4.6 Introduction to Titration
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
22 Terms
An analytical lab technique where a known volume and molarity of a titrant is dispensed from a buret into a flask containing a measured quantity of the analyte
Titration
Can you determine the moles of titrant at equivalent point without doing the titration itself?
Yes
How to determine the moles of titrant at equivalent point without doing the titration itself?
By using the balanced chemical equation specific to the reaction
The equivalence point is when:
The moles of the titrant and moles of analyte are stoichiometrically equivalent
What does moles of titrant and moles of analyte being stoichiometrically equivalent mean?
The two react in a ratio determined by the balanced equation
How to know when the equivalence point is reached?
By using a color change to signal the end point
Three types of common titrations:
Acid-base, redox, and precipitation
What is the titrant in acid-base titrations?
Acid or base
How to determine the end point of a strong acid/base reaction?
By adding an acid-base indicator or by using pH meters
When does an acid-base indicator added to a strong acid/base reaction change color?
At a pH close to 7
When is a color change produced in redox reactions?
When the oxidation state of a metal ion in the reaction changes
What reacts with what in precipitation reactions?
A titrant with ions in the analyte
What happens when the titrant reacts with ions in the analyte?
A precipitation can occur
What signals the endpoint in precipitation titrations?
The formation of a precipitate or its color change
Equipment needed to perform a titration include:
Buret, volumetric pipet or lab balance, Erlenmeyer flask, acid/base indicator or pH meter
What is buret used for in titration?
Keeping track of titrant volumes
What is volumetric pipet used for in titration?
Dispensing a specific volume of the analyte solution
What is lab balance used for in titration?
To mass a sample
Diagram of titration setup equipment:
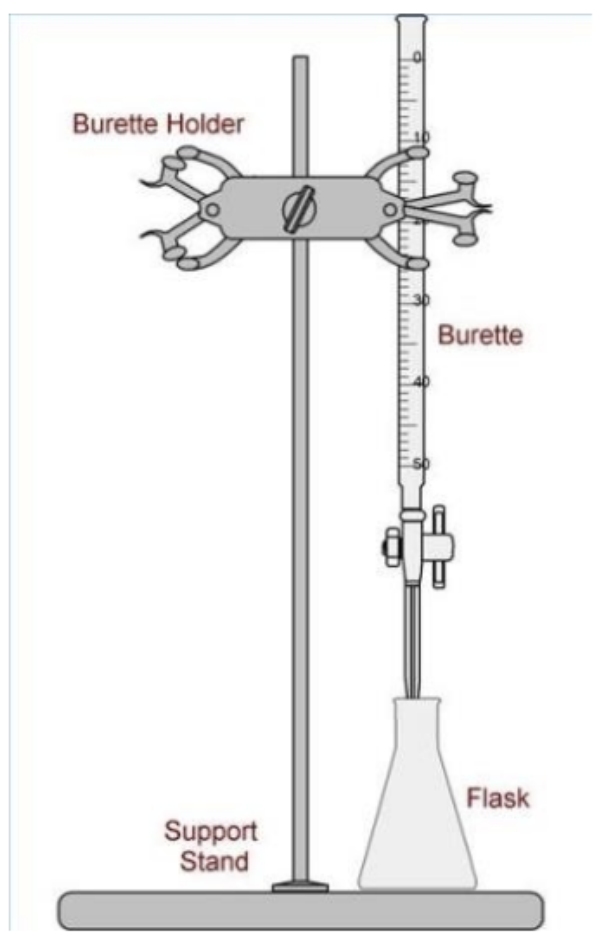
The data needed to be recorded when performing titration include:
Molarity of titrant, initial volume of titrant, final volume of titrant, volume or mass of analyte
If a pH meter is used to monitor an acid/base titration, it’s common to do what?
Plot pH and volume of titrant on a graph
How to determine amount of titrant needed to react fully with analyte from a titration curve?
By dropping a vertical line down at the equivalence point