Bioch 200 Post First Midterm
1/331
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
332 Terms
Fe2+ moved into plane
Conformation change that happens in Hb when transitioning from the T state to the R state.
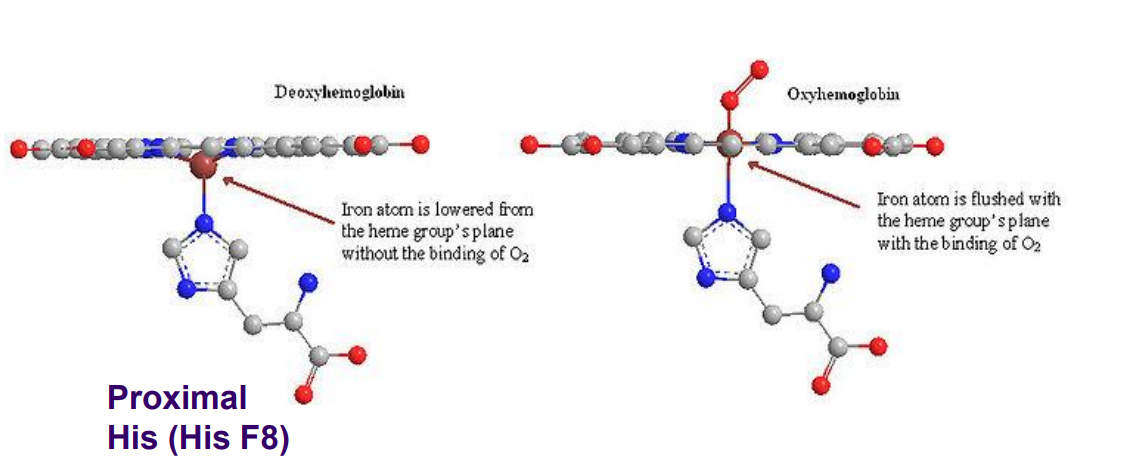
In the plane of the ring
The Fe2+ in Myoglobin is ALWAYS ________ (high affinity)
O2 impact on Hb
Favours the R state (Positive effector)
BPG impact on Hb
Favours the T state (negative effector)
H+ concentration impact on Hb
Favours the T state (negative effector)
CO2 impact on Hb
Favours the T state. Produced in large amounts during cellular activity, indirectly stabilizes.
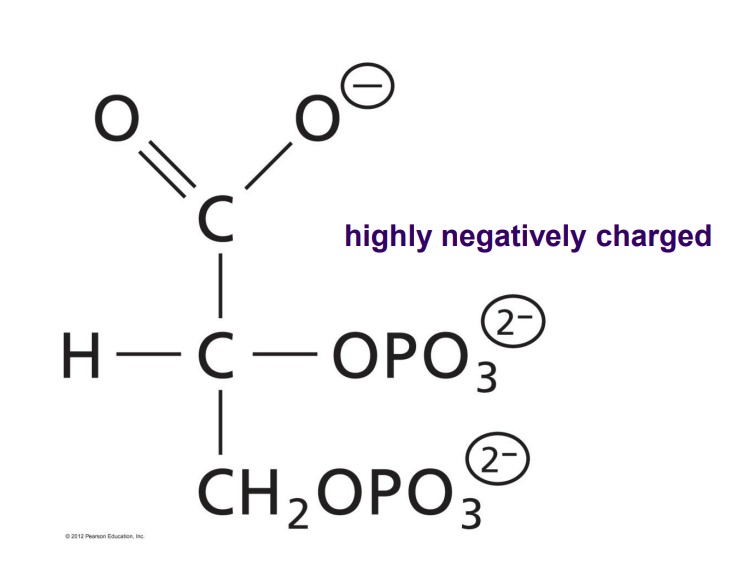
2,3-BPG binds to
Deoxyhemoglobin, right in the center cavity, during the T state. Stabilized by salt bridges. Interacts with 4 central His, 2 Lys and 2 N-terminus’.
Bohr effect
The effect of [H+ ] (pH) on Hb’s O2 binding behaviour
Lowering pH impact on Hb
Means more H+ are present, which protonate side chains (His→HIs+), and enhances BPG binding, reducing O2 binding (Bohr effect)
Blood in lungs (Hb)
Blue* (Low BPG)
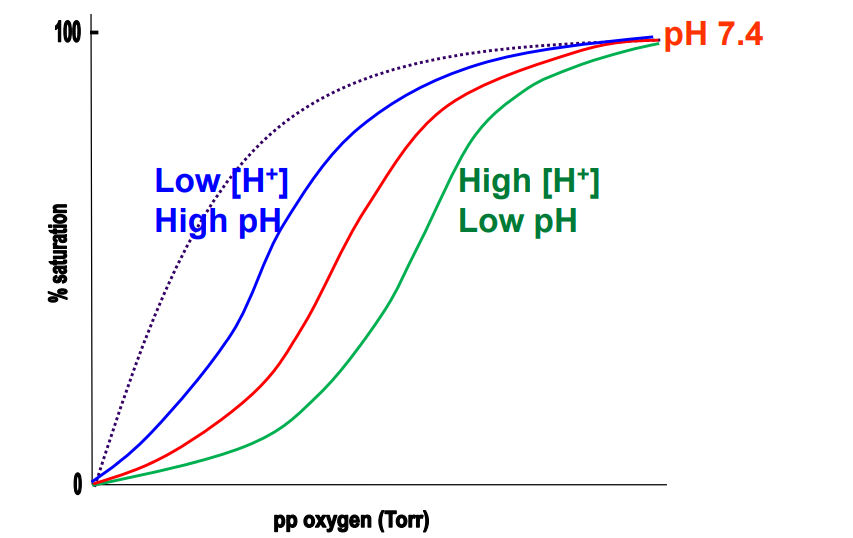
Blood in muscles (Hb)
Green (High BPG)
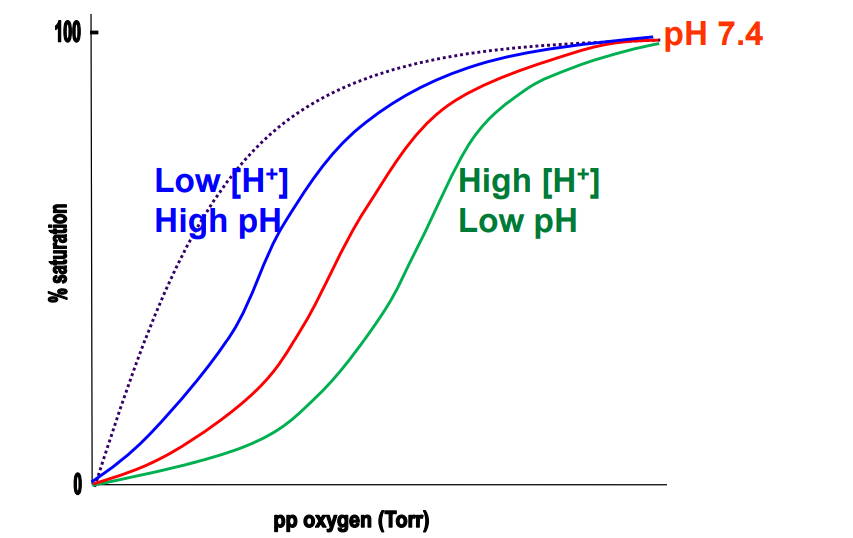
Mb binding curve
Grey - dotted
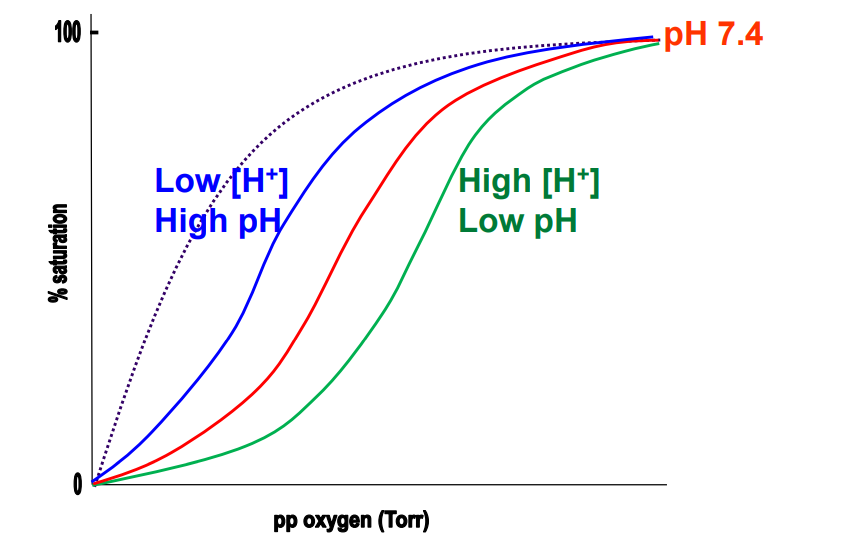
Lungs environment for binding
High proportion of O2, His deprotonated → high pH (relatively ~7.6), Low H+, BPG kicked out as salt bridges break → R state. High O2 binding. Low BPG binding.
Respiring tissues environment for binding
Low proportion of O2, Low pH (relatively), His protonated → High H+, High CO2 proportion, T state is favoured, Salt bridges form between His and BPG. High BPG binding. Low O2 binding.
Sickle Cell Anemia
Caused by replacing a Glu in Hb beta chain with a Val. Critical substitution, (Charged → Hydrophobic). Causes the potential for long fibers of Hb to form, distorting cell shape through hydrophobic interactions.

Fetal Hemoglobin
2 Hb Beta subunits are replaced by gamma subunits. Can bind O2 much better since losing 2 central His (1 from each subunit). Treatment for sickle cell. T state much less stable, Lower BPG binding.
Enzymes
Typically globular proteins, biological catalyst, always regenerated (never consumed during the rxn), some contain prosthetic groups but not required, highly specific.
Advantages of enzymes compared to non-bio catalysts
Higher rxn rates, milder rxn conditions (do not require extreme heat or pH), greater specificity, opportunity for regulation (because of conformational changes in protein structure).
High energy molecules/rxns
Release energy when breaking bonds, can from more stable products, total entropy can increase (ex. from 2 reagents → 3 products), energy released from bonds breaking is more than -25KJ
ATP energy
High energy due to -30KJ energy released when breaking phosphoanhydride bond. Products are more stable (less negative charges in ADP compared to ATP - 3 vs 4, inorganic phosphate resonance stabilized) and entropy increases (3 products formed from 2 reactants)
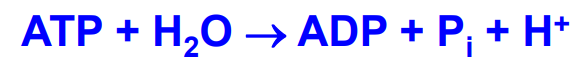
Negative ∆G
Thermodynamically favoured rxn (free energy of products more stable than reactants). Spontaneous. Exergonic. Rxn proceeds in the forward direction
Positive ∆G
Thermodynamically unfavourable (free energy of products less stable than reactants). Rxn proceeds in the reverse direction.
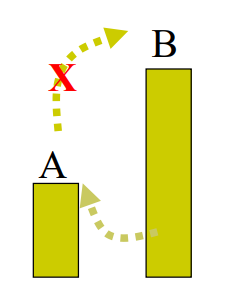
∆G ~ 0
Rxn may be considered reversible (either the forward or reverse could happen at any moment - At Equilibrium)
∆G << 0
Rxn may be considered irreversible (under cellular conditions; rxns in the lab are always reversible).
Reaction rate (SPEED!)
∆G cannot tell us anything about ______ only which direction the rxn will proceed.
Enzymes only impact
Transition state (ΔG‡), which then speeds up the rxn.
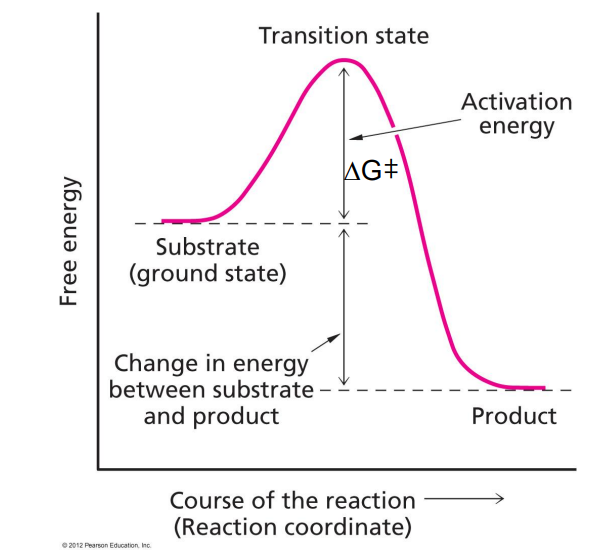
activation energy (Transition state ΔG‡)
This graph illustrates the change in ____ resulting from using an enzyme
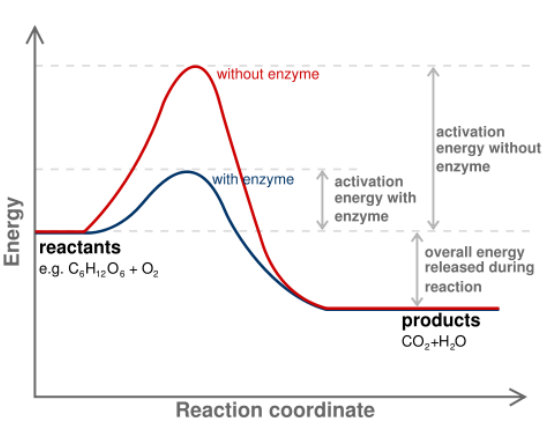
4 ways enzymes can speed up reactions
1. Removing substrates from aqueous solution (desolvation)
2. Proximity and orientation effects
3. Taking part in the reaction mechanism
4. Stabilizing the transition state
Desolvation
Enzymes speed up rxns by removing substrates from water (squeezing water out). Allows for more H-bonds to form. Only some enzymes do this.
Proximity and orientation
Done by all enzymes: Bringing substrates together in the specific way that they need to be orientated in order for the reaction to happen
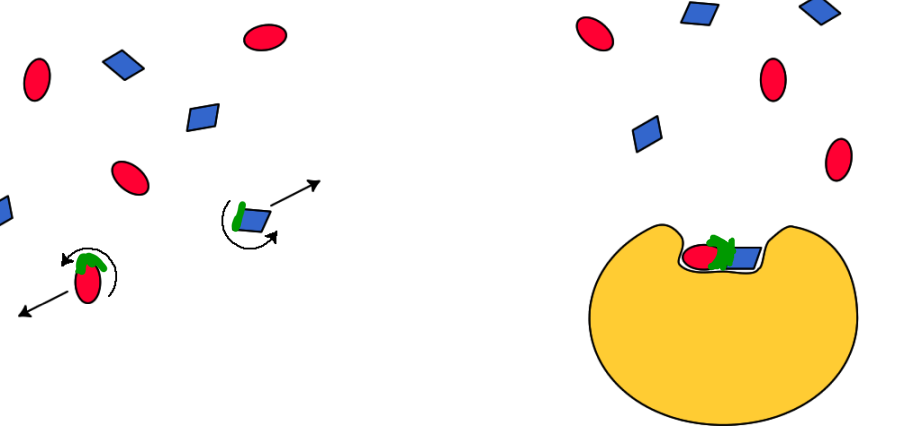
Taking part in the reaction mechanism
2 types: Positioning certain aa side chains in the active site where they can react with the substrates (acid base catalysis) and Provide other chemical substances at the active site (cofactors)
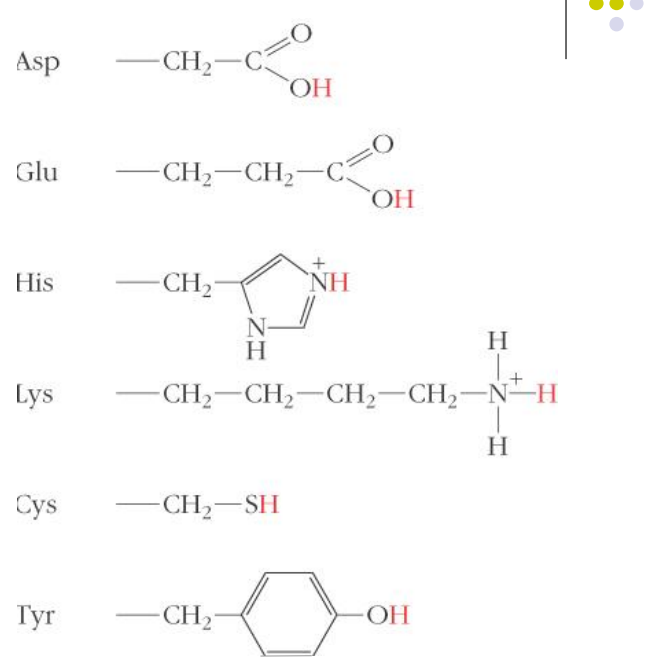
Amino acids in acid-base catalysis
These groups can act as acid or base catalysts, depending on their state of protonation.
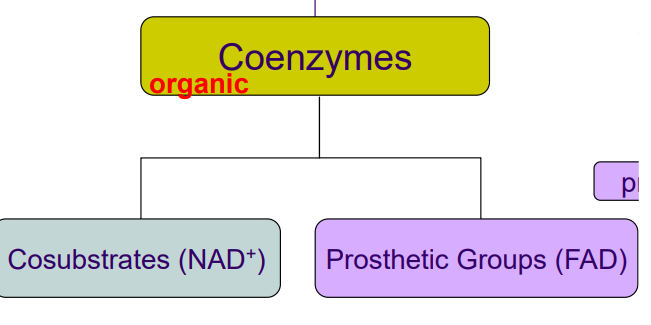
Organic Cofactors
Classified as Coenzymes. Can be tightly bound = Prosthetic groups (FAD) or loosely bound = Cosubstrate (NAD). Must be regenerated after reaction.
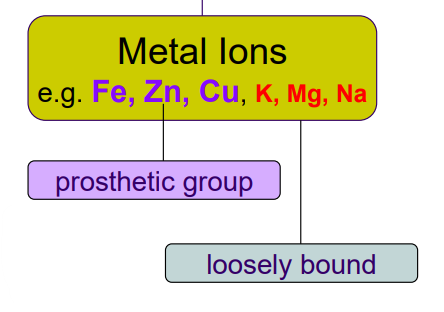
Inorganic Cofactors
Metal ions. Prosthetic = Fe, Zn, Cu.
Loosely bound = K, Mg, Na
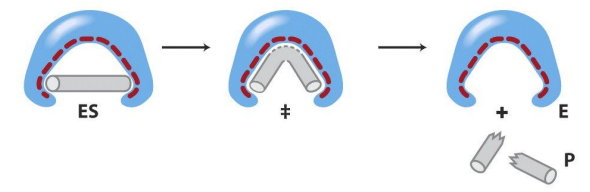
Stabilizing the Transition state
The active site of an enzyme looks more like (binds better) to the transition state than the actual substrate. This lowers the free energy of the transition state significantly (induced fit model)
Lock and key model
Incorrect theory for enzyme function. Perfect fit between substrates and active site. Demonstrates high specificity and affinity, but does not account for the transition state being lowered to any significant degree
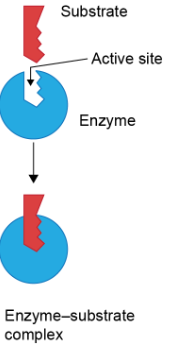
Induced fit model
More correct theory for enzyme function. Active site looks more like the transition state, causing the energy of the state to be lowered significantly, speeding up the reaction.
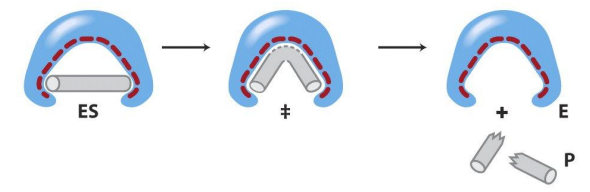
Rational drug design
Drugs today are designed to look like the transition state, allowing for increased affinity when interacting with enzymes (better fit, more like the lock and key model).
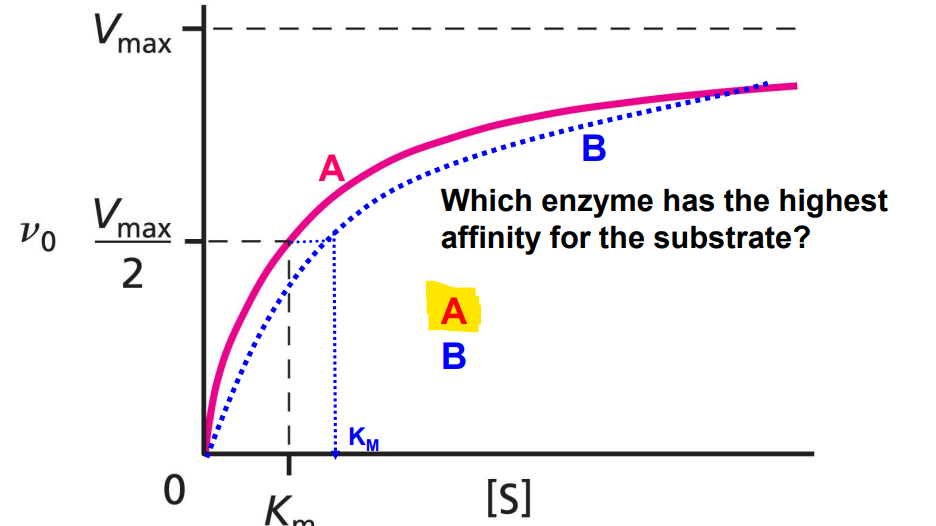
Km
Used for enzymes; when the max reaction velocity is at exactly half (Vmax/2). Lower Km = higher affinity for a given substrate (closer to y-axis).
Enzyme mechanisms of regulation
Fast: Competitive inhibition, Allostery (ionic signals) and reversible covalent modification (phosphorylation of serine, threonine and tyrosine).
Slower: Regulation of gene expression and changes in subcellular localization.
Enzymes may be regulated by multiple/all of these mechanisms.
Competitive Inhibition
Inhibitors are similar to the substrate in shape and size but differ chemically in such a way that they cannot react; competition over the same active site. ex) CO for O2 in Hb
True Km
Looking at only the enzyme and its intended substrate.
Apparent Km
Impacted by competitive inhibitors. Bigger than true Km, provided inhibitors are present (lower relative affinity, curve shifted to the right, further from y-axis).
Enzyme binding
Is only covalent (for both substrates and inhibitors). Remember that it is only a temporary connection (enzyme must release substrate eventually)
Better
Transition analog inhibitors bind ____ compared to inhibitors modeled after the intended substrate
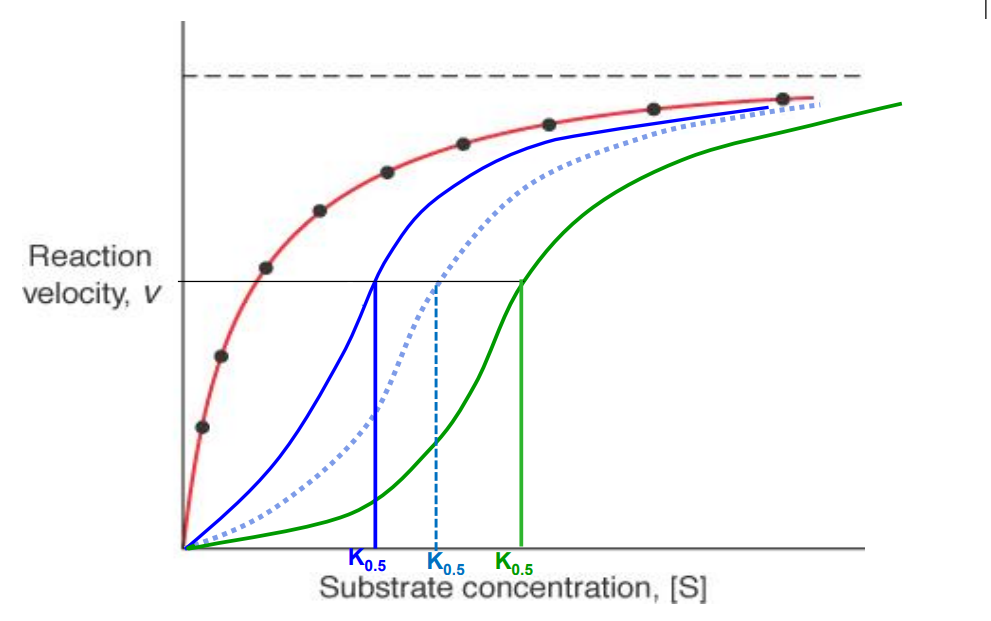
Allosteric enzymes
Have sigmoidal shaped rxn velocity vs substrate curves. Think of 40 structure, positive cooperation, homoallostery
K0.5
Used for allosteric enzymes rather than Km
Enzyme homoallostery
When the binding of the substrate at one active site increases the enzymes ability to bind with another substrate at a seperate active site.
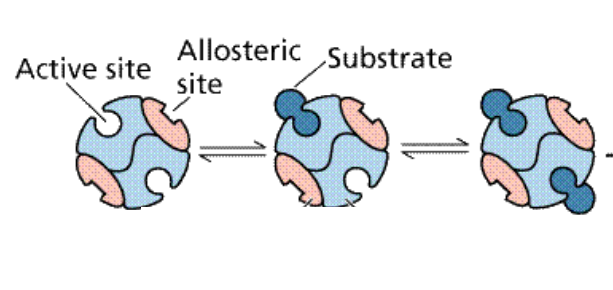
Enzyme heteroallostery
When an activator or inhibitor binds to an enzyme somewhere other than the active site, impacting the enzymes ability to bind with the substrate (can be positive or negative)
Hyperbolic curve (non allosteric enzyme)
Red curve
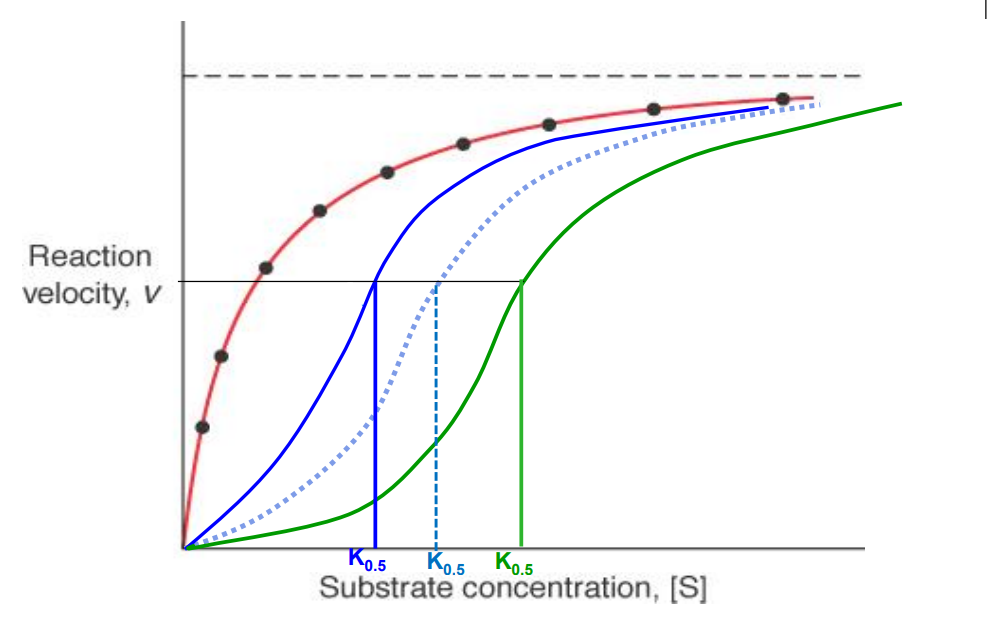
Allosteric enzyme activator present
Blue curve (K0.5 decreases)
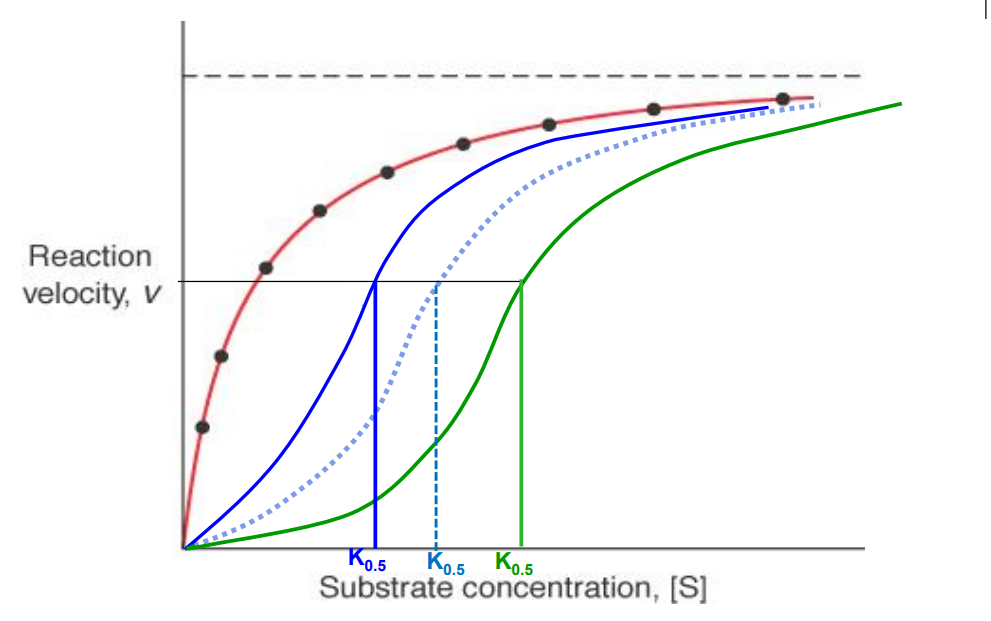
Allosteric enzyme inhibitor present
Green curve (K0.5 increases)
Protein Kinase
Enzyme which phosphorylates only proteins (membrane channels, receptors, transporters, other enzymes, etc) via ATP → ADP.
Ser, Thr, Tyr
Main amino acids (when apart of a protein) that become phosphorylated by protein kinase. OH group becomes phosphate group via the oxygen. Important for regulation
Protein Phosphotase
Enzyme that dephosphorylates proteins only. Occurs via hydrolysis (H2O put in) and releases an inorganic phosphate.
Addition of phosphates to enzymes
Increases size, increase in polarity/hydrophilicity, addition of two negative charges and the capability of making multiple new H bonds.
Changing the 3d shape
Phosphorylation impacts enzymes activity by
Reversible
Phosphorylation of enzymes is
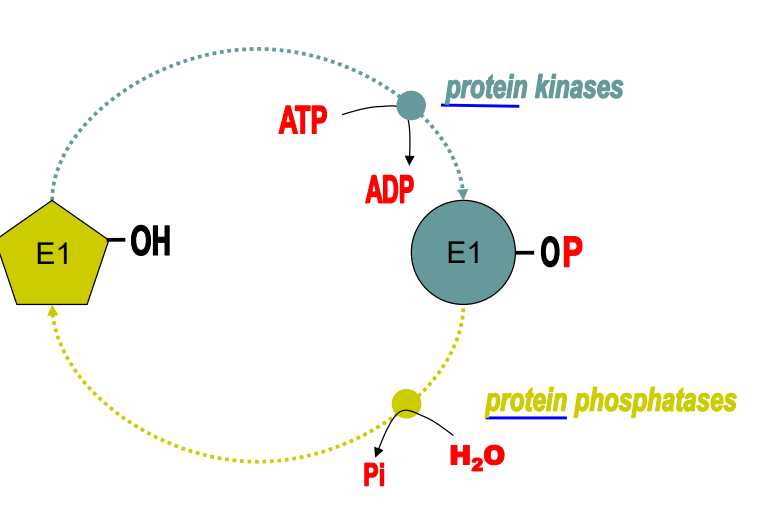
Increase or decrease activity
Phosphorylation of enzymes can __________, depending on the enzyme and conditions.
Fatty acids
Long Hydrocarbon chained carboxylic acids. Amphipathic, deprotonated at pH 7 (pKa ~5)
Saturated FA
Only single bonds (alkane)
Unsaturated FA
1 or more (poly) double bonds present. Cis more common than trans
Cis FA
Big kink
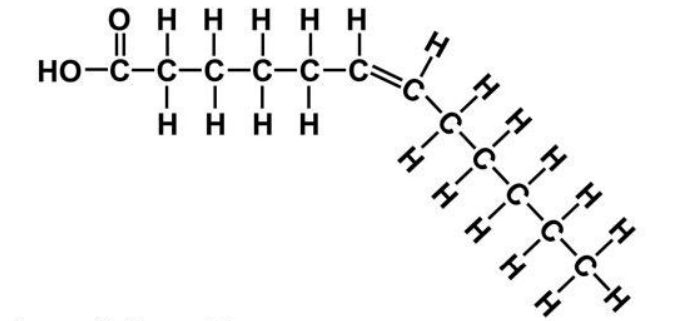
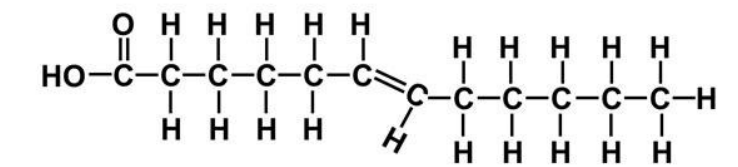
Trans FA
Lesser kink
FA alpha carbon
The carbon attached to the carboxyl group carbon (technically the 2nd carbon in the molecule). The first carbon to make up the hydrocarbon chain.
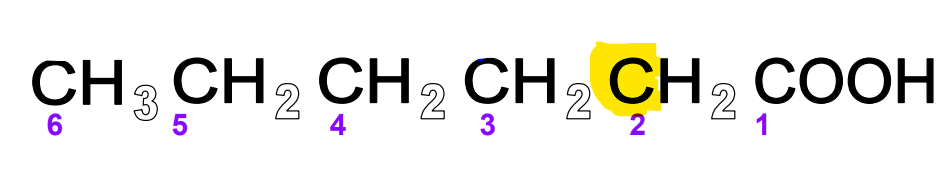
FA beta carbon
The carbon next to the alpha carbon in the hydrocarbon chain
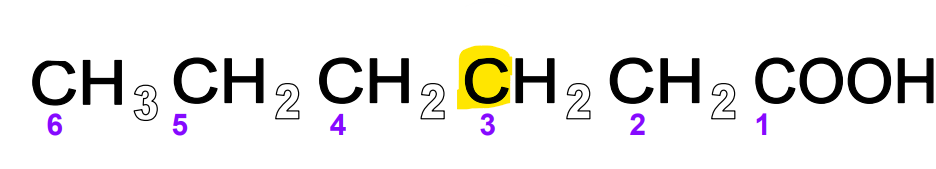
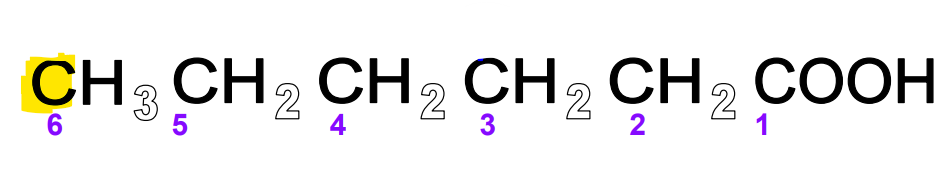
FA omega carbon
The carbon at the end of the hydrocarbon chain
Unsaturation causes
Kinks in the FA, meaning they cannot be packed together as tightly as normal.
High melting point
Longer chained, saturated FA
Low melting point
Shorter chained, unsaturated FA
Trans FA being less kinked
Have a slightly higher melting point than cis FA.
Melting point
The energy required to separate individual molecules in a solid from one another
Beef fat
Example of a fat that is solid at room temp; therefore can be predicted to contain less double bonds (more saturated)
Oil
Example of a fat that is liquid at room temp; therefore can be predicted to contain more double bonds (more unsaturated)
Triacylglycerides (TAG)
How FA are stored; 1-3 FA chains (acyl) attached to a glycerol molecule via an ester bond
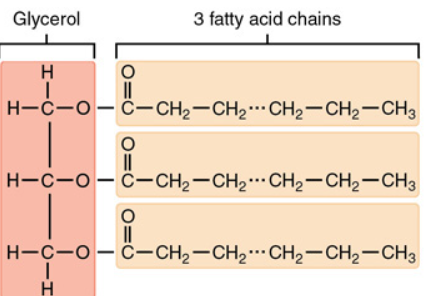
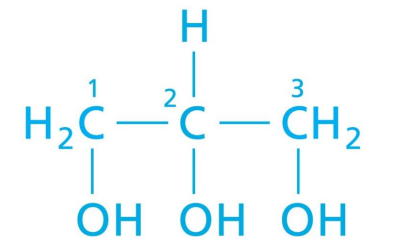
Glycerol
Has the potential to carry 3 FA chains at the hydroxyl group sites. Forms new bonds with FA via dehydration rxn.
Non-amphipathic
When FA combine with glycerol they become _______, due to the lose of the polar hydroxyl group. (They form blobs in water - no recognizable/organized structure)
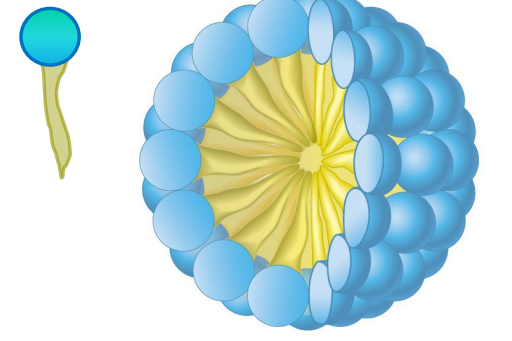
Micelle
FA naturally form a micelle in water, because they are amphipathic (polar hydroxyl head, non-polar carbon chain)
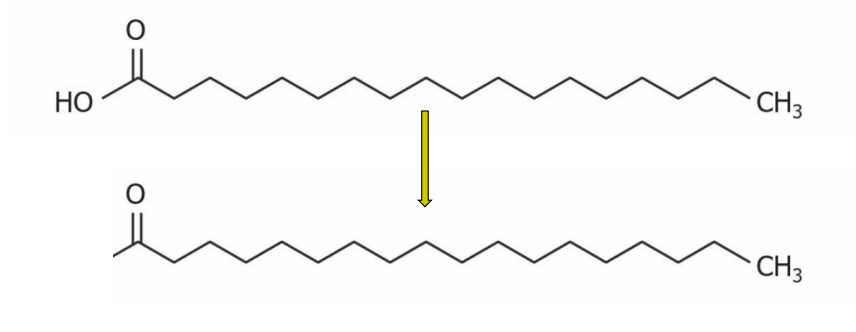
Acyl group
Formed (in the case of FA), when the carboxyl group combines with the glycerol hydroxyl group. The OH on the FA is lost, leaving behind a double bonded O and alkyl chain (A in TAG)
Simple TAG
3 identical FA attached to a glycerol; not very common as most are mixed
Sphingolipids
Can (but not necessarily must) have a very large polar head group compared to glycerophospholipids
Glycerophospholipids (GPL)
Contains glycerol in the center. Attached at the top is a polar phosphate containing head group. To the remaining 2 OH positions on the glycerol, acyl FA tails can attach.
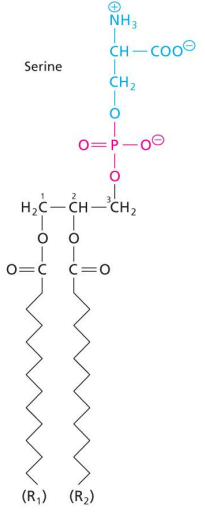
Potential for glycerophospholipid variation
Differing in head groups
differing in the lengths and saturation of acyl tails
Cholesterol
Important membrane lipid. Weakly amphipathic. Largely contained within the membrane (not significantly exposed to outer environment). At high temps maintains rigidity of membrane, at low temps prevents lipids from clustering/stiffening.

Bacteria
Cholesterol is not found in _____
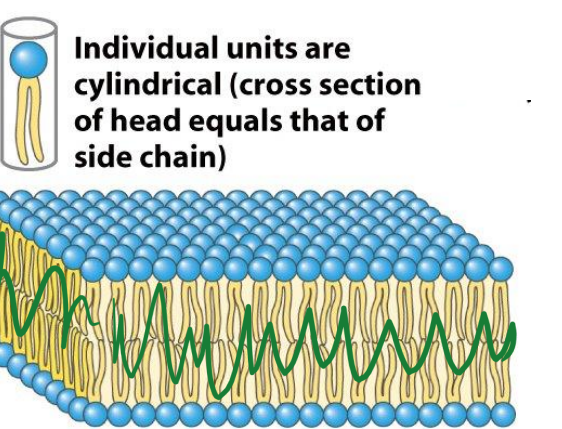
Bilayers
GPL naturally form _____ in water
Does not
Cholesterol _____ form membranes by itself.
Hydroxyl on cholesterol
Associates with the polar headgroup of GPL, when in the bilayer.
Completely hydrophobic
A FA can attach to the OH in cholesterol which makes it
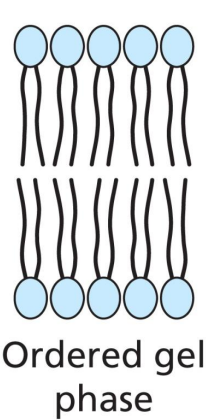
At low temps
Lipid bilayer exists as an ordered gel phase. Without the presence of cholesterol, the acyl chains can pack tightly together, in Van der Waal contact
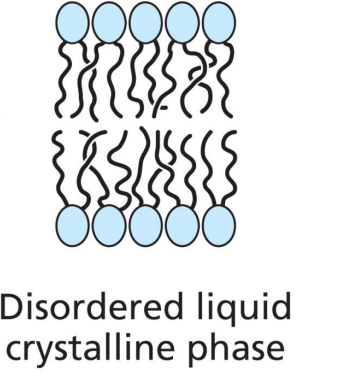
At high temps
Lipid bilayer exists as an disordered liquid crystalline phase. Molecules and chains move freely and rapidly
Melting (transition) temp
In a lipid bilayer, described as the temperature which transition from an ordered gel phase to a more fluid (liquid) crystalline phase occurs
Factors impacting membrane fluidity
Temperature
Lipid composition: (Cholesterol presence, FA chain length, Degree of saturation)
Higher Transition temp
membranes with longer, more saturated chains
Lower Transition temp
membranes with shorter, more unsaturated chains
Diversity of GLP’s present in membrane
Why is transition temp for biological membranes typically not sharp
Membrane adjustments for a colder environment
The membrane will incorporate more unsaturated FA and shorter FA (in GLP’s), in order to increase fluidity
Membrane adjustments for a warmer environment
The membrane will incorporate more saturated FA and longer FA (in GLP’s) in order to increase rigidity
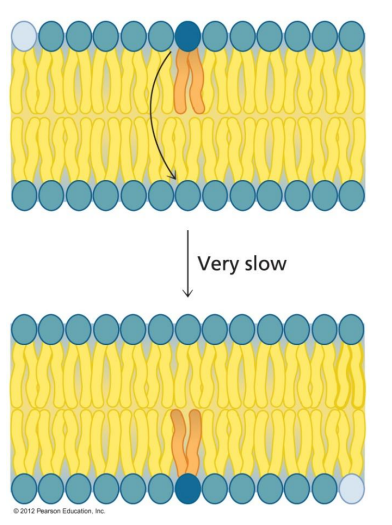
Laterally (side to side)
Lipids can move freely and rapidly in the bilayer, but only _____
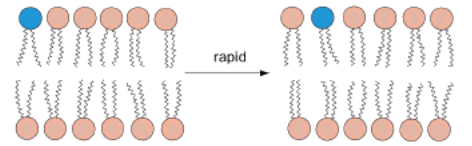
Transverse diffusion
When a lipid moves across from one leaflet in the membrane to the other. Takes a significant amount of energy (polar head interacting with non polar inner) and is very slow. Utilizes flippases and floppases enzymes.