Physics - 6.6 Half Life
1/14
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
15 Terms
What do the nuclei of radioactive atoms do because they are unstable?
They decay to form a completely different type of atom.
What sort of process is radioactive decay?
It is a random process and is impossible to predict when an individual nucleus will decay.
Define ‘half-life of the radioactive isotope’.
The time taken for the number of radioactive nuclei to reduce to one half of its initial value. Different isotopes have different half-life values.
As the isotope decays, what happens to the fraction of the original sample do?
The fraction of the original sample remaining (represented by the pink) decreases and the fraction that has decayed (represented by the grey) increases.
As the number of undecayed nuclei decreases, the number of decayed nuclei increases.
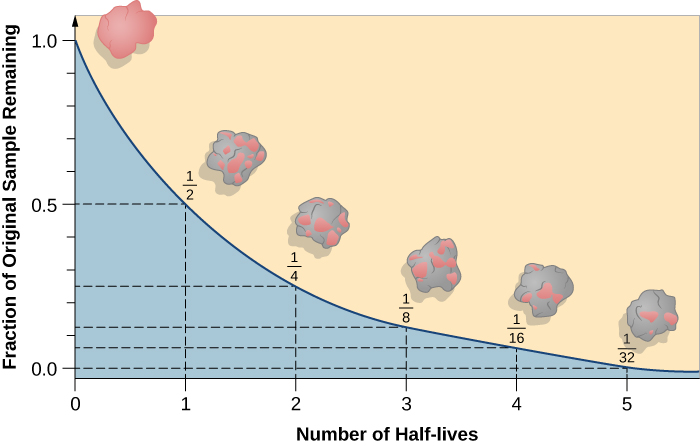
What does a longer half-life mean?
The substance will take longer to decay.
What is radioactive decay measured in?
Becquerel, Bq.
What two things does the activity of radioactive material depend on?
The number of radioactive/unstable atoms present.
The half life of the atoms.
What is carbon dating?
It uses Carbon-14, to estimate the age of an object.
How does carbon dating work?
Living things take in small amounts of radioactive carbon-14. After death, the amount of carbon-14 in the object decreases because the carbon-14 atoms decay. The amount of carbon-14 remaining can be used to date the object (for example, bones from a skeleton or objects made from wood).
When selecting a suitable isotope for a specific application, what needs to be considered? (2)
the half-life of the isotope
the penetrating power and biological effects of the radiation.
How are radioactive trackers used?
Radioactive tracers are used for medical imaging. A radioactive material is injected or ingested into the body and radiation detectors placed outside the body detect the radiation emitted. Cameras use this information to produce detailed images of inside the body.
How do you choose a suitable isotope for radioactive trackers?
Must have a short half-life so that once inside the body, it decays quickly.
The radiation emitted must penetrate the body easily and do as little damage as possible. Gamma radiation is suitable as it is the most penetrating and least ionising. Technetium-99m is commonly used in radioactive tracers. It decays by gamma emission and has a half-life of around six hours.
A radioactive material can be placed inside the tumour to kill the cancer cells. The radiation emitted needs to be what? (2)
strongly ionising so that it kills the cells
moderately penetrating so it can pass through the tumour to kill the cancer cells without damaging healthy ones.
What should the half-life be for cancer treatments?
A relatively short half-life is required but long enough, so the isotope remains active enough to treat the tumour. For example, palladium-103 is used in this way and has a half-life of about 17 days.
Why is a long half-life sometimes suitable to treat cancer.
A radioactive source outside the body can also be used to treat cancer. Radiation is aimed at the tumour from outside the body in order to kill the cancer cells. The radiation needs to be:
very penetrating so that it can pass through the body to reach the cancer cells
weakly ionising to limit any damage to healthy cells.
This means that it will not decay quickly and doesn’t need to be replaced often.