biochem final study guide
1/59
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
60 Terms
There are two major types of metabolism (regardless of oxygen levels). Name them.
Anabolism & Catabolism
The three primitive functions of metabolism are to:
1) Generate chemical energy
2) Transport electrons
3) Synthesize macromolecules needed for growth and survival
For every molecule of glucose that enters glycolysis, how many acetyl-CoA molecules are produced that enter into the TCA (Krebs, citric acid cycle, tricarboxylic acid cycle)?
2
Enzymes called phosphatases function to remove a phosphate from a substrate (be it protein or small molecule) whereas _________ are known to do the opposite.
Kinases
Name the 3 irreversible steps in glycolysis.
Phosphofructokinase-1, pyruvate kinase, hexokinase (PFK1, PK, HK)
Glycolysis can be divided broadly into 2 sections called the investment/preparatory and payoff phases. Why do we call it the investment phase?
Because we need to put energy in (using ATP) in order to break glucose into 2x molecules of pyruvate
T/F: The reaction catalyzed by GAPDH (glyceraldehyde-3-phosphate dehydrogenase) generates one molecule of NADH from NAD+ and Pi (inorganic phosphate)
True
Which two reactions in glycolysis produce ATP?
Pyruvate kinase (PK) and Phosphoglycerate kinase (PGK)
Phosphoglucose isomerase (PGI) converts the six membered ring of glucose into the five membered ring of fructose. How much energy input does this transformation require?
None
2-phosphoglycerate (2PG) is converted to phosphoenolpyruvate (PEP) via what kind of reaction catalyzed by the enzyme enolase?
Dehydration (loss of water)
T/F: The products of the enzyme aldolase are G3P/GAP (glyceraldehyde-3-phosphate) and DHAP (dihydroxyacetone phosphate).
True
Why do some cells undergo fermentation, either producing lactate or ethanol from pyruvate, even though it is less efficient than other metabolic processes?
To regenerate NAD+ quickly and make ATP equivalents under anaerobic conditions
In yeast anaerobic fermentation of pyruvate to acetaldehyde to ethanol, the enzyme pyruvate carboxylase uses the cofactor TPP (thiamine diphosphate) to catalyze what kind of reaction?
Decarboxylation
T/F: While technically all cells in the body have the ability to generate glucose via gluconeogenesis, muscle tissue is the primary site of glucose production helping it during exercise.
False
Glycolysis and gluconeogenesis are examples of opposing processes in cells. If both were happening at the same time this would be an example of:
Futile cycle
T/F: Like in animals, plants and fungi use starch as the primary storage form of glucose.
False
Which 2 enzymes are essential for bypassing two of the irreversible reactions in glycolysis?
Fructose-6-phosphatase (F6Pase) & Glucose-6-phosphatase (G6Pase)
What is one main reason why pyruvate can't be directly converted back into phosphoenolpyruvate (PEP) in gluconeogenesis?
Pyruvate is shuttled into the mitochondria as soon as it's made
How much net energy does it require to reform glucose from pyruvate in gluconeogenesis?
6 ATP equivalents
Insulin drives glucose uptake from the blood and into cells. This function of insulin as a hormone stimulates activation of all the following intracellular processes except:
- Gluconeogenesis
- Glycolysis
- Glycogen synthesis
- Lipid biosynthesis
Gluconeogenesis
Glycogen is a collection of highly branched glucose molecules used for energy storage, however it cannot be easily transported/shared without being broken down first. Which is the primary site of glycogen storage?
Muscles & Liver
In pyruvate dehydrogenase (PDH), which cofactor is found in the E3 subunit and functions to regenerate the E2 cofactor?
FAD/FADH2
Pyruvate dehydrogenase (PDH) has three subunits (E1, E2, and E3). Which subunit is the site where CO2 is released?
E1
Oxidative decarboxylation is catalyzed by pyruvate dehydrogenase (PDH) and pyruvate decarboxylase (fermentation) which share the same cofactor. Name the cofactor.
TPP (thiamine diphosphate)
Glycogen synthase and glycogen phosphorylase enzymes are regulated through reversible protein phosphorylation in a process referred to as _________.
Reciprocal control
Low blood glucose stimulates glucagon production via the pancreas which can lead to activation of protein kinase A (PKA) which phosphorylates both pyruvate kinase (PK) and phosphofructokinase-2 (PFK2). What impact does glucagon signaling have on overall rate of glycolysis?
Decrease
Pyruvate dehydrogenase (PDH) is 1) positively and 2) negatively regulated directly by high levels of which compounds?
1) NAD+
2) NADH
T/F: Uneven (odd) chain fatty acids enter β (beta) oxidation after first being reduced by FADH2 (hence they make fewer ATP equivalents than even numbered fats).
False
What is propionyl-CoA?
Biproduct of uneven (odd) chain fatty acid catabolism
The 'Mad Hatter' character from the Alice in Wonderland stories was inspired by hat makers suffering from the effects of chronic mercury poisoning (neurological/behavioral issues). What critical metabolic enzyme (among many other cellular functions) does mercury impact?
Pyruvate dehydrogenase (PDH)
Both pyruvate dehydrogenase (PDH) and isocitrate dehydrogenase (IDH) are positively regulated indirectly by Ca2+. How?
Ca2+-dependent dephosphorylation of the active site which opens it up to bind substrate
If citrate levels are suddenly sufficiently high in cells, what is the expected change to both the rate of glycolysis and rate of lipid biosynthesis?
Rate of glycolysis decrease & rate of lipid biosynthesis increase
While TCA cycle intermediates can be siphoned off toward the production of other important biomolecules (i.e. nucleotides, amino acids, lipids) these other molecules can also be reversibly broken down to reform critical TCA intermediates like α-ketoglutarate in a process known as _________.
Anaplerosis
α-ketoglutarate dehydrogenase (α-KGDH) possesses E1, E2, and E3 subunits similar to pyruvate dehydrogenase (PDH). α-KGDH is also therefore regulated by NADH & what other molecule through product inhibition?
Succinyl-CoA
Fats are stored primarily in what form in the body?
Triacylglycerides
When ATP levels in the cell are low, all of the following processes would be stimulated except:
- Fatty acid biosynthesis
- Glycolysis
- Fatty acid beta-oxidation
- TCA cycle
- Pyruvate dehydrogenase
- Electron transport chain
Fatty acid biosynthesis
Hormone stimulated lipases do what important reaction in metabolism?
Use water to cleave triacylglycerides into glycerol and free fatty acids
At what point in metabolism would the compound glycerol-3-phosphate come into play?
Produced in the oxidation of triacylglycerides
The oxidation of fatty acids is referred to as β (beta) oxidation because:
The carbon-carbon single bonds in the acyl chain of fatty acids are named based on location relative to the carboxylic acid, and oxidative cleavage of fats occurs most commonly after the β (beta) carbon
Per molecule of glucose, the core TCA cycle (no other pathways) produces how many molecules of CO2?
4
Per turn of the TCA cycle (one acetyl-CoA enters), how many NADH are produced?
3
T/F: Zymogens are enzymes involved in the breakdown of fats in the diet, playing an important role in preparing fatty acids for transport through the digestive tract into the blood.
False
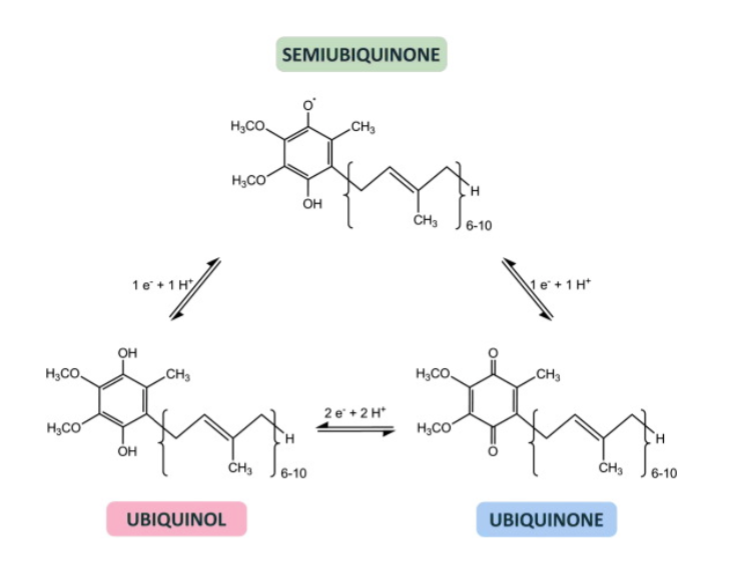
T/F: Ubiquinone is a post-translational modification that often targets proteins for degradation by the proteasomal machinery.
False
Transaminases (also known as amino transferases) are an enzyme family that converts α-ketoacids into amino acids. What enzyme family does the opposite reaction?
Transaminases
All of the following reactions of the urea cycle occur in the cytosol except for:
- Carbamoyl phosphate synthase
- Arginosuccinate synthetase
- Arginase
- Arginosuccinate lyase
Carbamoyl phosphate synthase
Ornithine, considered to be the 21st amino acid, combines with carbamoyl phosphate to form citrulline which has how many nitrogen-containing functional groups?
3
Arginosuccinase is an enzyme in the urea cycle which cleaves arginosuccinate to generate arginine with 4 nitrogen atoms and what other molecule?
Fumarate
What does the shunt do?
Connects the urea cycle to the TCA cycle by feeding critical intermediates
In the example of glutamine degradation by glutaminase and glutamate dehydrogenase in the kidneys, how many ammonium (NH4+) are liberated and excreted in the form of urea?
2
Albinism (lack of melanin) is a genetic condition attributed to mutation in which metabolic pathway?
Amino acid oxidation/degradation
The glycerol-phosphate and the malate-aspartate shuttles exist because:
Electrons generated from the glycolysis need to be moved into the mitochondria for ETC
The ACP acetyl-carrier domain of the FAS (fatty acid synthase) and Coenzyme-A (CoA) are similar in that they both:
Use a reactive cysteamine-phosphopantotheine flexible cofactor to covalently interact with substrate molecules
What is essential for transporting all the electrons of the electron transport chain to complex IV?
Cytochrome C
Why does FADH2 provide fewer ATP equivalents than NADH by the end of the ETC?
Complex II, which binds FADH2, does not pump protons
Why would anaerobic exercise and glucagon both inhibit the ACC (acetyl-CoA carboxylase)?
Energy levels are low, we don't need to be making fats
Lipoic acid and biotin are similar in that they both:
Are swing arm prosthetic groups
All of the following statements are true except:
- Fatty acid biosynthesis and β (beta) oxidation require NAD+ as an electron carrier
- Fatty acid biosynthesis and β (beta) oxidation occur in different cellular compartments
- The majority of fatty acid biosynthesis is catalyzed by one protein
- Malonyl CoA is important for fatty acid biosynthesis
Fatty acid biosynthesis and β (beta) oxidation require NAD+ as an electron carrier
In the process of fatty acid catabolism, which step involves an enzyme-bound acyl-adenylate intermediate?
Activation
The reaction catalyzed by fumarase in the TCA cycle requires fumarate and what else?
H2O
Why is it important that ubiquinone undergoes this cycling between states (characterized by color changes associated with its protonation state)?
Because some components of the ETC are obligate 1e- acceptors