Medicinal Chemistry Exam 2
1/168
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
169 Terms
what are the 3 steps in drug development?
1) discovery
2) optimization
3) development
what is the discovery step of drug development?
identify and produce new active substances (lead compounds)
what is a lead compound?
- new active substance that has pharmacological or biological significance
- compound that acts as the starting point for drug design and development
where can lead compounds originate from? (2)
- synthetic organic chemistry
- natural sources of biotech processes
what is the optimization step of drug development?
synthetic modification of the lead structure to improve potency, selectivity, and reduce toxicity
the characteristics of the optimization step revolve around what analysis?
structure-activity relationship (SAR)
what is the development step of drug development? (2)
- optimization of the SYNTHETIC ROUTE FOR BULK PRODUCTION
- modification of active substance to be used clinically (properties like solubility, palatability, reduction of pain at injection site, etc)
what are the 3 early events in medicinal chemistry drug development?
1) understand the disease
2) identify the molecular target
3) find a lead compound
what do the 3 early events in drug development all do?
develop an assay
what are the 3 later events in medicinal chemistry drug development?
1) optimize INTERACTIONS of drug with target
2) optimize PK properties (ADME)
3) optimize SAFETY
what do the 3 later events in drug development all do?
optimize formulation properties
what is a pharmacophore?
part or parts of a molecule that are the key parts of the molecule
what is an orphan drug?
drug developed for disease that affects less than 200,000 people and does not make a large profit
drug discovery and development process requires what 2 official applications from the FDA?
IND and NDA
ex of possible drug targets?
lipids, proteins, nucleic acids, and carbs
ex of mechanisms of drug action?
enzymes, receptors, ion channels, transporters, and DNA
2 types of target selectivity? ex of each?
1) b/w species (antibacterial, antifungal, and antiviral agents)
2) within the body (selectivity b/w enzymes, receptors, tissues)
what is a bioassay?
measurement of the concentration or potency of a substance by its effect on living cells or tissues.
what comes first: bioassay or lead compound
bioassay
put the following in order:
- identify target
- identify lead compound
- identify disease
- file NDA
- bioassay
- file IND
- develop animal testing
- start human clinical trials
- disease
- target
- bioassay
- lead compound
- animal testing
- IND
- human clinical trials
- NDA
drug testing is done in what 2 stages? what are they?
1) lead generation (find a compound that binds)
2) lead optimization (find a compound that binds better)
what is important for both stages of drug testing?
chemical similarity
tests are required to find lead and optimized drug compounds. what are the 3 tests that can be done? explain each
1) in vivo (in live animal or humans)
2) in vitro (separate enzyme in test tubes or in certain organ/tissues but not in live animal or human)
3) in silico (on computer)
pro and con of in vivo tests?
- pro: identify possible side effects
- con: difficult to rationalize b/c of number of factors
what can you measure with in vivo tests?
the TI
pro and con of in vitro tests?
- pro: easier to rationalize
- con: does NOT demonstrate clincial effect, possible side effects, or pro drug
what can you and can you not measure with in vitro tests?
the INTERACTIONS with a target (NOT THE ABILITY)
enzyme inhibition tests help us identify what?
competitive or noncompetitive inhibition
testing with receptors helps us identify what?
affinity (strength of binding), efficacy (max biochemical effect), and potency (concen. required to produce 50% max effect)
when developing a lead compound generation, what is important in the process in terms of the compounds themselves?
chemical diversity with synthesized combinatorial "libraries" of compounds
purpose of the chemical "libraries" of compounds for chemical diversity in developing a lead compound?
idea is to cover as much of the "CHEMICAL SPACE" as possible
3 steps in identifying a lead compound?
1) starting point
2) design
3) test
what can be used for the starting point of developing a lead compound?
finding a compound that can be therapeutically useful like an active principle
what can be used for the design of developing a lead compound?
NMR (molecular modeling) or screening
what can be used for the test of developing a lead compound?
identifying a suitable test in either test tube, animal, or computer
what is an active principle?
- compound that is often used as the lead compound
- it is isolated from a natural extract and is principally responsible for the extract's pharmacological activity
why are active principles useful?
b/c they are the single molecule responsible for pharmacological activity so by finding the active ingredient, you can make a drug out of it
___ = the "best chemist"
nature--rich source of complex and highly varied structures that are unlikely to be synthesized
when a lead is identified, what is the next stage?
find compounds that are similar to it
what is the step called in which compounds similar to the lead compound are searched for?
lead optimization
3 sources of lead compounds?
1) natural world
2) synthetic world
3) virtual world
what is included in the synthetic world of finding lead compounds? (2)
1) chemical synthesis
2) combinatorial and parallel synthesis
as soon as a lead compound is found, what must be filed?
patent to secure the 20 year life and avoid competition
after a lead compound is found, and similar compounds to the lead compound are found, what is done to these discoveries?
medicinal chemists make specific changes to the lead compound with SAR (ex: improving binding affinity)
what is ethnobotany?
the study of traditonal use of plants
what did each of the following lead compounds from the natural world give us?
1) opium
2) cinchona bark
3) artemisinin
4) yew tree
5) willow tree bark
6) cyanobacterium
1) opium - morphine and opioid analgesics
2) cinchona bark - quinine anti-malarials
3) artemisinin - more effective anti-malarial than quinine b/c of 2 oxygens
4) yew tree - taxol anti-cancer drugs
5) willow tree bark - anti-inflammatory agents like aspirin
6) cyanobacterium - anti-tumor
-
-
what is de novo drug design?
drug design on computers to optimize design of drug to fit into receptor
what is a scaffold in a lead compound?
very stable part of structure that reinforces the skeleton of a molecule but is NOT RESPONSIBLE FOR BIOLOGICAL ACTIVITY
how is lead optimization different from lead discovery?
in lead compound discovery, we WANT DIVERSITY but in lead optimization, we're looking for SIMILARITIES
3 pathways in which lead compounds are discovered?
1) screening
2) chemical modification
3) rational
what 3 pathways are part of the screening pathway of discovering lead compounds? (3)
1) random screening: compound tested in bioassay w/o regard to structure
2) non-random screening: compound tested in bioassay w/ regard to structure resemblance
3) combinatorial approach: automated synthesis of mixtures w/ different structures to see if useful
pro and con to random screening pathway in discovering a lead compound?
- pro: no bias, can just test structures
- con: very low success rate
what is the chemical modification pathway of discovering lead compounds?
use functional group modifications and drug metabolites to work on side effects produced from another molecule
functional group modification can be used in the discovery of lead compounds for what purpose?
to enhance or inhibit a side effect produced by a lead compound already
what is the rational pathway of discovering lead compounds?
start with natural ligand for receptor, then natural substrate for enzyme, and then computer aided (CADD) knowledge of target structure (in silico)
what lead compound molecule was found by serendipity?
monoamine oxidases (MAO)
how does MAO work?
it's an enzyme that deaminates monoamines (NE, serotonin, dopamine) and functions in the NS
MAO inhibitors should not be used with what?
cheese, wine, beer, yeast (can lead to increased BP)
other lead compounds found with serendipity?
penicillan, libirium, viagra
what are "me too" drugs?
- patentable modifications of existing drugs on the market
- aka not very original but makes a lot of money; initially a company finds a molecule and others make it with slight modifications that aren't on the patent
why is a lead compound not the final point of drug design?
it is just the starting point b/c binding may not be strong and it may be toxic
purpose of SAR?
identify which functional groups are important for binding and/or activity
what does in vitro testing test for in SAR?
binding abilities with target
what does in vivo testing test for in SAR?
target binding interactions and/or PK properties
if in vitro/in vivo activity is unaffected by the addition of a group, it implies that a group is not important
in vivo
if in vitro activity drops, it means that the group was important/not important for binding/interactions
group was IMPORTANT for BINDING part of the pharmacophore
analogue vs lead compound?
analogous compound has core structure but is not as diversified as the lead compound
analogue of a lead compound allows for the identification of what 2 things?
- allows identification of important groups involved in binding
- allows identification of the pharmacophore
pharmacophore vs auxophore vs toxiphore
- auxophore: not important in activity of molecule
- toxiphore: part of molecule that generates toxicity
SAR on alcohols: purpose and what if you replaced the OH to make it an ester, ether, or alkane
- purpose: see if an alc is part of the pharmacophore or not (meaning is it even important to the effect produced by the molecule)
- how to do it:
OH can be a HBA or HBD and if you change the OH group to an ether, ester, or alkane and there was NO CHANGE in the drug's activity, then it means the OH was not part of the pharmacophore (even after changing the group it can still be a HBD or HBA)
SAR on alcohols: what if you replaced the OH with a methyl
OH can e a HBA or HBD and if you change the OH group to a methyl, then the methyl cannot be HBA or HBD b/c of steric hindrance so there is a CHANGE in the drug's activity (meaning that HBA/HBD activity is important for the drug to work)
summary of SAR and what it tells us when we apply it to a lead compound
- if changing a functional group changes the activity of the molecule, then that group is important and possible part of the pharamcophore
- if changing a functional group does not change the activity of the molecule, then that group is not important and not part of the pharmacophore
understand SAR in each functional group
3 ways to express a pharmacophore?
2D pharmacophore
3D pharmacophore
3D bonding type pharmacophore
what does the 2D pharmacophore define?
the minimum skeleton connecting important binding groups
what does the 3D pharmacophore define?
defines relative positions in space of important binding groups using ANGLES AND DISTANCE
what does 3D bonding type pharmacophore define?
defines relative positions ins pace of the IMPORTANT BINDING INTERACTIONS which are required for activity
what is an important point of consideration in the 3D bonding type pharmacophore?
the active conformation
identification of the ___ is required in order to identify the 3D pharmacophore
the active conformation
what is the active conformation?
the conformation adopted by the drug when it binds to its target and produces a desired effect
what analysis helps us find possible conformations?
conformational analysis
how can the alkyl substituents be varied to optimize drug-target interaction?
by varying the length and bulk of an alkyl chain to optimize the interactions in a hydrophobic pocket
what happens in varying the aryl substituents to optimize the drug-target interaction?
vary the location of the substituents and their H bonds on an aromatic to affect the strength of the intermolecular H bond interactions
what happens in varying the extension to optimize drug-target interactions?
make extra functionality to explore target binding site for further binding regions and achieve additional binding interactions
what happens in varying the chain extensions/contractions to optimize drug-target interactions?
unsure what the best distance is so add linkers to enhance interaction
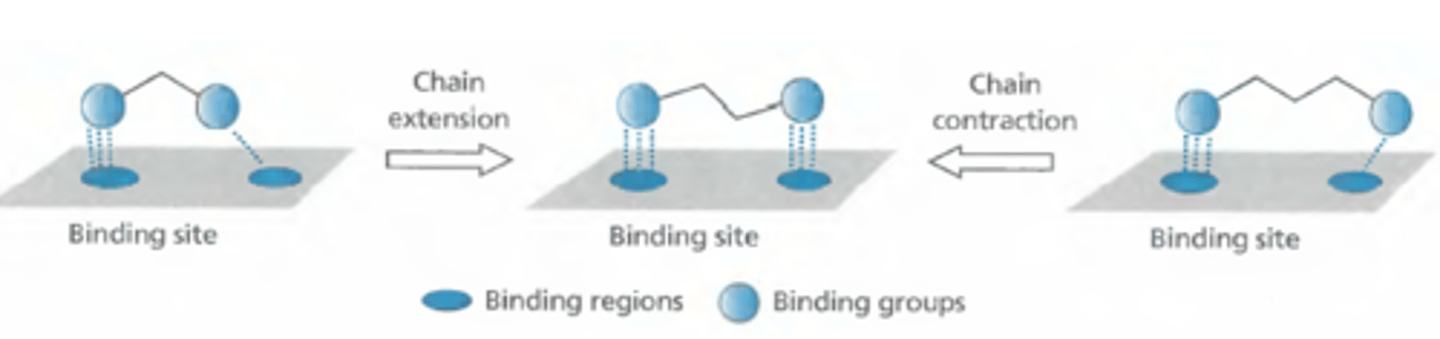
what happens in varying the ring extensions/contractions to optimize drug-target interactions?
add ring to improve overlap of binding groups to increase binding regions activity
what happens in varying the ring variation to optimize drug-target interactions?
2 types:
1) replaces aromatic rings with other rings (usually done for patent reasons) to find strongest binding affinity
ex: making a 6 membered ring into a 5 membered ring
2) ring fusion in which 2 things are made into 1
what happens in varying the 2 isosteres to optimize drug-target interactions?
1) isosteres: if the steric activities and valence of functional group are similar, they are isosteres and can be interchanged to affect the binding and stability
- ex: OH replaced with SH, NH2, CH3
2) bioisosteres: if the biological activities of functional groups are similar, they are bioisosteres and can be interchanged to affect the biolical properties
what are bio-isosteres?
substituents or groups that produce similar biological properties
classical vs nonclassical bio-isosteres?
- classical: atoms, ions, and molecules in which the peripheral layers of electrons can be considered identical
- nonclassical: do not have the same number of atoms but still produce similar biological effects
- ex of nonclassical: replacing sulfonamide for a phenol
which one is STRUCTURALLY similar: bio-isostere or isostere
isostere
which one is structurally not similar but ACTIVITY similar: bio-isostere or isostere
bio-isostere
what happens in varying the simplification to optimize drug-target interactions?
retain pharmacophore and remove unnecessary functional groups
con of simplification of drug to optimize drug-target interactions?
- may OVERSIMPLIFY resulting in decreased activity and selectivity
- simpler molecules have MORE CONFORMATIONS so bioactivity will decrease
- more likely to interact with more than one target site
- may result in INCREASED SIDE EFFECTS
what happens in varying rigidification to optimize drug-target interactions? what is used to rigidify?
- conformations are a result of flexible compounds so rigidifying the molecule can limit conformations and increase activity b/c there's more of a chance to get the desired conformation
- rigidify with rings, rigid functional groups and double bonds, amides, and steric blockers
pharmacokinetics deals with what characteristic of a drug?
it's ADME and whether or not it will reach an active site
can a drug that is active in vitro be inactive in vivo?
yes
factors affecting whether a drug will reach its target has to do with pharmacokinetics/dynamics
kinetics (body affects the drug)
drug design should consider what 2 things?
PK and binding interactions