Alkene Addition Reactions (CH 8)
1/11
Earn XP
Description and Tags
These flashcards cover key organic reaction mechanisms related to alkene transformations, their definitions, and the processes involved.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
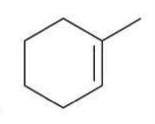
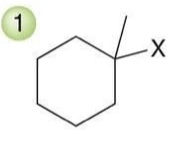
Hydrohalogenation (Markovnikov)
A reaction where a hydrogen halide adds to an alkene, with the hydrogen atom preferentially adding to the more substituted carbon.
Reagent: HX
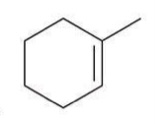
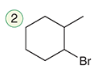
Hydrohalogenation (anti-Markovnikov)
A reaction where a hydrogen halide adds to an alkene, with the hydrogen atom preferentially adding to the less substituted carbon.
Reagent:
HBr
ROOR
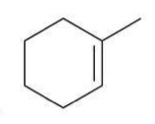
Acid-catalyzed hydration
The addition of water across a double bond in the presence of an acid, resulting in the formation of an alcohol.
Reagent: H3O+
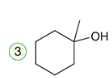
Oxymercuration-Demercuration (Anti-Markovnikov)
A two-step reaction involving the addition of mercuric acetate followed by reduction to form an alcohol.
Reagent:
1)Hg(OAc)2, H2O
2) NaBH4
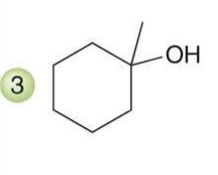
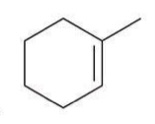
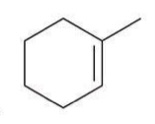
Hydroboration-Oxidation
A two-step reaction in which an alkene is converted to an alcohol via the addition of borane and subsequent oxidation.
Reagent:
1) BH3.THF
2) H2O2, NaOH
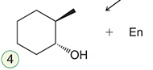
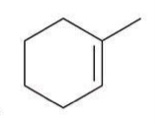
Hydrogenation
The addition of hydrogen across a double bond, typically in the presence of a catalyst such as palladium, platinum, or nickel, converting alkenes into alkanes. The addition of hydrogen removes the double bond.
Reagent:
H2
Pt/Pd
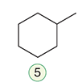
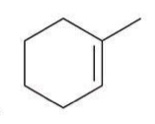
Bromination
The addition of bromine across a double bond, resulting in the formation of a vicinal dibromide.
reagent: Br2
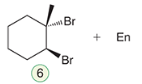
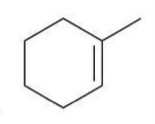
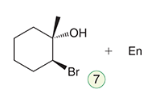
Halohydrin formation
The addition of a halogen and water across a double bond, yielding a halohydrin. This reaction involves the formation of an alcohol and a halide, leading to the addition of -X and -OH at adjacent carbons.
Reagent: Br2, H2O
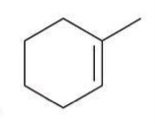
Anti Dihydroxylation
A reaction where two hydroxyl groups are added to a double bond from opposite sides.
Reagent:
1) MCPBA
2) H3O+
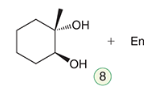
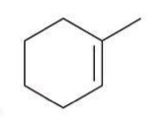
Syn Dihydroxylation
A reaction where two hydroxyl groups are added to a double bond from the same side.
2 Possible Reagents:
a) 1st Reagent
1) OsO4
2) NaHSO3, H2O
b) 2nd Reagent
KMnO4
NaOH, cold
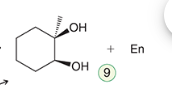
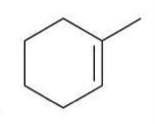
Ozonolysis
A reaction where ozone reacts with alkenes to cleave the double bond and produce carbonyl compounds.
Reagent:
1) O3
2) DMS
Chapter 8 Splurge
Section 8.1
Addition reactions are characterized by the addition of two groups across a double bond.
Section 8.2
Alkenes are abundant in nature.
Ethylene and propylene, both formed from cracking petroleum, are used as starting materials for a wide variety of compounds.
Section 8.3
In addition to their systematic and common names, alkenes are also classified by their degree of substitution, which refers to the number of alkyl groups connected to a double bond.
Section 8.4
Addition reactions are thermodynamically favorable at low temperature and disfavored at high temperature.
Section 8.5
Hydrohalogenation reactions are characterized by the addition of H and X across a π bond, where X is a halogen.
For unsymmetrical alkenes, the placement of the halogen represents an issue of regiochemistry. Hydrohalogenation reactions are regioselective, because the halogen is generally installed at the more substituted position, called Markovnikov addition.
In the presence of peroxides, addition of HBr proceeds via an anti-Markovnikov addition.
The regioselectivity of an ionic addition reaction is determined by the preference for the reaction to proceed through the more stable carbocation intermediate.
When one new chiral center is formed, a racemic mixture of enantiomers is obtained.
Hydrohalogenation reactions are only efficient when carbocation rearrangements are not a concern.
Section 8.6
Addition of water (H and OH) across a double bond is called hydration.
Addition of water in the presence of an acid is called acid-catalyzed hydration, which generally proceeds via a Markovnikov addition.
Acid-catalyzed hydration proceeds via a carbocation intermediate, which is attacked by water to produce an oxonium ion, followed by deprotonation.
Acid-catalyzed hydration is inefficient when carbocation rearrangements are a concern.
Dilute acid favors formation of the alcohol, while concentrated acid favors the alkene.
When generating a new chiral center, a racemic mixture of enantiomers is expected.
Section 8.7
Oxymercuration-demercuration achieves hydration of an alkene without carbocation rearrangements.
The reaction is believed to proceed via a bridged intermediate called a mercurinium ion.
Section 8.8
Hydroboration-oxidation can be used to achieve an anti-Markovnikov addition of water across an alkene. The reaction is stereospecific and proceeds via a syn addition.
Borane (BH3) exists in equilibrium with its dimer, diborane, which exhibits three-center, two-electron bonds.
Hydroboration proceeds via a concerted process, in which borane is attacked by a π bond, triggering a simultaneous hydride shift.
Section 8.9
Catalytic hydrogenation involves the addition of H2 across an alkene in the presence of a metal catalyst.
The reaction proceeds via a syn addition.
Heterogeneous catalysts are not soluble in the reaction medium, while homogeneous catalysts are.
Asymmetric hydrogenation can be achieved with a chiral catalyst.
Section 8.10
Halogenation involves the addition of X2 (either Br2 or Cl2) across an alkene.
Bromination proceeds via a bridged intermediate, called a bromonium ion, which is opened by an SN2 process that produces an anti addition.
In the presence of water, the product is a bromohydrin or a chlorohydrin, and the reaction is called halohydrin formation.
Section 8.11
Dihydroxylation reactions are characterized by the addition of OH and OH across an alkene.
A two-step procedure for anti dihydroxylation involves conversion of an alkene to an epoxide, followed by acid-catalyzed ring opening.
Section 8.12
Syn dihydroxylation can be achieved with osmium tetroxide or potassium permanganate.
Section 8.13
Ozonolysis can be used to cleave a double bond and produce two carbonyl groups.
Section 8.14
In order to predict the products of an addition reaction, the following three questions must be considered:
What is the identity of the groups being added across the double bond?
What is the expected regioselectivity (Markovnikov or anti-Markovnikov addition)?
What is the expected stereospecificity (syn or anti addition)?
Section 8.15
The position of a halogen or OH group can be changed via elimination followed by addition.
The position of a π bond can be changed via addition followed by elimination.