electronegativity, dative and polar bonds
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
18 Terms
what is a covalent bond?
a shared pair of e-
atoms share e- to gain a full outer shell and increase stability
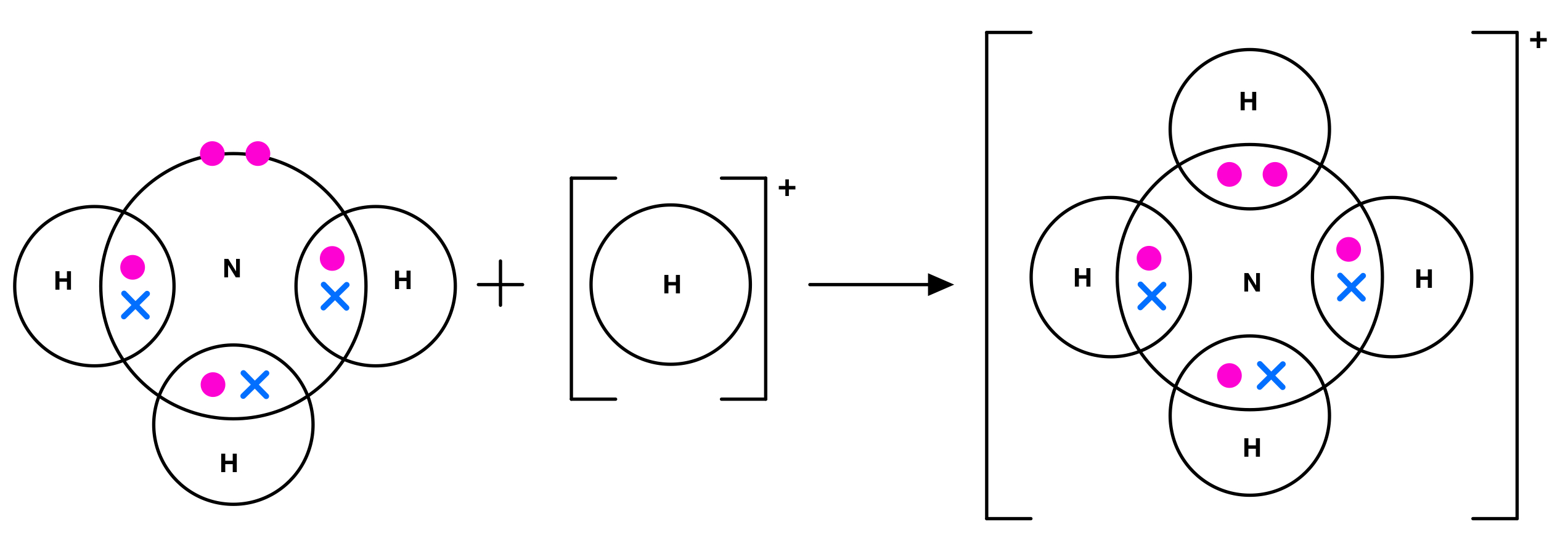
what is a dative covalent bond/coordinate bond?
both of the e- forming the covalent bond come from the same atom - they are donated
e.g. ammonium ion
what is electronegativity?
the tendency of an atom to attract a pair of e- in a covalent bond
name the 3 factors affecting electronegativity
nuclear charge
atomic radius
shielding
explain how nuclear charge affects electronegativity
the more protons, the stronger the attraction between nucleus and bonding pair of e-
therefore the more electronegative it is
explain how atomic radius affects electronegativity
the closer to the nucleus, the stronger the attraction between nucleus and bonding pair of e-
therefore more electronegative it is
explain how shielding affects electronegativity
the lower the number of shells of e- between nucleus and e-, the less shielding (less repulsion)
this means there is stronger attraction between nucleus and bonding pair of e-
therefore electronegativity increases
how does electronegativity change across the periodic table?
increases from left to right and bottom to top
(except noble gases - this means that F is the most electronegative element)
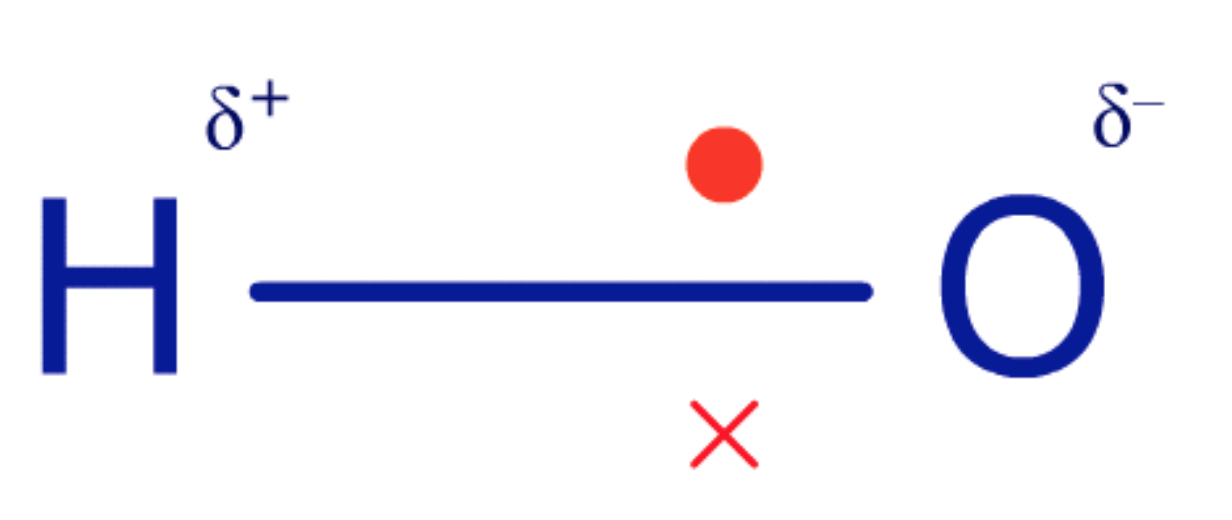
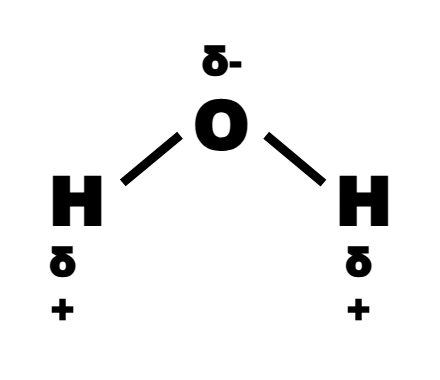
which atom is more electronegative and why? what does this mean?
O is more electronegative than H
this is because the e- are closer to the O atom
this creates a partial charge (shown by delta positive/negative)
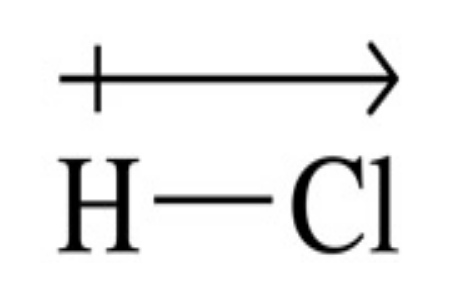
in this diagram, what does the arrow represent?
the arrow shows the direction of the dipole, w/ the arrow pointing towards the more -ve atom
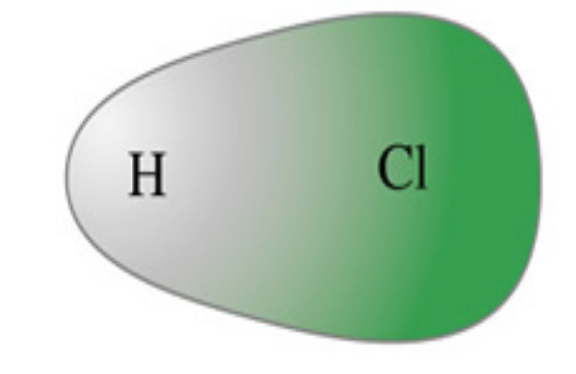
in this diagram, what does the cloud repesent?
the unequal sharing of a cloud of e- density
what does a significantly large difference in electronegativity cause?
ionic bonding! (or in extreme cases, the atoms just wouldn’t bond)
what causes polar bonding?
a large difference in electronegativity between the atoms in the covalent bond
what causes polar molecules?
when there are dipoles (due to polar bonds) in a molecule that don’t cancel out, e.g. H2O
can a molecule have polar bonds but not be polar?
yes - the dipoles must cancel each other out, e.g. CO2
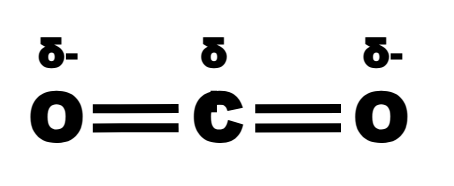
what is a bond dipole moment?
a measure of the strength and direction of the polarity in a bond
what does the bond dipole moment depend on?
the bigger the difference in electronegativity, the bigger the bond dipole moment
in organic molecules, which bonds are not regarded as being polar, despite the small difference in electronegativity between the atoms?
C-H bonds