3. Macromolecules in solution
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
24 Terms
Macromolecules
Large molecules built up by small molecules (monomers), that are covalently linked
Examples:
Biological polymers:
Polysaccharides such as starch, cellulose etc.
Sugar monomers
Lignin
Phenolic monomers
Proteins: Not strictly polymers
Amino acid monomers
Synthetic polymers:
Polyethylene (PE)
Polyethylene glycol (PEG)
Polyvinylalcohol (PVA)
Polystyrene (PS)
Polyacrylamide etc.
What is homopolymer, copolymer, block copolymer and branched homopolymer?
Homopolymer- same type of monomer
Copolymer- alternating monomer
Block copolymer- alternating in block→ making them surface-active
Branched homopolymer- branching can occur for others
Polymer size - Hydrodynamic radius
Hydrodynamic radius is obtained from the Stokes-Einstein equation. It describes how diffusion of a particle or polymeric structure depends on its radius.
D= diffusion coefficient [m2/s]
k= Boltzmann constant (J/K)=1.38×10−23 J/K
T= temperature (K)
η=dynamic viscosity of continuous phase [Pa•s]
d=hydrodynamic diameter of particle [m]
Polymer size - Radius of gyration
Based on the distance of each monomer unit to the polymer’s center of mass. Used for random coil polymer
Debey’s definition of radius of gyration
It is most common one. Distance of every mass to the center of mass. The average distance is rg
rg=average distance
Mi=molar mass
ri=distance to the center of mass
Polymer conformation in solutions depends on
Interactions with solvent molecules
pH
Salt
Temperature
Interactions with other polymer molecules
Really likes solvent: polymer swell
Doesn’t like it as much: polymer minimises
Conformation in dilute solutions
Rod: stiff
Double helix: two molecules
Flory-Huggins theory
Describes solubility of a polymer and thermodynamics of macromolecular solutions.
Flory-Huggins parameter (χ)
– 0.1< χ<0.5 ”Good” solvent (molecule swell)
– χ ≈ 0.5 ”θ-solvent” (transition between good and poor solvents) (reduce it bellow to precipitate, increase to allow it dissolve)
– χ > 0.5 ”Poor” solvent (molecule shrink)
– χ >> 0.5 Insoluble
• χ can change with temperature, pH, ionic strength etc.
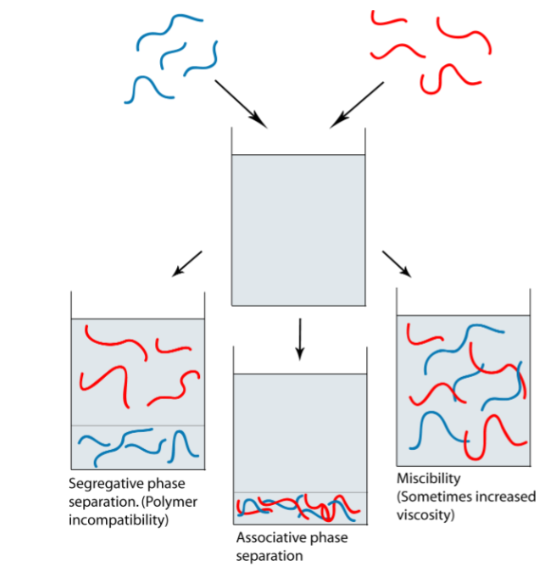
Phase behaviour of macromolecule solutions (miscibility, segregative phase separation and associated phase separation)
Polymer 1 and polymer 2 are chemically different
Miscibility: polymers like the solvent, don’t care about each other
Segregative phase separation: Polymers don’t like each other but like solvent
The rule for mixtures of non-ionic polymers
Associated phase separation: Polymers like each other better than solvents (e.g polymer could have opposite charges)
Polyelectrolytes of opposite charge, proteins-polysaccharides
Applications of macromolecules
• Thickening to increasing viscosity to preferred value and flow properties in liquid formulations. Example: Pharmaceutics, Foods and Paints and coatings
• Stabilization of dispersions
Rheology
Rheology is the study of the flow and deformation of matter, including liquids, solids, and soft materials, under applied forces or stresses.
Shear stress
Shear stress is the external force applied parallel to the surface, which causes deformation.
σ=shear stress [Pa]
F=applied external force [N]
A=cross-sectional area [m2]
Shear rate
Shear rate 𝛾 is the rate at which strain changes over time, which is the rate of deformation.
γ=shear rate [1/s]
v=velocity [m/s]
y=distance between moving plates [m]
Describe viscosity is in pure liquid and solution of polymer
In a pure liquid: friction between molecules
In a solution of polymers: resistance to a deforming flow compared to pure solvent → increase in viscosity
Dynamic viscosity
γ=shear rate [1/s]
σ=shear stress [Pa]
η=dynamic viscosity [Pa・s=N・s/m2]
Newtonian flow
• Shear stress proportional to shear rate → viscosity independent of shear rate.
• Low molar mass liquids (water, organic solvent etc.).
What happens viscosity from flow field is distrub?
Viscosity increase in dispersions arises from disturbances in the flow field
Non-Newtonian flow
• The viscosity is shear rate dependant.
• Pseudo-plastic flow: The viscosity decreases with increasing shear rate (as known as shear-thinning). For example paint
• Dilatent flow: The viscosity increases with increasing shear rate. For example wet sand (particle volume fraction increase locally)
• Apparent viscosity
Herschel-Bulkley flow
• Above the yield stress the system starts to flow.
• Below the yield stress the system appears solid (i.e. gel)
Example: Tooth paste, tomate ketchup
Overlap concentration (c*)
The point where solution viscosity increases sharply
with polymer concentration
• c* depends on the volume occupied by polymer in the solution.
– Large volume → lower c*
– Small volume → higher c*
• Note that the volume is 3D!
– i.e. rod-like conformation occupies a large volume
Relation to overlap concentration (c*) for different concentrations
c correlation to c*
Thixotropy
Thixotropic fluids are a type of non-Newtonian liquids that exhibit shear thinning during increased shear rate and a slow recovery to the original viscosity after the shear rate is removed.
• Time-dependent viscosity decrease during constant shear
• Obtained with associative thickeners – dissociation and orientation give a decrease in viscosity
• The viscosity is recovered when shearing/deformation stops
• Example: Important for controlling paint viscosity and application
Associative thickeners
Hydrophobic interaction can play a role in polymer network formation
Surfactant micelles can associate with hydrophobic groups