Raman Spectroscopy
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
14 Terms
Explain what effect changing the excitation laser wavelength will have on the Raman spectrum obtained.
Changing the excitation wavelength will have no effect on the Raman spectrum obtained
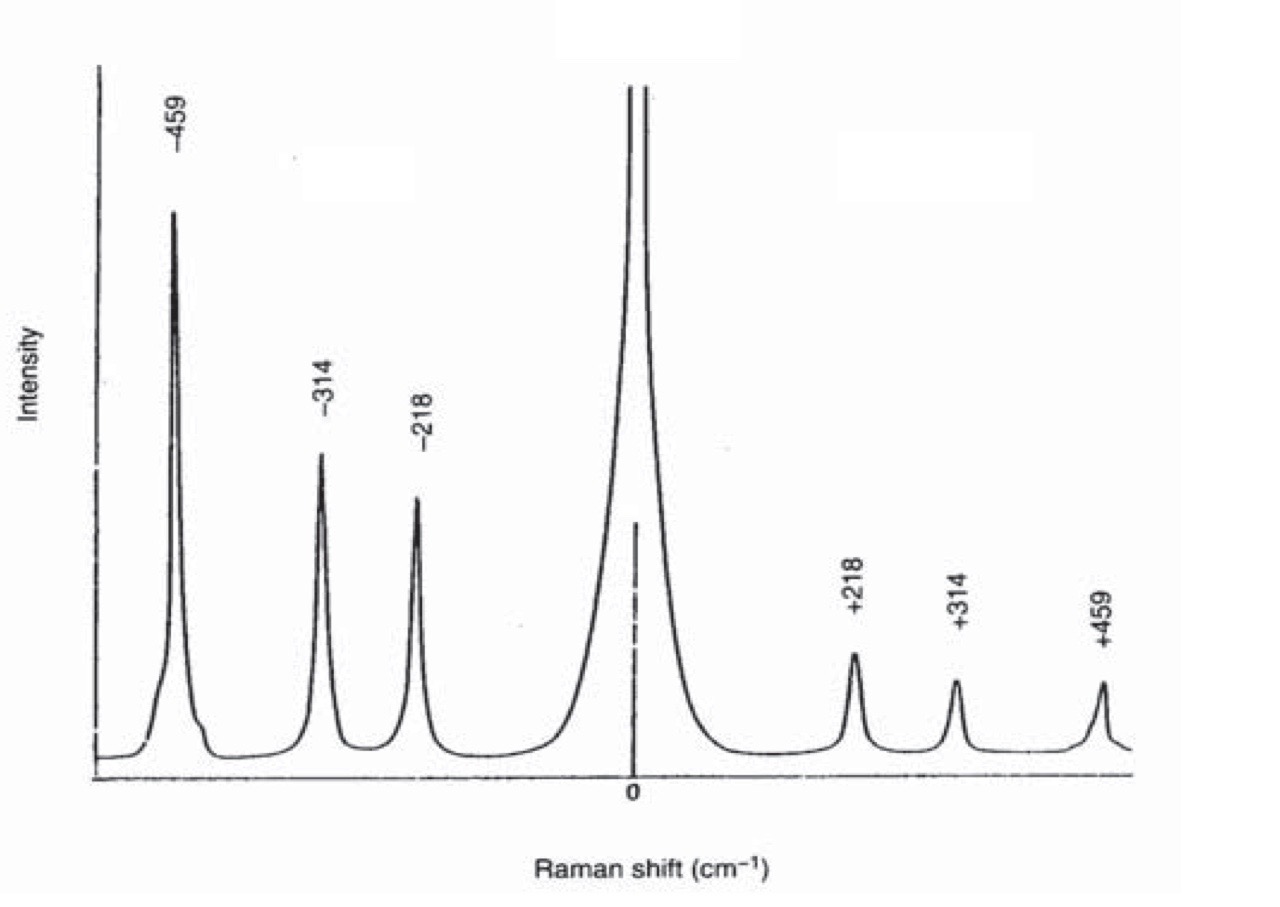
The figure below shows the Raman Spectrum for CCl4.obtained using laser excitation at 488nm.
Explain the transitions responsible for each peak observed.
Intense peak at 0cm-1 is due to Rayleigh scattering
Peaks at Raman shift of -218 cm-1, -324cm-1, -459 cm-1 are due to Stokes shift Peaks at Raman shift of +218 cm-1, +324cm-1, +459 cm-1 are due to anti-Stokes shift
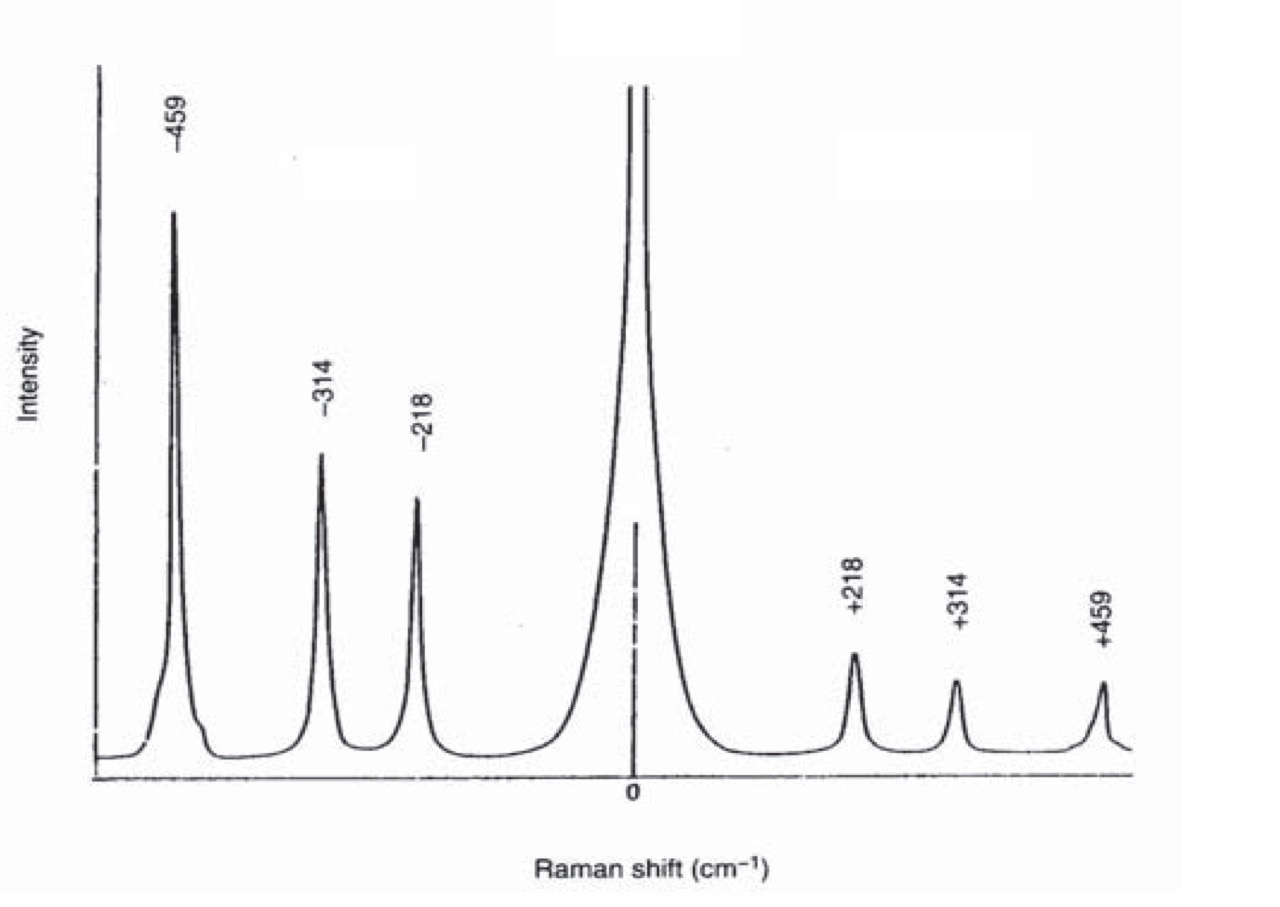
The figure below shows the Raman Spectrum for CCl4.obtained using laser excitation at 488nm.
Calculate the wavelengths and wavenumbers associated with these transitions.
λex = 488nm Wavenumber = 1/λex =1/488nm = 20 492 cm-1
Stokes Wavenumbers = 20 492 – 218 = 20274cm-1;
Following similar calculation the other lines are at: 20168 cm-1; 20033cm-1Which corresponds to wavelengths of 493nm; 496nm; 499nm
Anti-Stokes Wavenumbers = 20492 + 218 = 20710cm-1; and for the other lines. 20816cm-1; 20951cm-1
Which corresponds to wavelengths of 483nm; 480nm; 477nm
Explain why some compounds absorb infrared radiation, others are Raman active but these two phenomena are not observed within simple molecules.
IR transitions require change in dipole moment and Raman transitions require change in electron polarisation. In simple symmetrical molecules Raman will be observed for the symmetric stretch only and IR for all the other vibrational modes.
A Raman shift is seen associated with a relaxation of electrons to a less energetic state; will a Stokes line or an anti-Stokes line be observed?
Less energy → Longer λ → negative Raman shift, therefore Stokes Shift.
A Raman spectrum for a compound is obtained following excitation using 435.8 nm light. Raman lines are observed at 443 nm and 463 nm. Calculate the Raman shift for both these lines.
The IR spectrum for acetylene (C2H2) contains a strong absorbance at 3290 cm-1, but the Raman spectrum contains absorbance at 3370 cm-1 and 1970 cm-1.
a. What are the number of vibrational modes for acetylene?
b. Explain, in detail, the source of the differences in the IR and Raman spectra.
a. For linear molecules, number of types of vibrations: 3N-5 = 3(4) – 5 = 7

Under what circumstances would a helium/neon laser, λex = 633nm, P ~35mW, be preferable to an argon-ion laser, λex = 488nm, P ~ 1.5W, as a Raman source?
For studying samples where photodecomposition may be an issue and therefore a laser of lower power output is required. Also 633nm laser would be required if the sample under study was fluorescent at 488nm excitation.
Raman spectroscopy is increasing being used in identification of counterfeit pharmaceuticals. Discuss why Raman spectroscopy is becoming a popular tool for this application.
Little or no sample preparation required, can measure sample through cheap glass / plastic containers, water is weak scatterer, portable Raman instruments now available.
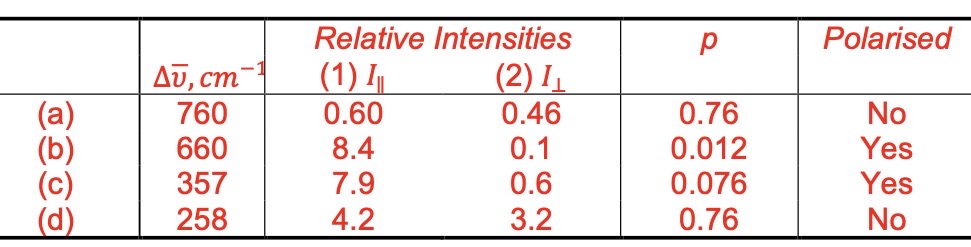
The following Raman data were obtained for CHCl3 with the polarizer of the spectrometer set (1) parallel to the plane of polarization of the laser and (2) at 90o to the plane of polarization of the source.
Calculate the depolarization ratio for each peak and indicate which Raman peaks are polarized.
Explain the meaning of the following terms commonly used in Raman spectroscopy:
Anti-Stokes shift, Polarisability Ellipsoid, Resonance Raman
Anti-Stokes Shift: Raman scattering resulting in higher energy scattered photons with respect to incident light.
Polarisability Ellipsoid: A 3-D surface where the distance from the centre of mass is proportional to 1 / √ai. aI is the polarizability of the molecule.
Resonance Raman: The Raman scattering signal observed when the molecule is promoted to the first excited singlet state.
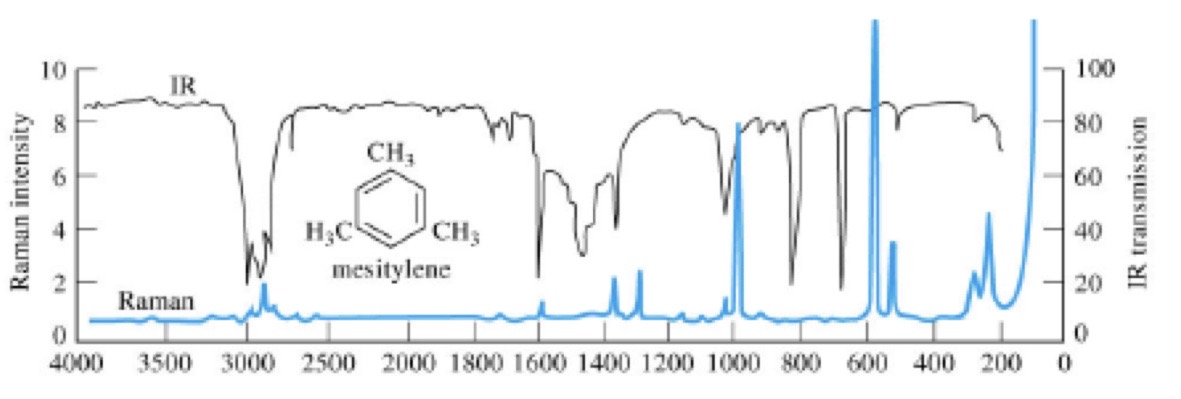
The FT-IR and Raman spectrum of Mesitylene is shown below. With reference to the chemical structure of Mesitylene discuss the similarities and differences between these two spectra.
Raman predominantly sensitive to changes in polarizability of molecule, hence intense peaks can be attributed to symmetric stretching of the ring structure, the C-C, C=C and CH3 bonds.
IR is sensitive to dipole moment changes, hence sensitive to antisymmetric stretching, bending and twisting of the molecule.
Because of the highly symmetric form of Mesitylene, peaks observed in Raman are generally not found in the IR spectrum or are extremely weak.
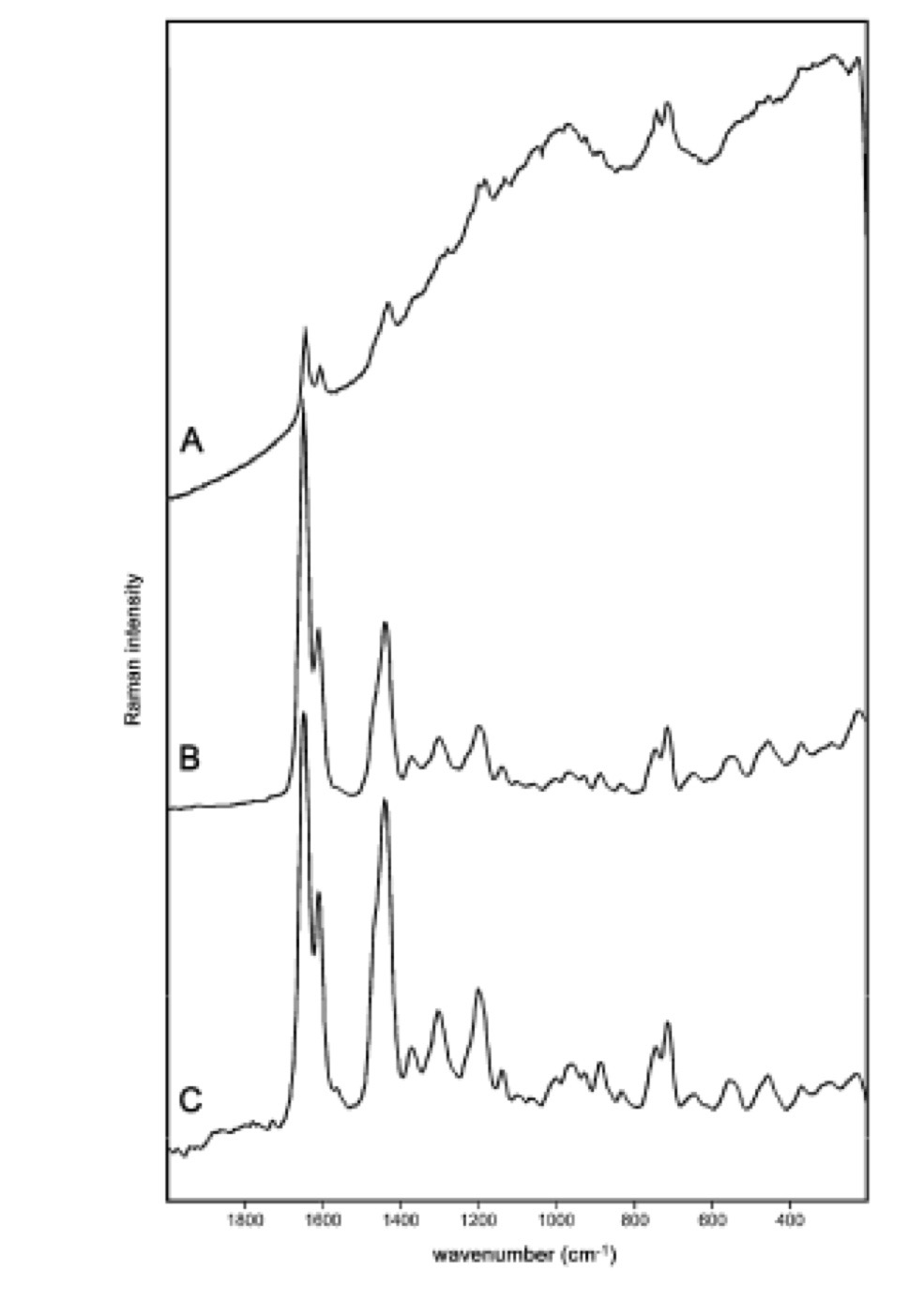
The Raman spectra given below for Baltic Amber have been collected with 3 different instruments, (A) and (B) with a bench top instruments equipped with a 785 nm and 1064 nm laser respectively and (C) a portable instrument employing a 1064 nm laser source.
i)Fully discuss why the two laser sources give rise to different spectral shapes.
785 nm source shows evidence of a fluorescence background.
1064nm is far enough into the IR part of the spectrum that excitation to the 1st excited state is not observed hence no fluorescence background.
ii) One of the C-H stretching modes is observed at 1450 cm-1. Determine the wavelength at which this peak is observed for both the 785 nm and 1064 nm laser source.
