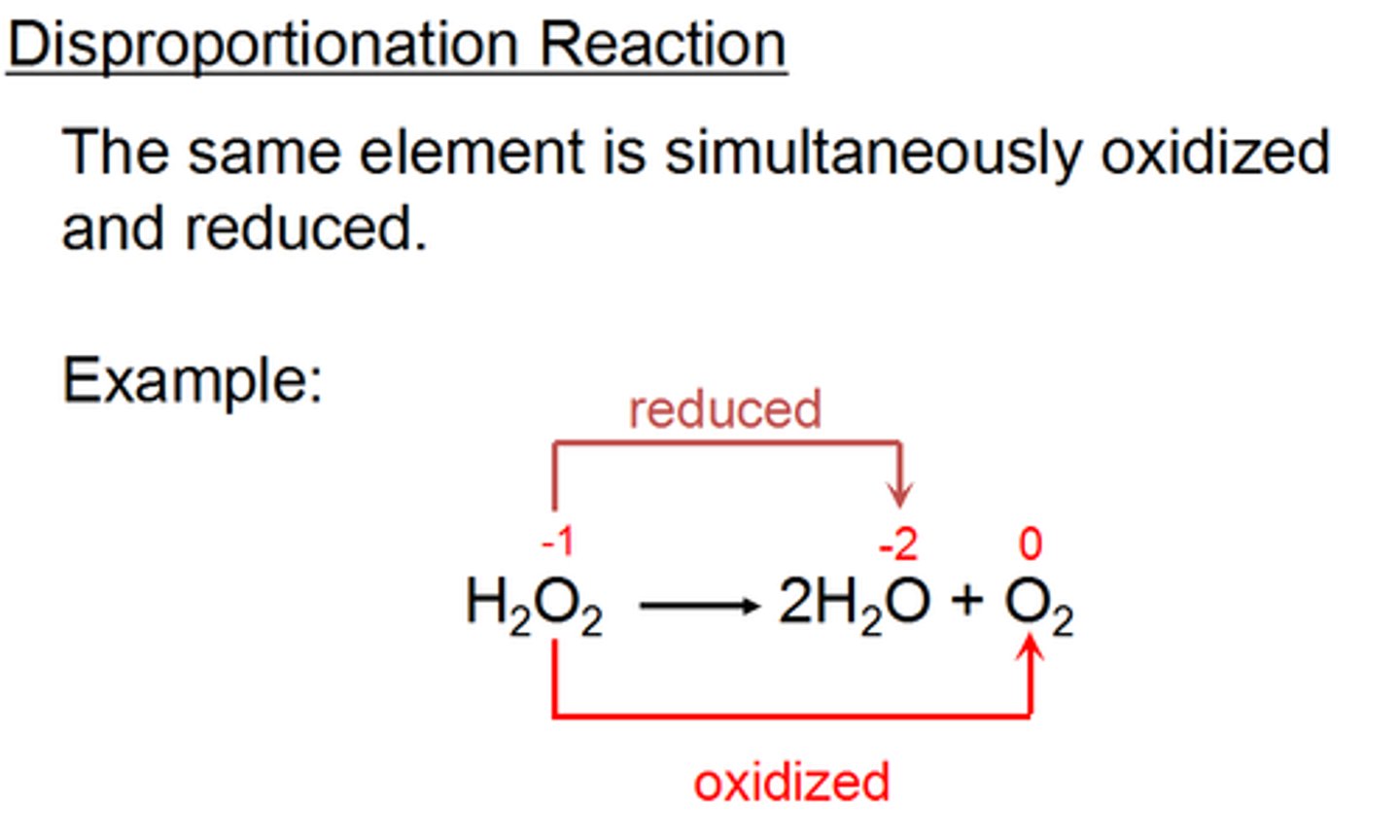
Is the following reaction a disproportionation reaction? Why or why not?: 2Cu+ -> Cu(s) + Cu2+
2Cu+ -> Cu(s) + Cu2+ is a disproportionation reaction because Copper is both oxidized and reduced. In a disproportionation reaction, an element is both oxidized and reduced.
Balance the following redox reaction in acid: MnO4-(aq) + I-(aq) → Mn2+(aq) + I2(s)
10I-(aq) + 2MnO4-(aq) + 16H+(aq) → 5I2(s) + 2Mn2+(aq) + 8H2O(l)
See Chem Libre's "Balancing Redox Reactions: Examples" Page for a detailed solution.
CRB Fill in the blanks: In older naming systems, cations that have multiple positive oxidation states would have their more charged oxidation state end with ______, and their less charged oxidation state end with ______.
(A) -ous , -ic
(B) -ite , -ate
(C) -ic , -ous
(D) -ate , -ite
(C) -ic , -ous
In older naming systems, cations that have multiple positive oxidation states would have their more charged oxidation state end with -ic, and their less charged oxidation state end with -ous.
CRB Fill in the blanks: In older naming systems, elements that can form more than one oxyanion (anion with Oxygen) would have their compound with more Oxygen end with ______, and their compound with less Oxygen end with ______.
(A) -ous , -ic
(B) -ite , -ate
(C) -ic , -ous
(D) -ate , -ite
(D) -ate , -ite
In older naming systems, elements that can form more than one oxyanion (anion with Oxygen) would have their compound with more Oxygen end with -ate, and their compound with less Oxygen end with -ite.
Examples: NO2 "Nitrite" and NO3 "Nitrate"
CRB True or false? In larger series of oxyanions, the oxyanion with the most Oxygens has the prefix "Hypo-" and the oxyanion with the fewest Oxygens has the prefix "Per-".
False. In larger series of oxyanions, the oxyanion with the LEAST amount of Oxygens has the prefix "Hypo-" and the oxyanion with the MOST Oxygens has the prefix "Per-".
Examples: ClO4 "Perchlorate" and ClO "Hypochlorite"

CRB Define electrolytes, and explain why they are much better at conducting than solid salts.
Electrolytes are solutes that enable solutions (often aqueous) to carry currents. Electrolytes are free to move around in the solution, so the charges can move, unlike in solid salts where charged particles are rigidly set in place by lattice structures.
CRB True or false? Even a very polar, pure uncharged liquid (like water) will not be a good conductor without electrolytes dissolved in it.
True. Even a very polar pure liquid (like water) will not be a good conductor without electrolytes dissolved in it. This is because there are no charges that could freely move around in a pure, uncharged liquid.