Radical Reactions (CH 10)
1/6
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
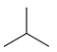
Bromination of Alkanes
Reagent:
Br2
hv (light)
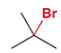
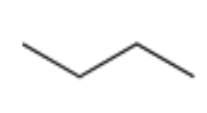
Chlorination of Alkanes (Halogenation That Creates a New Chiral Center)
Reagent:
Cl2
hv (light)
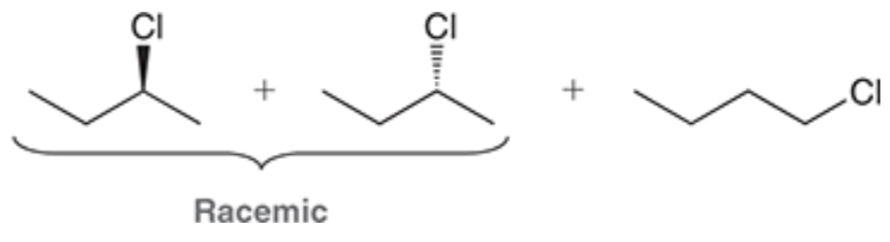
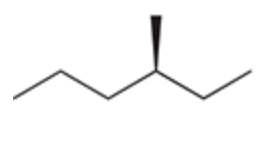
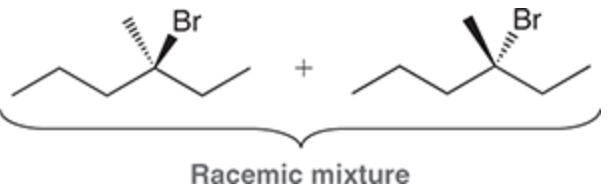
Halogenation at an Existing Chiral Center
Reagent:
Br2
hv (light)
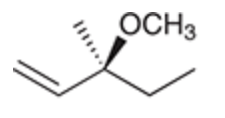
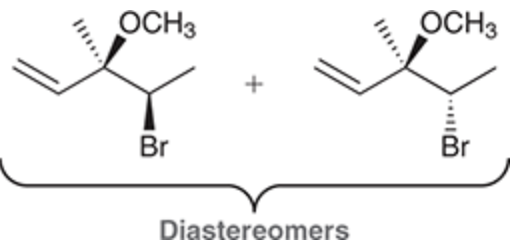
Formation of Diasteromers (use radical bromination)
Reagent:
Br2
hv
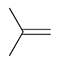
Anti-Markovnikov Addition of HBR to Alkenes
Reagent:
HBr
ROOR
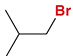
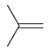
Allylic Bromination
Reagent:
NBS
hv (light)
Chapter 10 Splurge
SECTION 10.1
Radical mechanisms utilize fishhook arrows, each of which represents the flow of only one electron.
The order of stability for radicals follows the same trend exhibited by carbocations.
Allylic and benzylic radicals are resonance stabilized. Vinylic radicals are not.
SECTION 10.2
Radical mechanisms are characterized by six different kinds of steps: (1) homolytic cleavage, (2) addition to a π bond, (3) hydrogen abstraction, (4) halogen abstraction, (5) elimination, and (6) coupling.
Every step in a radical mechanism can be classified as initiation, propagation, or termination.
SECTION 10.3
Methane reacts with chlorine via a radical mechanism.
The sum of the two propagation steps gives the net chemical reaction. These steps together represent a chain reaction.
A radical initiator is a compound with a weak bond that readily undergoes homolytic bond cleavage. Examples include alkyl peroxides and acyl peroxides.
A radical inhibitor, also called a radical scavenger, is a compound that prevents a chain process from either getting started or continuing. Examples include molecular oxygen and hydroquinone.
SECTION 10.4
Only radical chlorination and radical bromination have practical use in the laboratory.
Bromination is generally a much slower process than chlorination.
SECTION 10.5
Halogenation occurs more readily at substituted positions. Bromination is more selective than chlorination.
SECTION 10.6
When a new chiral center is created during a radical halogenation process, both possible stereoisomers are obtained.
When a halogenation reaction takes place at a chiral center, a racemic mixture is obtained regardless of the configuration of the starting material.
SECTION 10.7
Alkenes can undergo allylic bromination, in which bromination occurs at the allylic position.
To avoid a competing ionic addition reaction, N-bromosuccinimide (NBS) can be used instead of Br2.
SECTION 10.8
Ozone is produced and destroyed by a radical process that shields the earth’s surface from harmful UV radiation.
A rapid decrease of stratospheric ozone is attributed to the use of CFCs, or chlorofluorocarbons, sold under the trade name Freons.
A ban on CFCs prompted a search for viable substitutes, such as hydrofluoroalkanes (HFAs), also called hydrofluorocarbons (HFCs).
SECTION 10.9
Organic compounds undergo oxidation in the presence of atmospheric oxygen to produce hydroperoxides. This process, called autooxidation, is believed to proceed via a radical mechanism.
Antioxidants, such as BHT and BHA, are used as food preservatives to prevent autooxidation of unsaturated oils.
Natural antioxidants prevent the oxidation of cell membranes and protect a variety of biologically important compounds. Vitamins E and C are natural antioxidants.
SECTION 10.10
Alkenes will react with HBr in the presence of peroxides to produce a radical addition reaction.
SECTION 10.11
Polymerization of ethylene via a radical process generally involves chain branching.
When vinyl chloride is polymerized, polyvinyl chloride (PVC) is obtained.
SECTION 10.12
Radical processes are used heavily in the chemical industry, particularly in the petrochemical industry. Examples include cracking and reforming. When cracking is performed in the presence of hydrogen, it is called hydrocracking.
SECTION 10.13
Radical halogenation provides a method for introducing functionality into an alkane.
When the starting compound has only one kind of hydrogen atom, chlorination can be used.
When different types of hydrogen atoms are present in the compound, it is best to use bromination in order to control the regiochemical outcome and avoid a mixture of products.