unit 8: cell-cell junctions, cell-matrix junctions
1/55
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
56 Terms
describe connective tissue
ex: bones and tendons
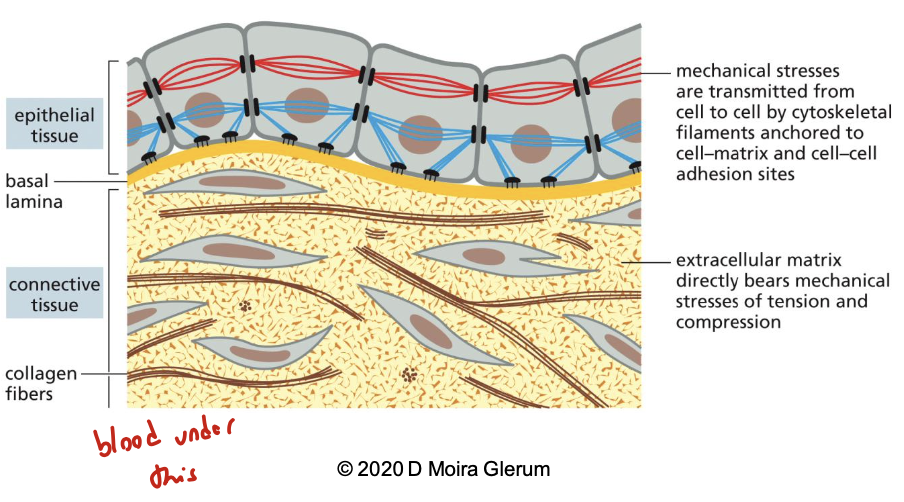
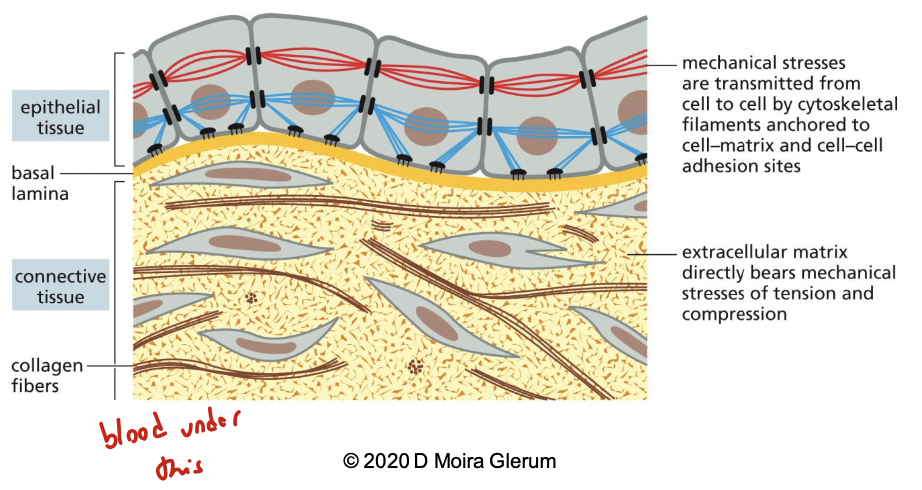
this tissue has lots of ECM. it’s a secreted network of proteins and polysaccharides, frequently rich in collagen. it bears most of the mechanical stress, provides support. it has a strong attachment with the ECM through cell-matrix junctions by cytoskeleton and few direct attachments with each other
describe epithelial tissue
ex: skin, lining of gut
these are sheets of cells tightly bound together by cell-cell junctions via the cytoskeleton. ECM is a thin mat on the basal side called the basal lamina (aka basement membrane), linked to the cytoskeleton via cell-matrix junctions
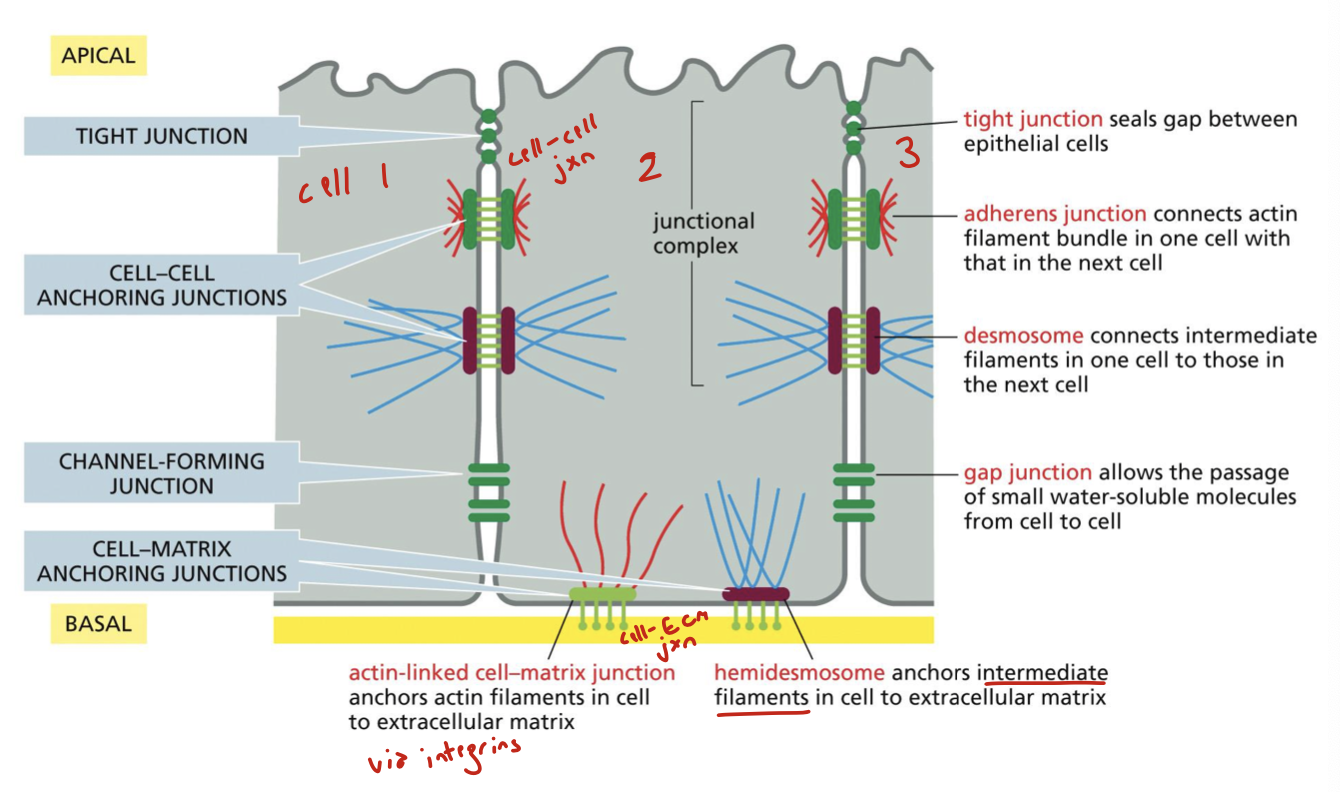
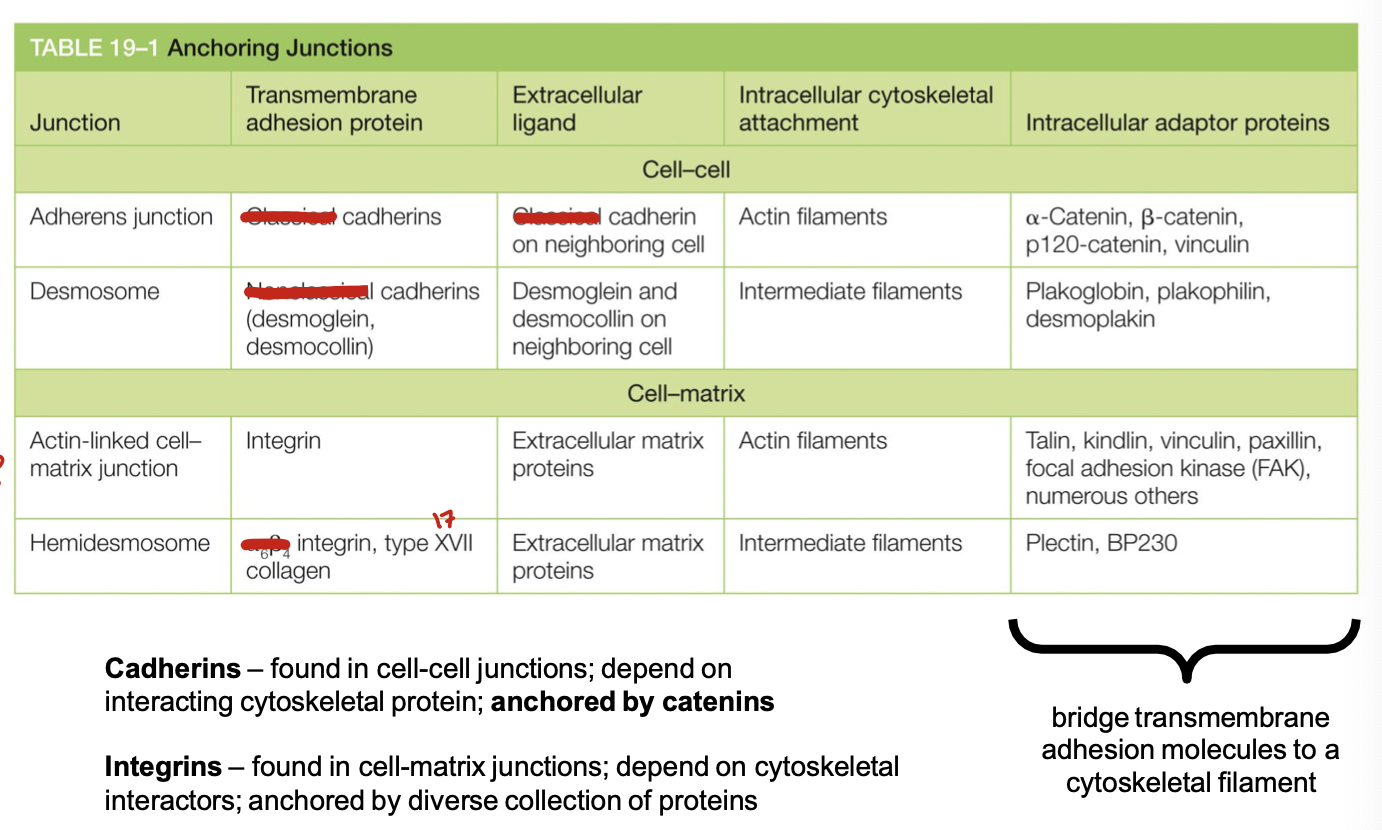
list and describe the 6 types of cell junctions
tight junction: cell-cell. seals gap between epithelial cells
adherens junction: cell-cell. connects actin filament bundle in one cell with that in the next cell
desmosomes: cell-cell. connects intermediate filaments in one cell to those in the next cell
gap junction: cell-cell. allows the passage of small water-soluble molecules from cell to cell
actin-linked cell-matrix junction: cell-matrix. anchors actin filaments in cell to ECM via integrins
hemidesmosome: cell-matrix. anchors intermediate filaments in cell to the ECM
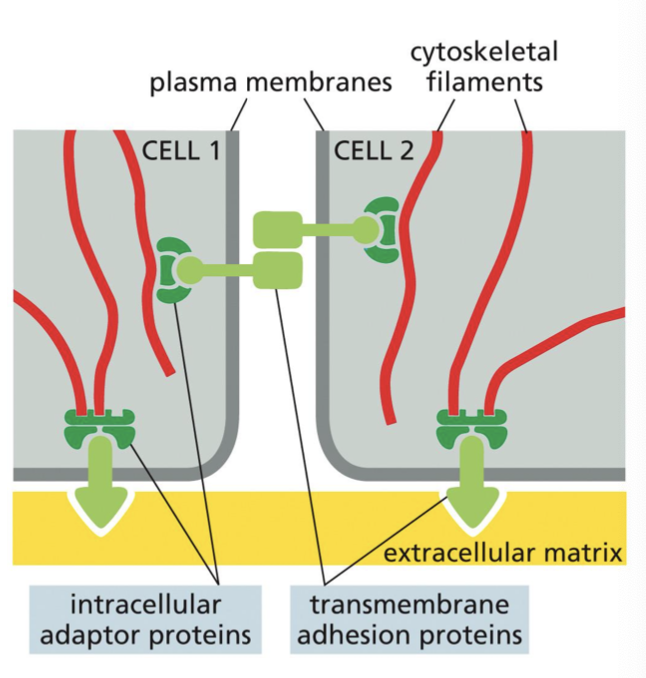
describe cell-cell adhesions. what are the two superfamilies of transmembrane adhesion proteins?
cell-cell adhesions are most clearly seen in mature epithelia where there is strong, direct anchorage of adjacent cells.
there are 4 main types of anchoring junctions: tight, adhesion, desmosome, gap.
transmembrane adhesion proteins span the PM
the two superfamilies:
cadherins: generally cell-cell attachments. anchored by catenins. depend on interacting cytoskeletal protein
integrins: generally cell-ECM attachment. anchored by diverse collection of proteins. depend on cytoskeletal interactors
what are the four types of anchoring junctions? categorize them as cell-cell or cell-matrix and list the transmembrane adhesion protein involved. mention which intracellular cytoskeletal attachment is involved
adherens junction: use cadherins. are cell-cell. has actin filaments
desmosome: use cadherins. cell-cell. has intermediate filaments
actin-linked cell-matrix junction: integrins. cell-ECM. actin filaments
hemidesmosomes: use integrins and type 17 collagen. cell-ECM. intermediate filaments
what organisms have cadherins?
all multicellular animals but not fungi and plants or prokaryotes
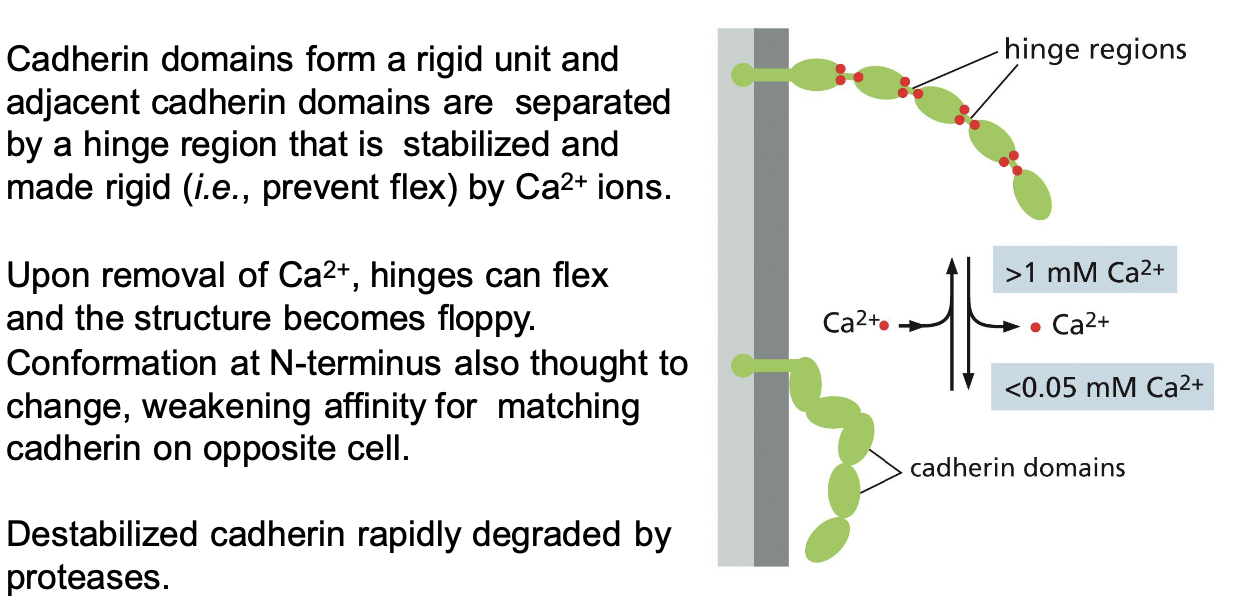
what gives us the cadherin name?
calcium-dependent adhesion
how were cadherins discovered?
some types of cells can be readily dissociated by removing calcium from the EC medium. sometimes the addition of a protease is also required. if dissociated cells are placed back into the normal medium, they can reassociate.
they were first found in: epithelial cells (E-cadherin), nerve cells (N-cadherin), and placenta+epidermis (P-cadherin).
these are called classical cadherins: they’re closesly related throughout their sequece and perform well-defined adhesive functions.
what is the nature of binding between cadherins of one cell to the cadherins of another?
homophilic interactions, making the anchoring junction symmetrical
describe how cadherins act as velcro
they bind to each other at N-terminal tips. binding of partners typically with low affinity but many weak bonds in parallel result in a strong overall attachment. they’re easy to disassemble by breaking the molecules sequentially from the side (just like velcro)
making and breaking of anchoring junctions is vital for development and tissue turnover
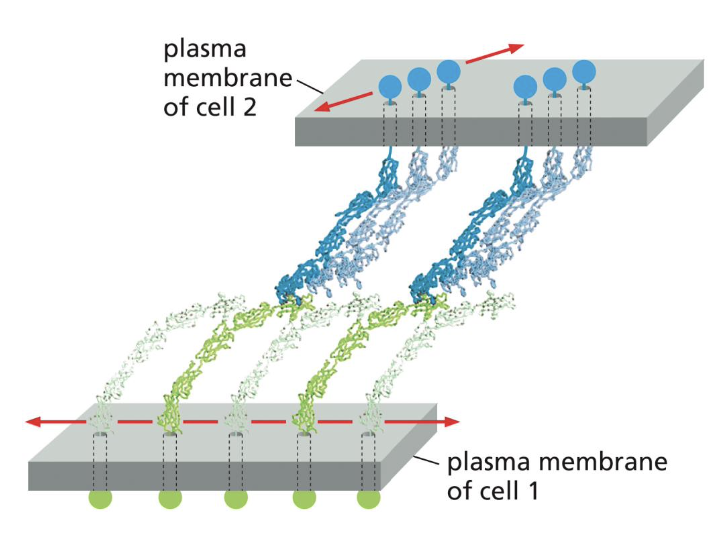
describe the homophilic binding mechanism of cadherins
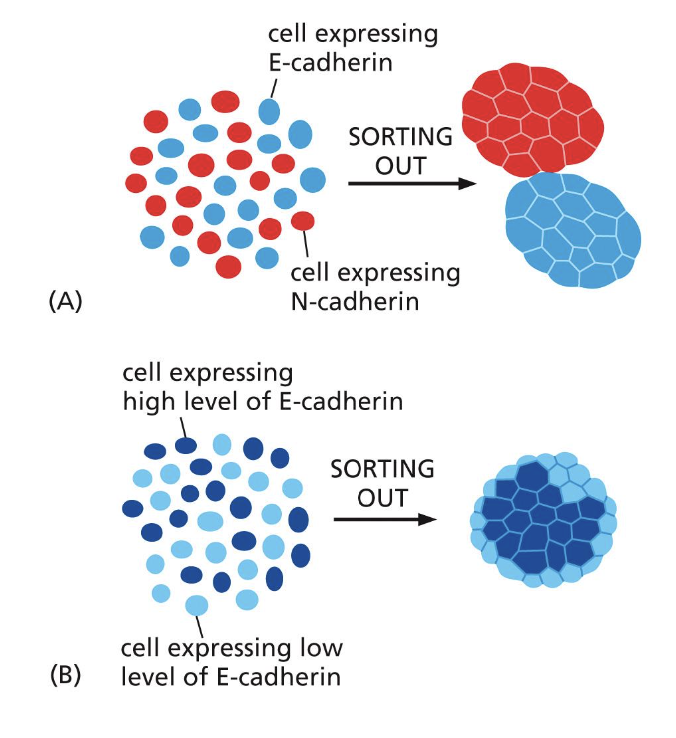
we see this binding in action when fibroblasts that normally don’t express cadherins transfect with E-cadherin: they end up able to adhere
if you transfect them with a different cadherin it’ll sort according to the specific type
this is how homophilic binding controls tissue segregation.
note that cells with higher levels of cadherins and homophoilic binding adhere more strongly and typically end up internally
differentiate between epithelial and mesenchymal calls
the expression of cadherin in dispersed unattached (aka mesenchymal) cells can cause them to come together and form an epithelium (ex: tissue regen)
the epithelial-to-mesenchymal transition is seen during development when cells leave the epithelium (ex: metastasis).
epithelial cells
form sheets connected by junctions
move in harmony
have a clear polar character
contacts only one surface of the cell
mesenchymal cells
loosely organized and loosely attached
can migrate as individual cells
can also adhere in 3D clumps
basal lamina may surround the cell
what are catenins?
accessory intracellular anchor proteins for cadherins
actin cytoskeleton in adherens is brought together via β-catenin
IFs in desmosomes via γ-catenin
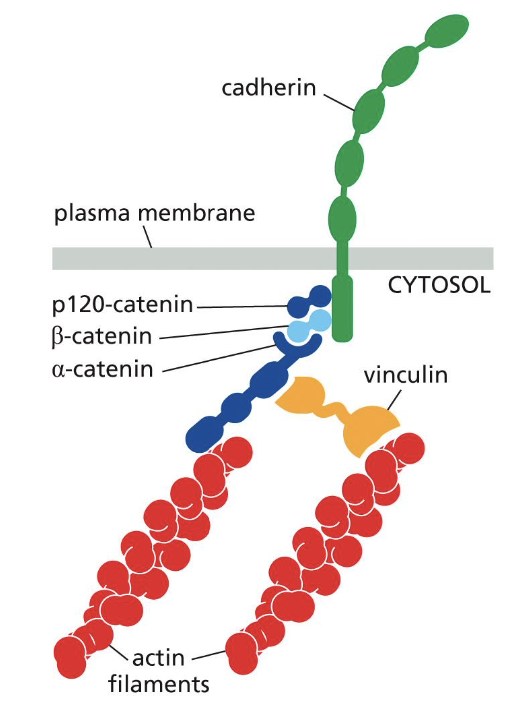
what does a loss of β-catenin in the heart lead to?
arrthythmogenesis
describe the assembly of adherens junctions
in an unattached cell, contraction of actin-myosin bundles maintain cortical tension
loss of cortical tension needs to occur for assembly of adheren junctions
this happens due to local Rac activation and Rho inhibition
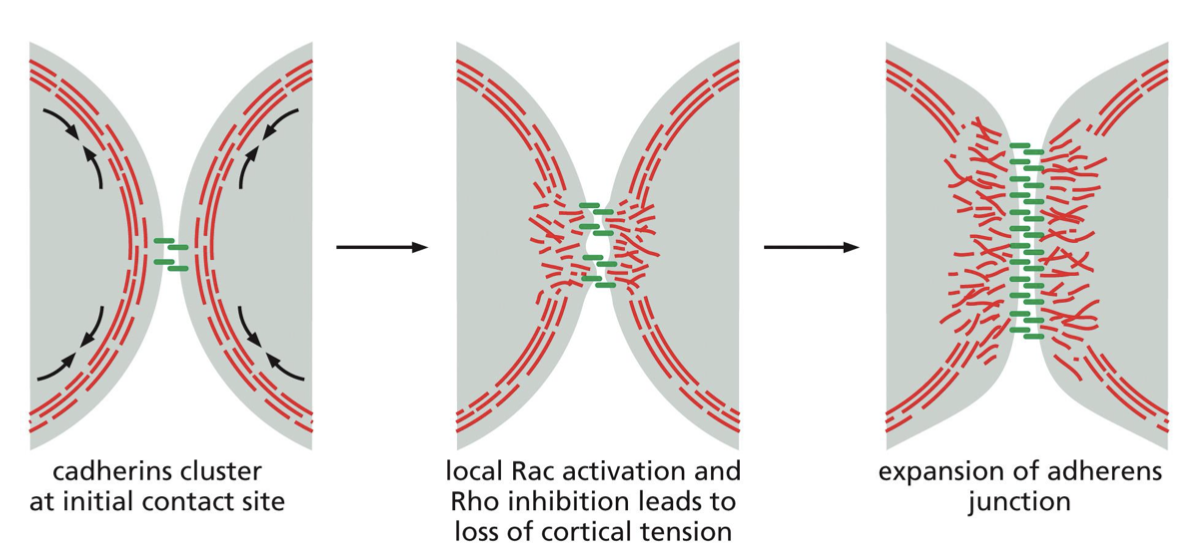
what are zonula adherins?
junctions organized as continuous adhesion belt beneath the apical surface of cells
define desmosomes
linkage of cadherins to IFs of cytoskeleton, organized in a button-like spot
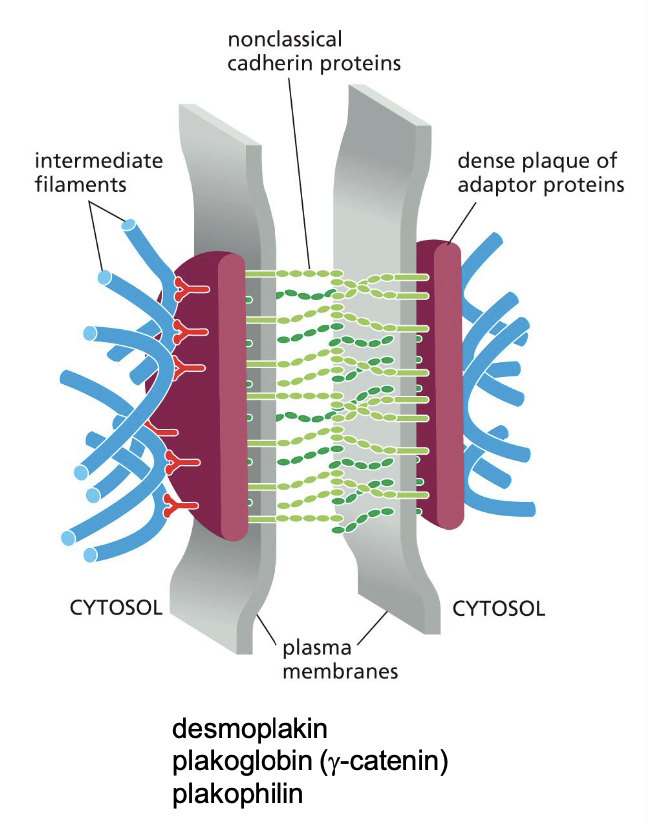
which organisms have desmosomes?
vertebrates, NOT invertebrates
what do desmosomes do? what is pemphigus?
they’re found in tissue subject to high stress: it provides strength
pemphigus = disorder resulting from disruption of desmosomes. an autoimmune disease that causes blistering of skin and leakage of fluids into loosened epithelium
describe the role of tight junctions
they form a selectively permeable barrier by connecting epithelia
adjacent cells are sealed by tight junctions, which prevents molecules from leaking back into original space. they also prevent apical and basolateral proteins from diffusing into wrong area
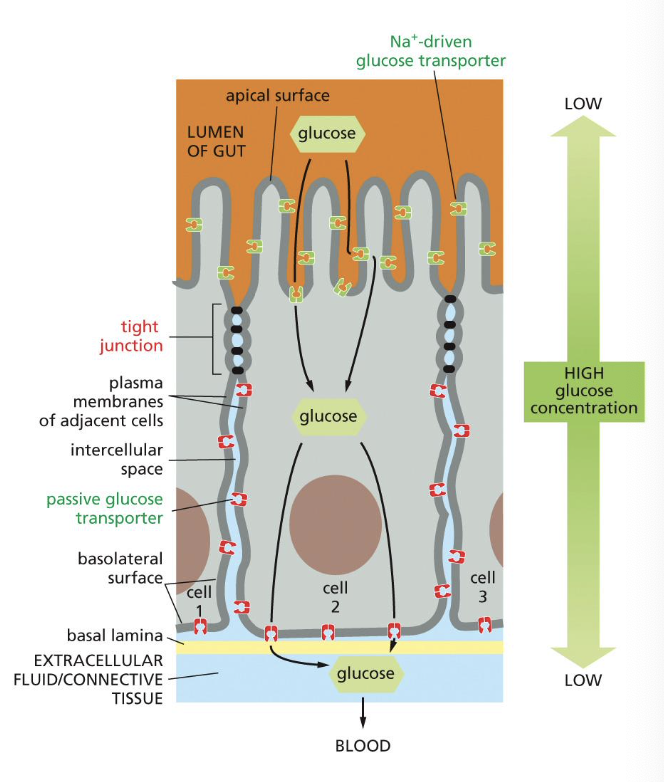
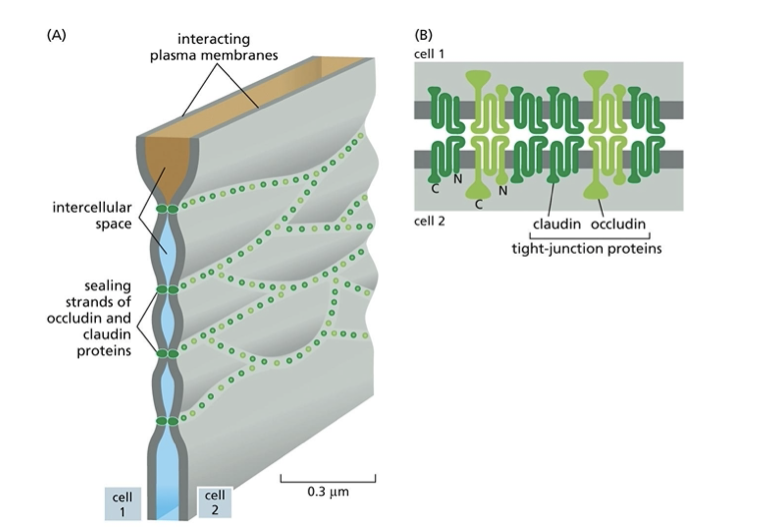
how can we observe tight junctions?
by freeze-fracture EM. this lets us see branching networks of sealing strands that encircle epithelial cell. conventional EM shows tight apposition.
what is claudin?
constituent proteins of tight junctions that form sealing strands
different claudin members are found in different cell types
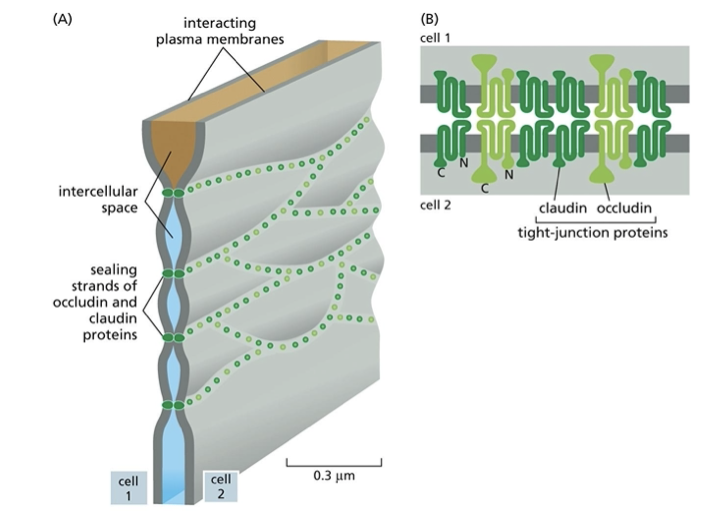
what are occludins?
constituents of tight junctions that limit permeability
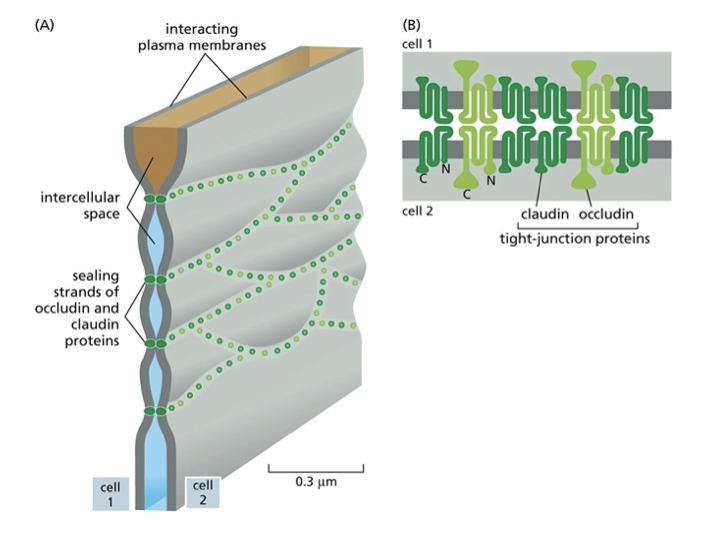
what is tricellulin?
a constituent of the tight junction that prevents leakage at them
describe the mechanism of clostridium perfringens enterotoxin (CPE) and any possible therapies
it’s a gastrointestinal, foodborne disease which binds to a subset of claudins. pore formation in the PM of host mucosal cells is mediated by N-terminal domains, leading to cell death. note that C-terminal domain CPE (aka cCPE) is NOT cytotoxic though! it’s just the N-terminal version…
so cCPE is thought to be a possible claudin modulator: it increases paracellular permeability and could be used to improve drug delivery across tissue barriers. it could also target claudin-overexpressing tumors and help deregulate claudin expression associated with tumor growth
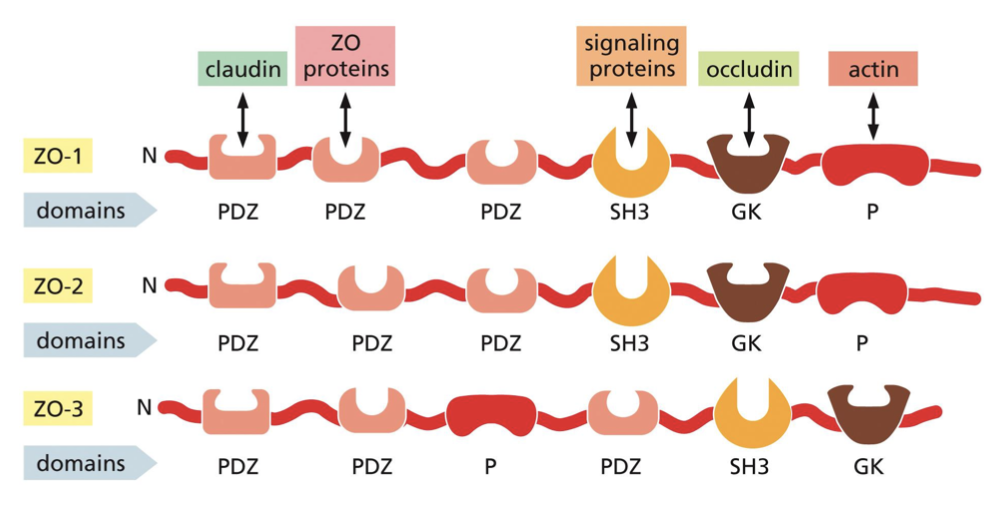
what are ZO proteins?
zonula occludens: intracellular scaffold proteins whose binding to cytoplasmic domains determines the organization of the junctional complexes made by claudins and occludins.
there are three types: ZO-1, 2, 3. each have multiple protein interacting domains and can interact with other ZO proteins
in order to form a junctional complex, they have to be in the right position. this will form a sealing strand, usually apical to adherens and desmosome junctions
what is the gap junction equivalent in plants?
plasmodesmata
define gap junctions and how we can see them
they bridge gaps between adjacent cells to create direct channels. they allow cells to exchange small molecules (less than 1000 Da) such as inorganic ions, sugars, amino acids, nucleotides, vitamins, and signalling molecules such as cAMP. the passage of inorganic ions and small water-soluble molecules means that epidermal cells are metabolically and electronically connected (ex: heart muscle cells)
can see with the TEM
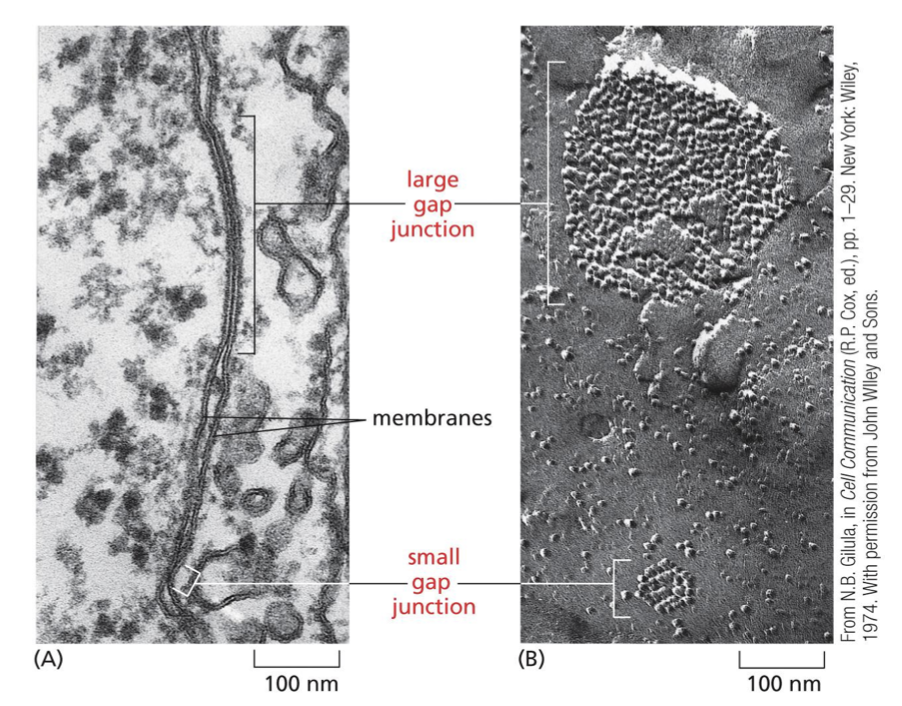
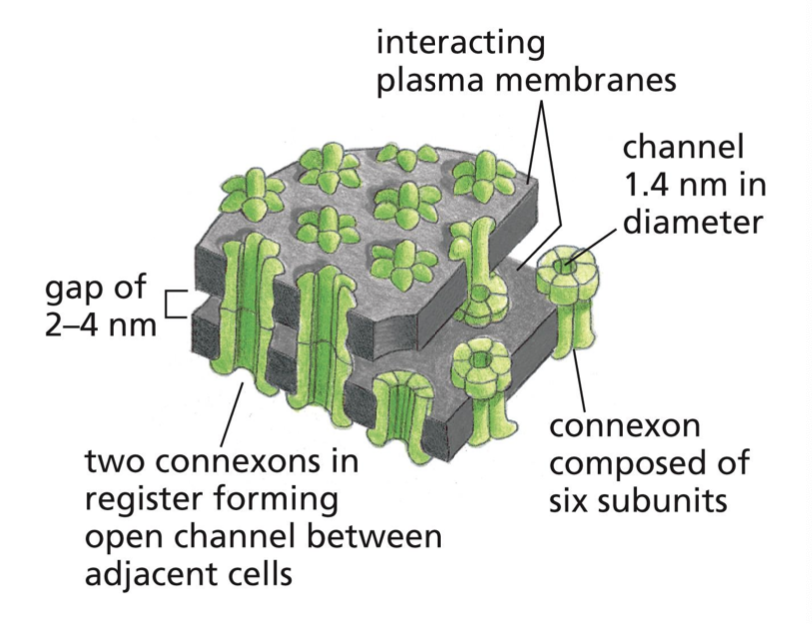
describe the composition of gap junctions
they are a collection of channels formed by proteins called connexins and innexins. vertebrates are mostly connexins, while invertebrates are mostly innexins
six connexins assemble to form a hemichannel called the connexon. when connexons in PMs of 2 cells in contact are aligned, they form a continuous aqueous channel
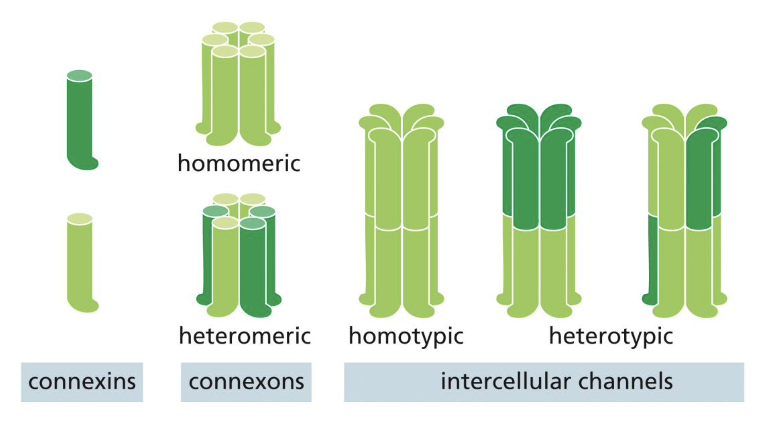
how does cell type change gap junction function?
they have different properties and permeabilities per cell due to different connexins
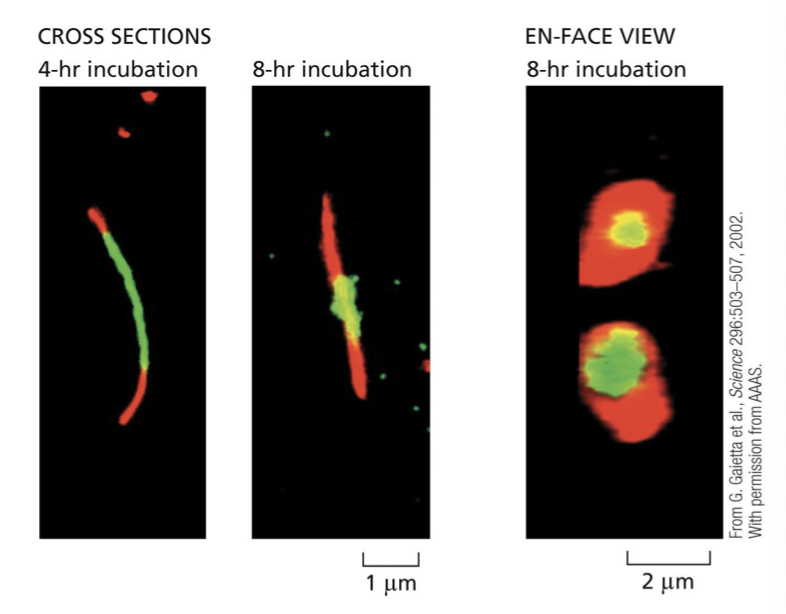
describe the gap junction states mechanics
they can switch between open and closed states in response to specific stimuli, like ion channels:
ex: voltage differences between 2 cells can change the channel’s state
what kind of diseases are caused by mutations in gap junctions?
congenital deafness, cataracts, peripheral nerve demyelinating disease
are gap junctions dynamic or static?
dynamic. they can be assembled, disassembled, remodelled
how do new connexons get sent around?
via exocytosis: new connexons are continually added around the periphery as old connexions are removed from the core. they then diffuse in plane of membrane until they encounter periphery of plaque and get trapped
what makes up the ECM?
tissues made of cells and a macromolecular network
what produces the ECM?
local cells
what does the ECM provide?
support, regulation, survival, development, migration, proliferation, shape, and function
define the basal lamina
specialized ECM found in epithelial tissue
what produces the ECM in connective tissue? cartilage? bone?
connective tissue: fibroblasts
cartilage: chondroblasts
bone: osteoblasts
what are glycoasminoglycans (GAGs)? describe the structure
unbranched polysaccharide chains composed of repeating disaccharides: 1 sugar is always either sulfated (negative) GlcNAc or GalNAc, 2nd sugar is usually uronic acid
it is highly negative and very hydrophilic. they are characterized by linkage between sugars, number and location of sulfation. it attracts cations that are osmotically active and causes large amounts of water to be sucked into the ECM
they occupy a huge volume (look at pic)
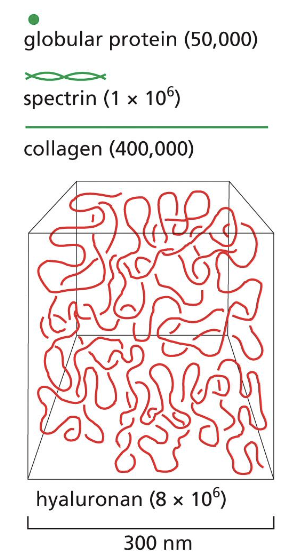
what are the 4 main groups of GAGs?
hyaluronan, chondroitin/dermatan, heparan, keratan
what does GAG create in the ECM?
turgor: enables matrix to resist compression as opposed to collagen, which resists stretching
describe hyaluronan
the simplest GAG available in variable amounts throughout the body. it’s strange: no sulfation, disaccharides are identical, and it isn’t linked to the protein core (all traits are completely opposite of normal GAGs. it also isn’t exocytosed like normal GAGs: it is created and spun out directly from enzyme complex (embedded in the PM) at the cell surface
what are linkage tetrasaccharides?
specific polysaccharide that forms chains assembled in the golgi. one sugar is added to it at a time by a specific glycosyl transferase and undergoes covalent modifications (such as sulfation) in the golgi
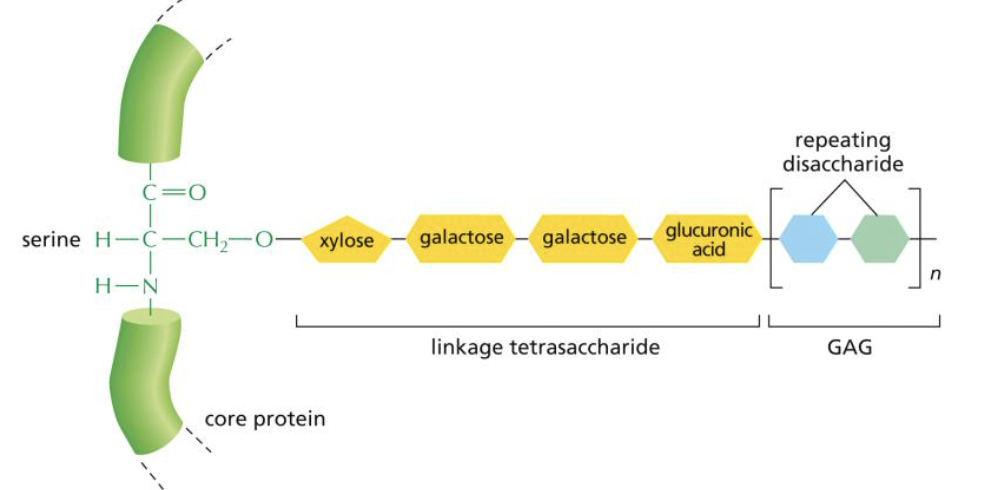
are all proteoglycans secreted from the cell?
no: some need to stay
do all GAGs have the same core proteins?
no
what are aggrecan and decorin?
two types of proteoglycans with vastly different structures: shows the hetergeneity amongst proteoglycans
aggrecans have lots of GAGs and are an important component of cartilage, while decorins have 1 GAG, found in fibroblasts, and binds collagen to regulate fibril assembly and size
note that GAGs and proteoglycans can further associate to form even larger polymeric complexes. ex: aggrecan can assemble with hyaluronan in cartilage matrix to form aggregates as big as a bacterium
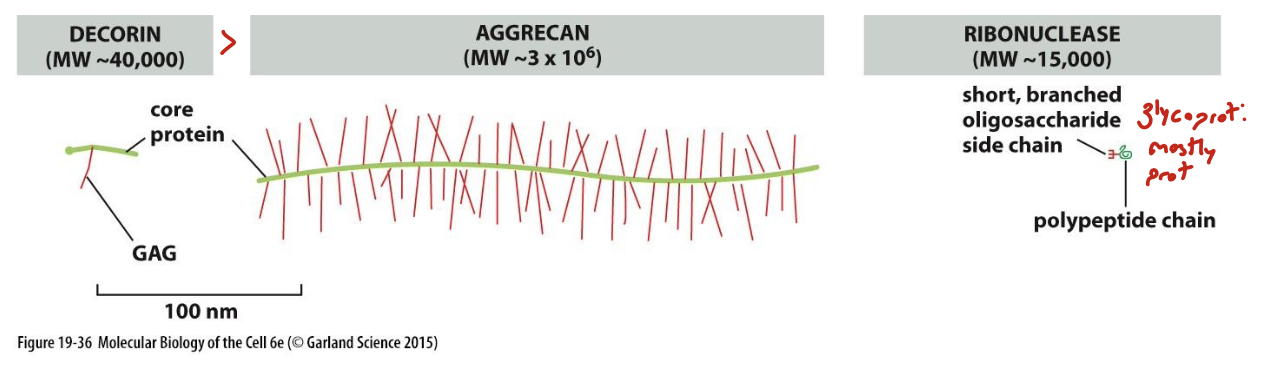
describe collagen structure
fibrous protein
found in skin and bones
long and stiff triple helix
extremely rich in proline and glycine
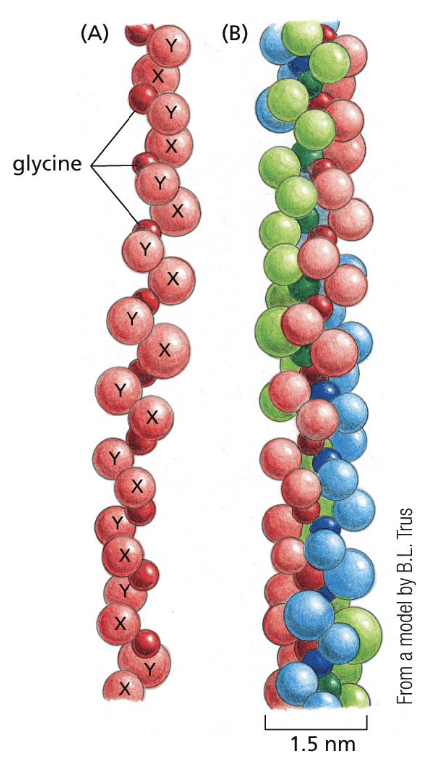
list 4 types of collagen and describe them
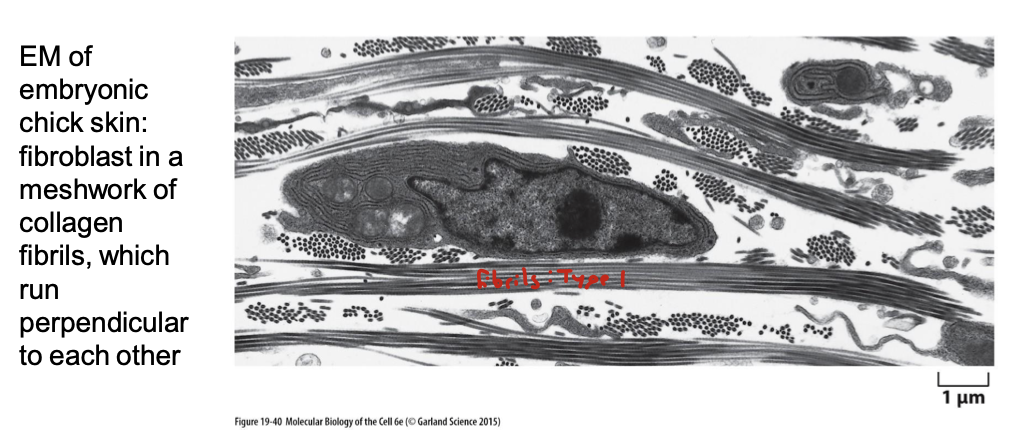
type I: most common and found in skin and bones. forms fibrils. super long and rope-like. can visualize it by TEM and light microscopy. can aggregate into larger cable-like bundles
type IX and type XII: fibril-associated collagens that associate with type 1. decorate surface of collagen fibrils. link fibrils to one another and to other ECM components (they’re just links)
type IX binds to type II in cartilage, cornea
type XII binds to type I in tendons
type IV: network-forming collagen. major component of basal lamina ONLY
describe the process of collagen fibril assembly
synthesis of pro-α chain
hydroxylation of selected prolines and lysines
glycosylation of selected hydroxylysines
self-assembly of three pro-α chains
procollagen triple-helix forms
propeptides are secreted
cleavage of propeptides
self-assembly into fibril
aggregation to form a fiber
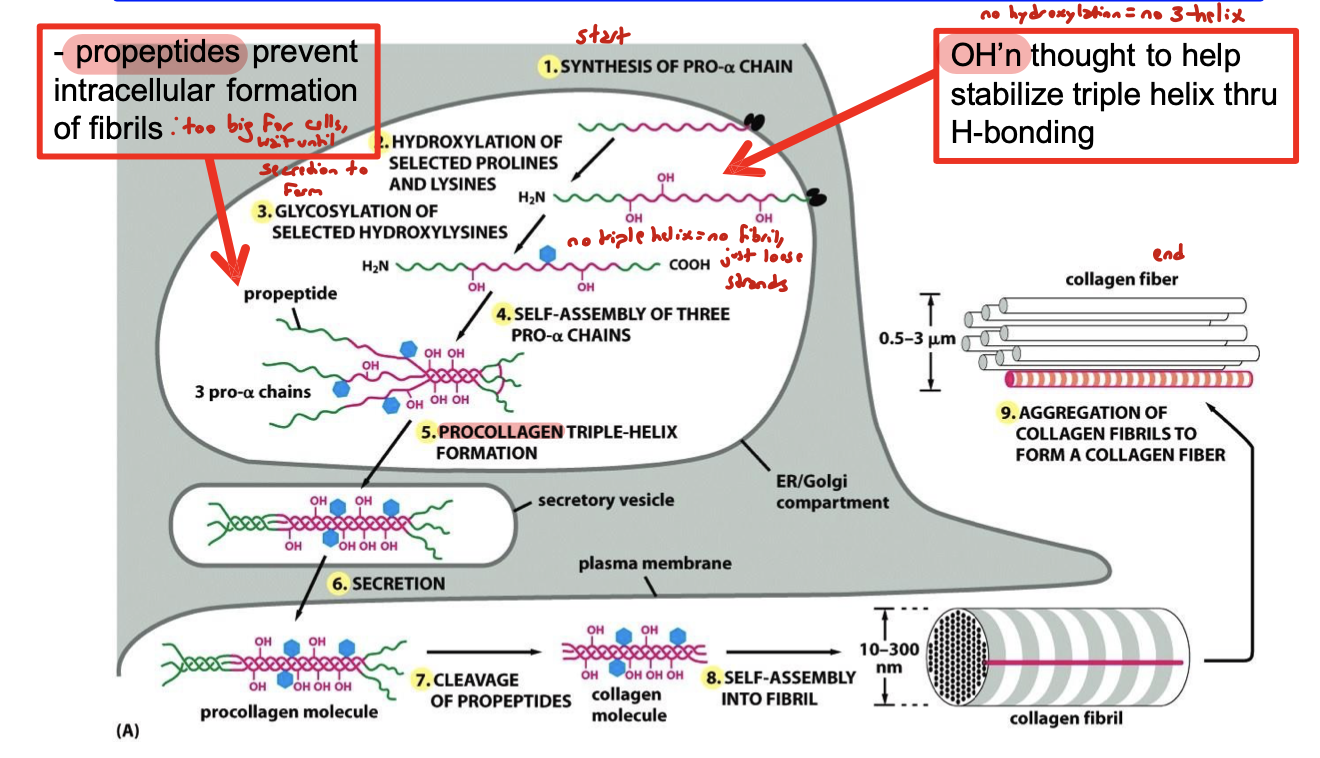
what do propeptides do?
prevent intracellular formation of fibrils because fibrils are too big for cells. so fibrils wait until after secretion to form
what helps stabilize triple helix of propeptide?
hydroxylation through hydrogen bonding
no hydroxylation = no triple helix
what collagen-related diseases can result from a lack of hydroxylation?
scurvy: vitamin C deficiency, which is a cofactor for hydroxylases that hydroxylate pro-α chains. detective pro-α chains fail to form triple helix and are degraded. synthesis of new collagen is then inhibited, causing a gradual loss of preexisting normal collagen due to the turnover nature of it. blood vessels become fragile, teeth become loose, and wounds fail to heal
osteogenesis imperfecta type 1: inherited defect of collagen that is autosomal dominant. it causes weak bones and easy fractures
describe elastic fiber properties
are resilient to recoil after transient stretch. they’re interwoven with collagen to limit extent of stretch, prevent tearing
you see these in aorta, where you definitely don’t want tearing…
describe elastic fiber structure
made of elastin
highly hydrophobic
has a high Pro and Gly content, but it’s not glycosylated
it has hydroxylated proline but not lysine
tropoelastin is a precursor that is secreted into the EC space and assembled into fibers close to the PM. they exhibit a high degree of cross-linking
hydrophobic segments of elastin are responsible for the elastic properties
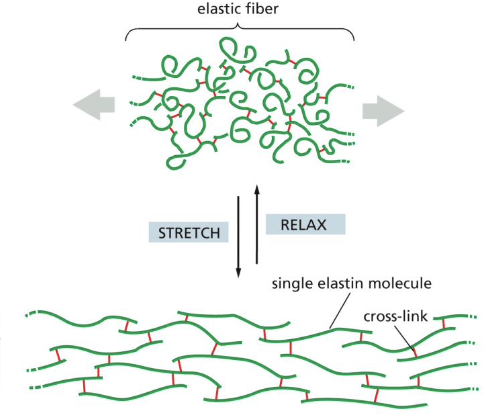
describe the components of elastic fibers
elastin
a dominant ECM protein in aerties and the aorta. deficiency of it causes narrowing of arteries and excessive proliferation of smooth muscle cells
microfibrils
sheath the core of fibers to provide scaffolding
fibrillin
glycoproteins that make up microfibrils. they bind to elastin and are essential for integrity of elastic fibers. mutations in them cause Marfan syndrome, which causes aortic rupture